Clear Sky Science · en
Sustainable superhydrophobic coating based on in-situ electrodeposited Ni-Al layered double hydroxide for enhanced corrosion protection of steel
Keeping Metal Safe from Harsh Environments
From ships and bridges to pipelines and power plants, much of our modern world is built on steel—and that steel is constantly under attack from water and salt. Corrosion silently eats away at structures, costing billions and threatening safety. This study explores a new, greener way to armor steel with a water‑repelling skin that not only keeps it dry but also stays tough in rough, real‑world conditions.
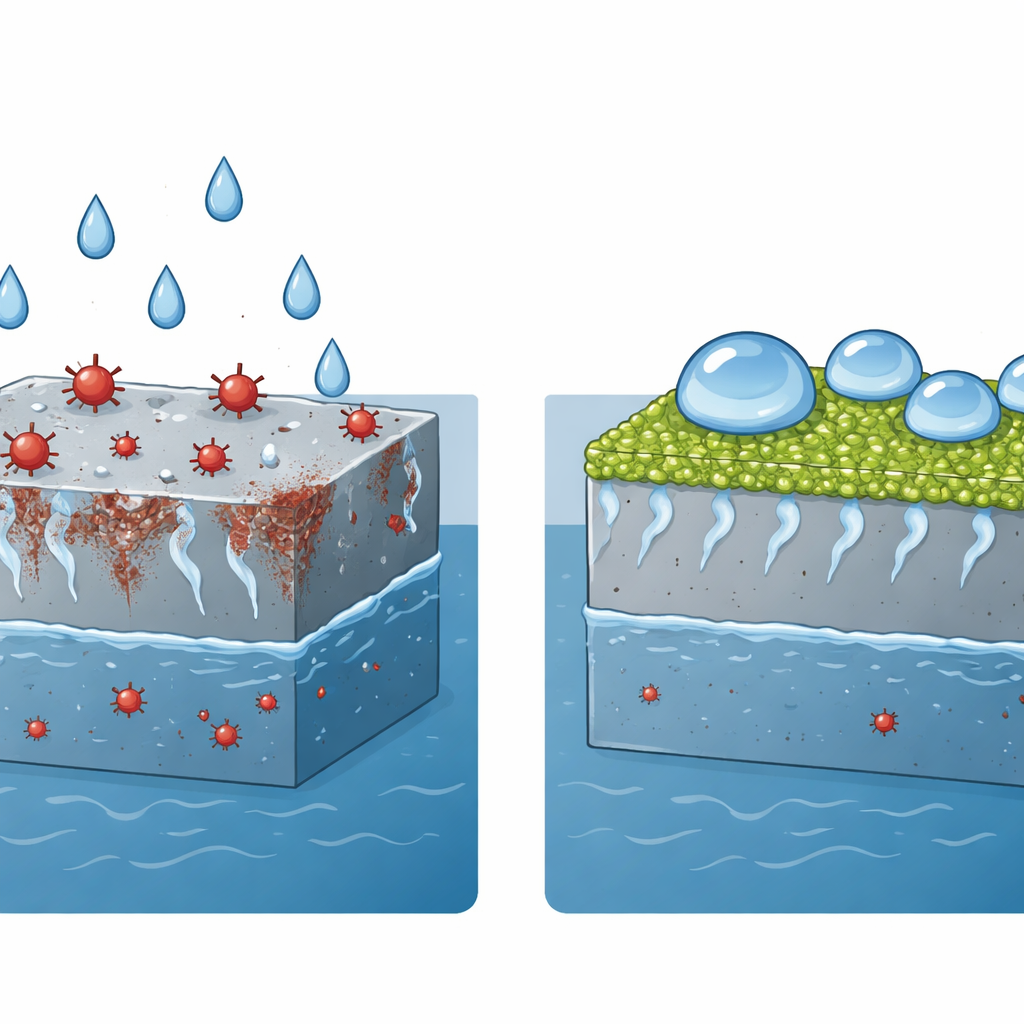
How a Water-Hating Skin Protects Steel
The researchers set out to make steel surfaces behave a bit like a lotus leaf: water droplets bead up and roll off instead of spreading and soaking in. Such "superhydrophobic" surfaces trap a thin layer of air between the metal and the liquid, acting like a microscopic raincoat that blocks salty water from reaching the steel underneath. The team aimed to combine this effect with a coating that is robust, long‑lasting, and environmentally friendlier than many existing water‑repellent treatments that rely on persistent fluorinated chemicals.
Building a Tiny Forest on Steel
To create this protective skin, the scientists used an electrochemical process that grows a special, layered material directly on the steel. This material, made of nickel and aluminum compounds, forms a dense forest of microscopic walls and needle‑like structures on the surface. Because the coating is grown in place rather than glued on, it bonds strongly to the metal. In a second step, they dipped the rough surface into a solution of stearic acid, a long‑chain fatty acid related to everyday substances found in plants and animals. This natural, low‑energy layer makes the already rough surface strongly repel water without resorting to fluorinated chemicals.
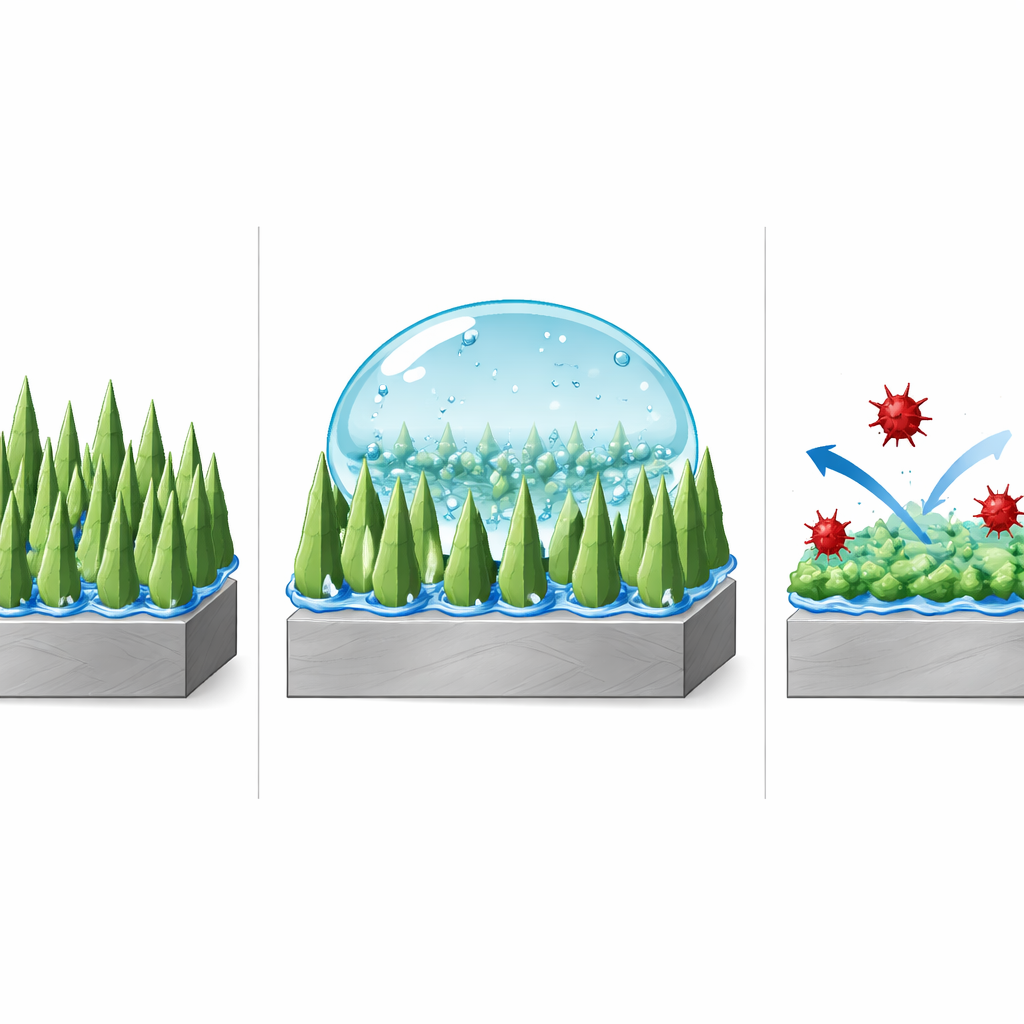
Finding the Sweet Spot for Maximum Protection
The team carefully tuned how long they ran the electrodeposition step, comparing coatings grown for 7.5, 15, and 22.5 minutes. They found that 15 minutes produced an especially effective "nano‑forest" of slender needles with very high surface roughness. Water droplets on this surface formed nearly perfect spheres with a contact angle of about 161 degrees and slid off with only a gentle tilt, showing extremely low sticking. Shorter growth times left the surface under‑developed, while longer times caused the tiny features to coarsen and smooth out, reducing the ability to trap air and weakening the water‑repellent effect.
Putting the Coating Through Tough Tests
To see how well this superhydrophobic skin could protect steel, the researchers exposed coated and uncoated samples to salty water similar to seawater and measured how easily corrosive reactions occurred. Electrochemical tests showed that the optimized coating drastically slowed the flow of corrosion‑related current, boosting protection efficiency to about 96.5 percent compared with bare steel. Just as important, the coating held up under stress: it kept its water‑repelling behavior after being rubbed over more than a meter of abrasive paper, and it stayed highly water‑repellent after immersion in liquids ranging from strongly acidic (pH 1) to strongly alkaline (pH 13). These results indicate that both the rough structure and the thin organic layer are chemically and mechanically resilient.
Why This Matters for Real-World Use
In simple terms, the study shows that steel can be given a tough, long‑lasting, and greener water‑repellent shield by growing a special microscopic structure directly on its surface and then coating it with a naturally derived fatty layer. When optimized, this combination traps air so effectively that salty, corrosive water struggles to touch the metal at all, greatly slowing down rust. Because the process is relatively simple, uses modest conditions, and avoids persistent fluorinated chemicals, it could be adapted to help protect infrastructure, marine equipment, and industrial hardware in demanding environments. While more work is needed to test long‑term weathering and large, complex shapes, this approach offers a promising new path toward sustainable, durable corrosion protection.
Citation: Ragheb, D.M., Zaki, M.M., Mahgoub, F.M. et al. Sustainable superhydrophobic coating based on in-situ electrodeposited Ni-Al layered double hydroxide for enhanced corrosion protection of steel. Sci Rep 16, 12184 (2026). https://doi.org/10.1038/s41598-026-44678-z
Keywords: superhydrophobic coating, corrosion protection, steel, electrodeposition, nickel aluminum layered double hydroxide