Clear Sky Science · en
Single cell profiling of ER stress in coronary artery disease and therapeutic mechanisms of Ginkgo biloba extract
Why heart stress matters
Heart disease remains the world’s top killer, yet many of the processes that quietly damage our coronary arteries unfold long before chest pain or a heart attack. This study looks inside blood and heart cells to ask a simple but powerful question: when the cell’s protein‑making machinery is under stress, does that push coronary artery disease forward—and can a natural extract from Ginkgo biloba leaves help ease that stress? By combining big genetic datasets, single‑cell analysis, and lab experiments, the researchers map a new layer of biology that could someday refine diagnosis and point toward gentler, more targeted treatments.
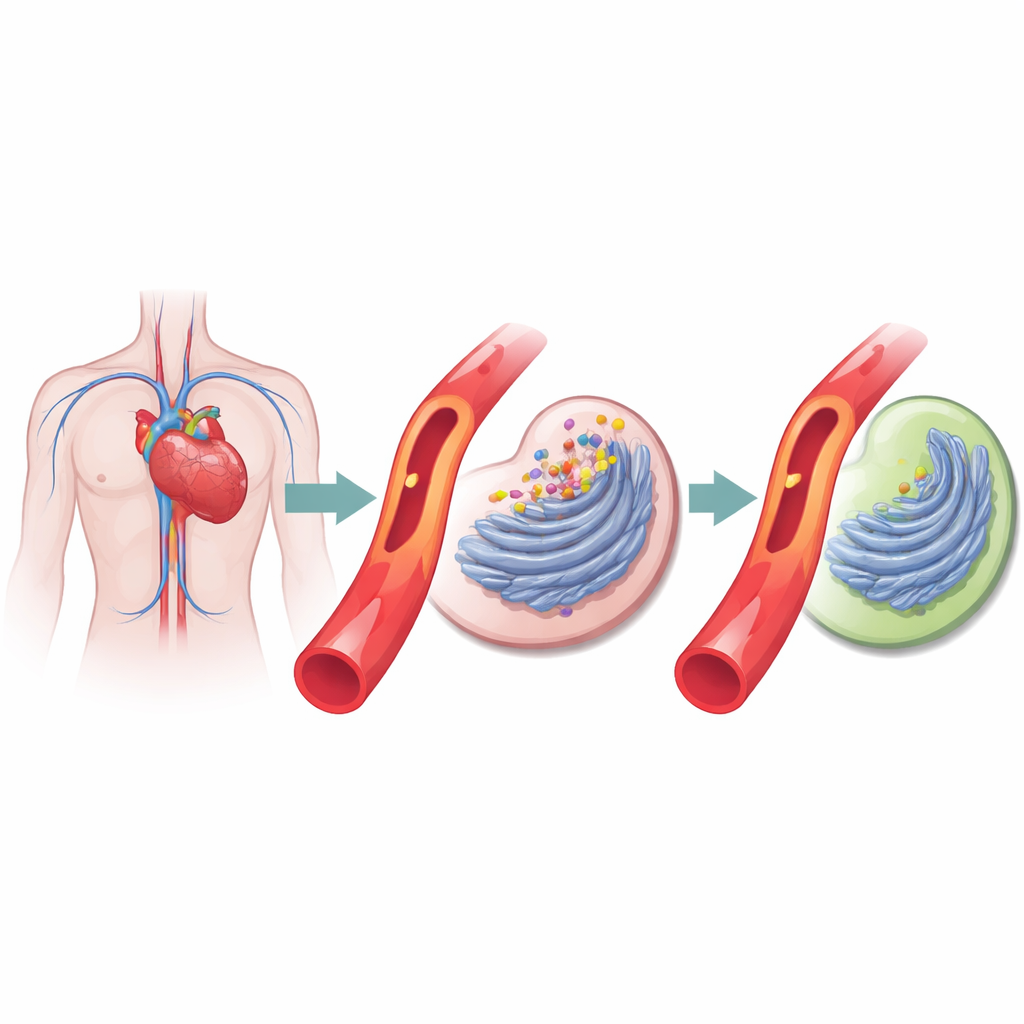
Inside the heart’s clogged pipes
Coronary artery disease arises when fatty, inflamed plaques narrow the vessels that feed the heart. The authors focused on a process called cellular stress in the protein factory of the cell, a folded membrane system where proteins are built and shaped. When this system is overwhelmed, damaged proteins pile up and a stress response switches on. Earlier work hinted that this response is active in diseased arteries. Here, the team pulled together gene‑activity data from hundreds of people with and without coronary disease, drawn from several international studies, and asked which genes differ consistently between patients and healthy volunteers.
Finding stress signals in the blood
Across the datasets, the researchers found 833 genes whose activity changed in people with coronary artery disease. Many of these genes were linked to inflammation and immune attack, especially involving neutrophils, a type of white blood cell that can damage vessel walls as it fights infection. Pathway analyses showed that stress‑related cleanup systems and inflammatory signaling routes were strongly switched on in patients. By overlaying these changes with a curated list of genes known to respond to protein‑factory stress, and then examining how these genes interact with each other, the team distilled a set of 10 core "stress‑related" genes that best marked the presence of disease.
Zooming in on key cell types
Blood is a mixture of many cell types, so the authors turned to single‑cell sequencing, which reads out gene activity one cell at a time. This allowed them to see exactly which cells carried the stress signatures. They identified five major cell populations and found that the stress‑related genes clustered mainly in monocytes and macrophages—immune cells that infiltrate vessel walls and help form unstable plaques. Several of these genes also showed a tight relationship with central stress‑response switches inside the protein factory, reinforcing the idea that they are not just bystanders but part of a coordinated damage program active in coronary disease.
From gene maps to risk scores and remedies
To test whether these signals might help in the clinic, the researchers built a simple gene‑based risk score using five of the most informative genes. In independent data, this score could distinguish people with coronary disease from controls with reasonable accuracy, hinting at a potential blood‑based aid for earlier risk assessment. Next, they searched drug‑gene databases for compounds that might modulate the stress‑linked genes and highlighted 177 candidates. Among these, ingredients from Ginkgo biloba emerged as especially promising. In heart muscle cells grown under low‑oxygen conditions, treatment with a Ginkgo extract reduced the levels of classic stress markers, and this benefit faded when stress was artificially forced back up—evidence that the extract’s protective effect depends on easing cellular stress.
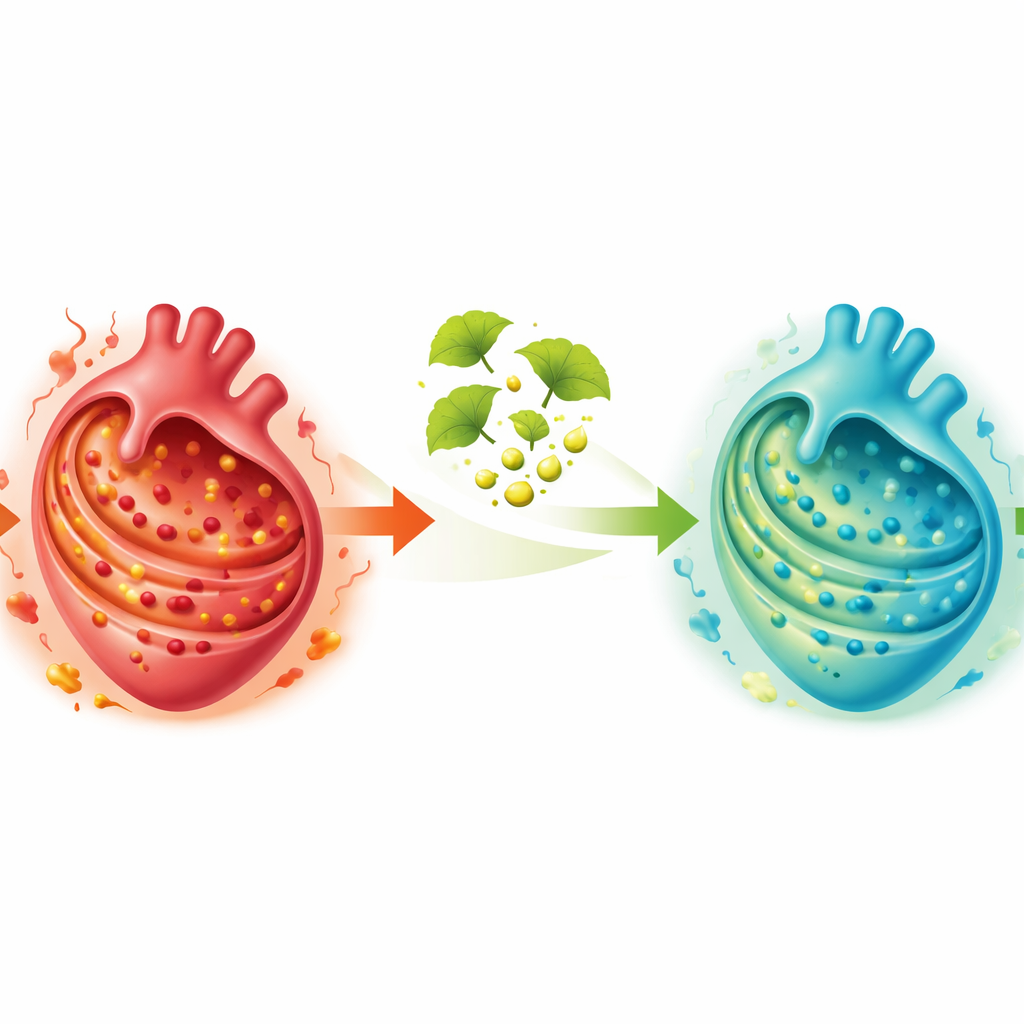
What this means for patients
Viewed together, the work supports a picture in which ongoing stress inside the cell’s protein factory helps drive inflammation and damage in the coronary arteries, particularly through specific immune cells circulating in the blood and entering vessel walls. The authors show that a small panel of stress‑linked genes can flag disease risk and that Ginkgo biloba extract can dial down stress signals in heart cells in the lab. While much more testing is needed before such findings reshape treatment, this integrated approach—from big‑data gene mining to single‑cell mapping and natural compounds—offers a fresh route toward more personalized diagnosis and new therapies that calm the heart from the inside out.
Citation: Zhao, Fj., Wang, F., Qin, C. et al. Single cell profiling of ER stress in coronary artery disease and therapeutic mechanisms of Ginkgo biloba extract. Sci Rep 16, 14508 (2026). https://doi.org/10.1038/s41598-026-44541-1
Keywords: coronary artery disease, cellular stress, immune cells, single-cell RNA sequencing, Ginkgo biloba extract