Clear Sky Science · en
Corilagin alleviates Staphylococcus aureus pathogenicity by interacting with amidase and α-hemolysin
Fighting Tough Germs in a New Way
Antibiotic-resistant “superbugs” like methicillin-resistant Staphylococcus aureus (MRSA) are a growing threat in hospitals, farms, and communities. Instead of trying to kill these bacteria outright—a tactic that often drives resistance—researchers are searching for treatments that disarm the germs so our bodies can clear them more easily. This study explores such an approach, showing how a plant-derived compound called corilagin can blunt the harmful powers of S. aureus, including a dangerous pneumonia-causing strain, without acting as a classic antibiotic.
A Plant Compound with Hidden Talent
Corilagin is a naturally occurring molecule found in several medicinal plants and already known for anti-inflammatory and liver-protective effects. Earlier hints suggested it could also weaken S. aureus, but how it worked was unclear. The authors focused on two bacterial tools that are central to infection. The first, amidase (amiA), helps bacteria divide and form protective biofilms—slimy communities stuck to tissues or medical implants. The second, alpha-hemolysin (Hla), is a toxin that punches holes in host cells, contributing to tissue damage and severe pneumonia. If corilagin could block both, it might offer a powerful “two-pronged” way to reduce disease.
Blocking Bacterial Growth and Sticky Biofilms
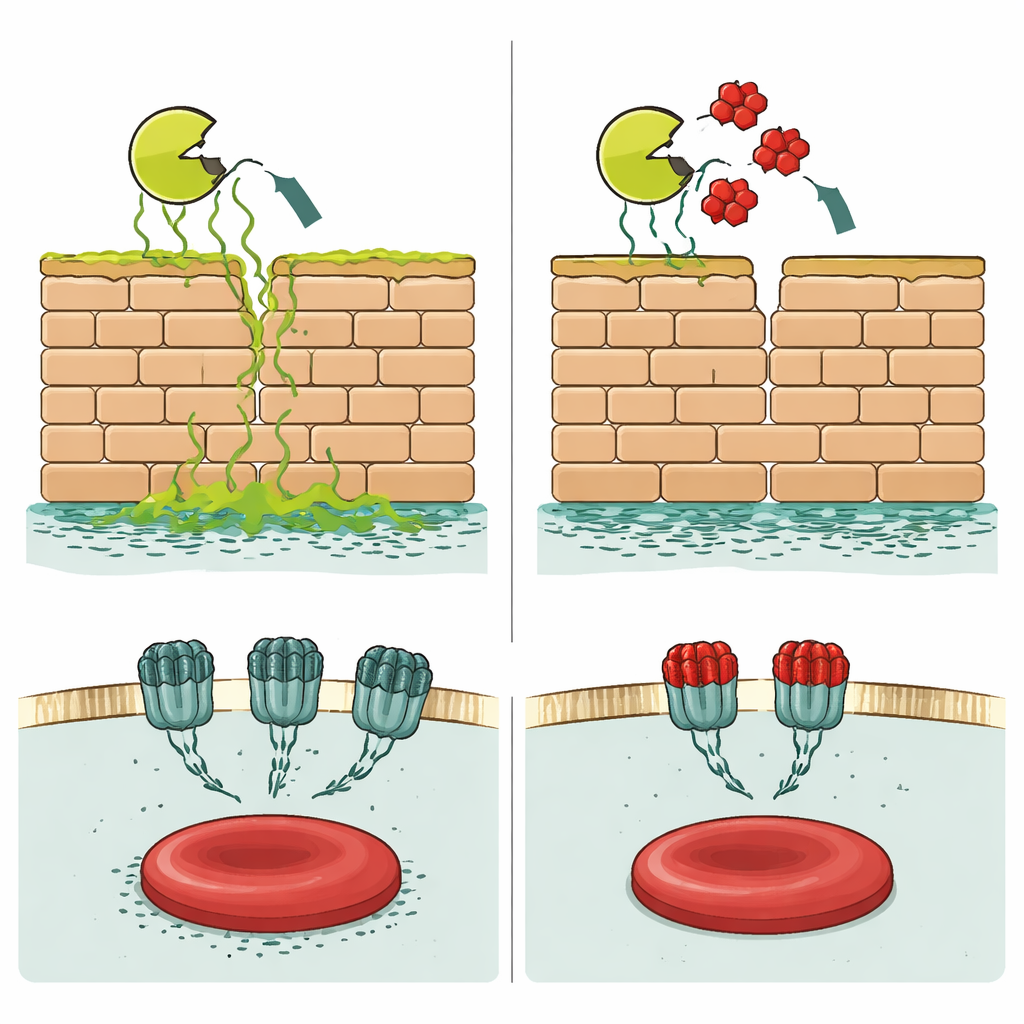
Using computer modeling, the team showed that corilagin fits snugly into the active pocket of amidase, contacting several key building-blocks of the protein. Laboratory experiments confirmed the functional impact. When MRSA strain USA300 grew in the presence of corilagin, its growth slowed even though the compound did not strongly kill the bacteria, indicating a targeted interference rather than outright poisoning. More strikingly, the bacteria formed far less biofilm—the dense, glue-like communities that protect them from antibiotics and immune attacks. With rising doses of corilagin, biofilm mass and the number of bacteria within these films dropped sharply. This suggests that by jamming amidase, corilagin disrupts cell separation and the early steps of surface attachment, leaving the microbes less able to entrench themselves.
Disarming a Potent Toxin
The second line of attack involves alpha-hemolysin, a pore-forming toxin that helps S. aureus destroy red blood cells and damage lung tissue. When the researchers grew USA300 with corilagin, the liquid surrounding the bacteria caused far less destruction of sheep red blood cells, showing that toxin activity was greatly reduced. Protein separation tests revealed that bacteria exposed to corilagin secreted less Hla. Even purified Hla became less damaging when mixed directly with corilagin, implying that the compound binds to the toxin itself. Simulations supported this, placing corilagin on a rim region of Hla that normally helps it latch onto cell membranes and form deadly pores.
From Cells and Insects to Infected Lungs
To see whether these molecular effects matter in living systems, the team tested corilagin in several models. In cultured mouse immune cells and human lung cells, S. aureus normally caused substantial cell death and clung tightly to cell surfaces. Adding corilagin greatly reduced both the toxic damage and the bacterial adhesion, yet the compound alone showed little harm to the cells. In mice infected in the lungs with the aggressive USA300 strain, animals treated with corilagin had fewer bacteria in their lung tissue, less swelling and fluid buildup, lower levels of inflammatory signaling molecules, and significantly better survival. A separate test in wax moth larvae, a common infection model, showed that corilagin protected the insects better than the standard antibiotic ampicillin, while also appearing safe across tested doses.
What This Means for Future Treatments
Instead of acting like a traditional antibiotic that aims to kill bacteria directly, corilagin works more like a scalpel that cuts away the germ’s most dangerous tools. By hindering amidase, it slows growth and prevents the formation of fortified biofilms, and by binding to alpha-hemolysin, it reduces the bacteria’s ability to rupture host cells and inflame tissues. In animal models, this strategy translates into milder lung disease and improved chances of survival. While further work is needed to refine dosing and delivery and to confirm safety in humans, the study highlights corilagin as a promising blueprint for next-generation anti-infection drugs that disable, rather than simply destroy, stubborn pathogens like MRSA.
Citation: Teng, F., Wen, T., Lu, J. et al. Corilagin alleviates Staphylococcus aureus pathogenicity by interacting with amidase and α-hemolysin. Sci Rep 16, 10829 (2026). https://doi.org/10.1038/s41598-026-44825-6
Keywords: Staphylococcus aureus, MRSA, corilagin, anti-virulence, biofilm