Clear Sky Science · en
Metabolic reprogramming of endothelial-related pathways in COVID-19 patients treated with hyperbaric oxygen therapy: a randomized clinical trial
Why oxygen under pressure matters
COVID-19 is often thought of as a lung infection, but doctors now know it can disrupt the whole body, damaging blood vessels and throwing basic chemistry inside our cells out of balance. This study asked a simple but important question: if we give hospitalized COVID-19 patients carefully controlled doses of pure oxygen in a pressure chamber—an approach called hyperbaric oxygen therapy—does it nudge their internal chemistry in a healthier direction? By tracking tiny molecules in the blood over time, the researchers looked for clues that could explain how this treatment might help the body cope with severe infection.
Looking inside the body’s chemical traffic
To explore this, the team ran a randomized clinical trial in a Polish hospital. Thirty adults hospitalized with COVID-19 pneumonia were enrolled; 28 were included in the final chemical analysis, with half assigned to standard care and half to receive five sessions of hyperbaric oxygen over the first five days. During each session, patients breathed 100% oxygen at more than twice normal atmospheric pressure inside a special chamber. Blood samples were collected at the start, after the first session, and again on days five and ten. Using high-resolution mass spectrometry, the scientists scanned these samples for hundreds of small molecules, then focused on 42 high-quality candidates and followed how their levels rose or fell over time.
Shifts in key building blocks and cell membranes
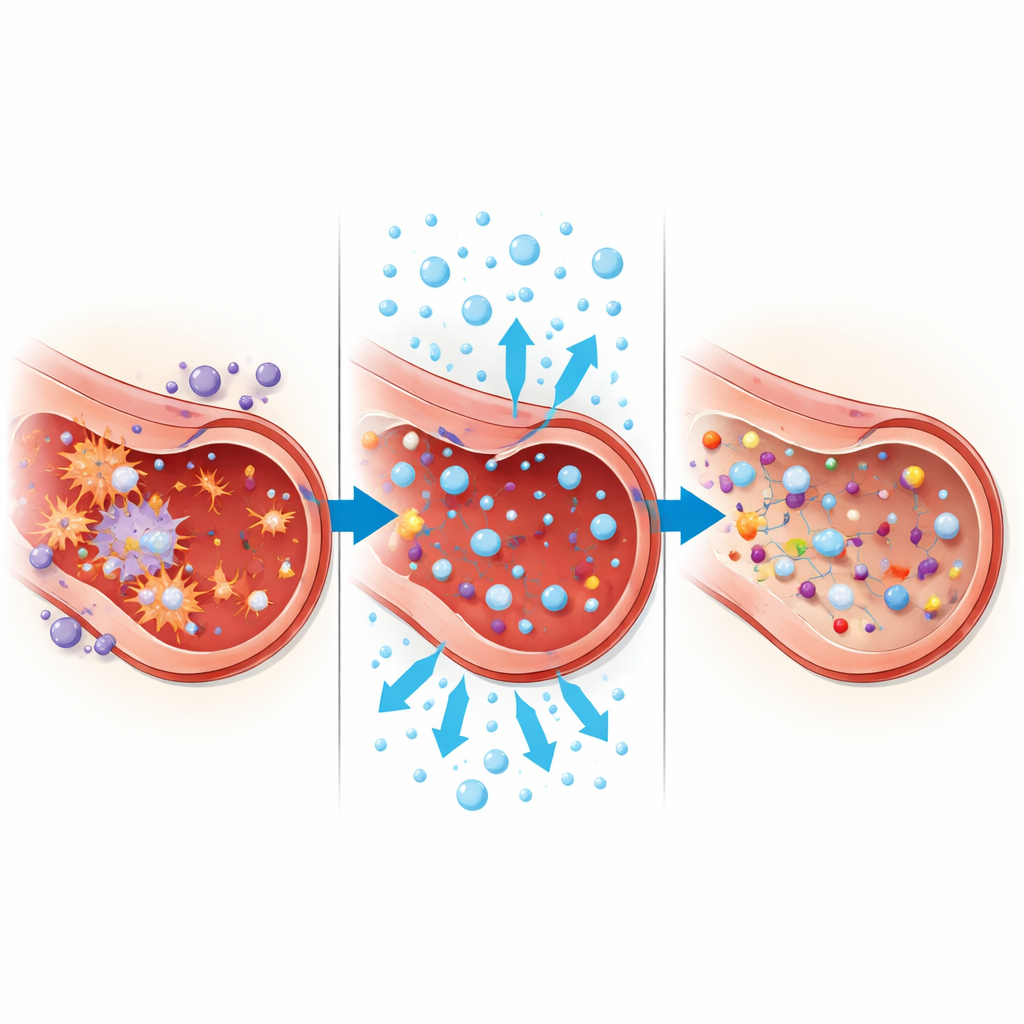
The analysis revealed ten molecules whose trajectories clearly differed between the hyperbaric and control groups. Several were related to arginine, an amino acid that fuels the production of nitric oxide, a gas crucial for keeping blood vessels flexible and healthy. In patients receiving hyperbaric oxygen, levels of arginine and a close cousin called homoarginine tended to fall, whereas they drifted upward in the control group. At the same time, molecules linked to cell membranes—certain phospholipids—and to choline metabolism changed, hinting that the outer shells of cells and the signaling fats embedded in them were being remodeled under the influence of the treatment. These changes do not yet translate directly into “better” or “worse,” but they flag biological systems that are being actively adjusted during therapy.
Stress signals and energy helpers reorganize
Two other groups of molecules told an equally intriguing story. Compounds called pterins, which are connected to folate chemistry and to the handling of oxidative stress, rose markedly in the hyperbaric group. Another marker, creatine riboside, related to the creatine system that helps cells manage bursts of energy demand, also increased. Meanwhile, intermediates in creatine production and the amino acid threonine showed distinct ups and downs. When the researchers examined how these molecules moved together rather than in isolation, they found that patients receiving hyperbaric oxygen developed a much denser web of connections among them. Pathway analysis pointed toward coordinated shifts in arginine and proline handling, choline and phospholipid metabolism, niacin-related energy chemistry, and folate–pterin routes tied to redox balance.
A more tightly coordinated chemical network
By treating the molecules as points in a network and looking at the strength of the links between them, the team saw that hyperbaric oxygen therapy led to a more interconnected chemical landscape. In these patients, amino acids, lipids, and redox-related compounds formed clusters that were more strongly synchronized than in the control group, where the network was sparser and more fragmented. This suggests that the treatment does not simply raise or lower a few markers; it appears to reorganize how multiple pathways—those governing blood vessel function, cell membranes, and energy use—talk to one another under the stress of infection and high oxygen exposure.
What this could mean for patients
For non-specialists, the upshot is that breathing oxygen under pressure during acute COVID-19 seems to push the body into a new, more coordinated chemical state, especially in systems that influence blood vessels and inflammation. The study does not prove that these shifts directly improve survival or long-term recovery, and the patient group was relatively small with no healthy comparison subjects. Still, the findings offer a biochemical map of how hyperbaric oxygen might help stabilize damaged vessels, adjust energy use, and fine-tune inflammatory responses. Larger, outcome-focused trials will be needed, but this work lays essential groundwork for understanding how a long-standing therapy for diving accidents and wound healing could also reshape the internal chemistry of patients battling a severe viral illness.
Citation: Jermakow, N., Brodaczewska, K., Kot, J. et al. Metabolic reprogramming of endothelial-related pathways in COVID-19 patients treated with hyperbaric oxygen therapy: a randomized clinical trial. Sci Rep 16, 13999 (2026). https://doi.org/10.1038/s41598-026-44520-6
Keywords: hyperbaric oxygen therapy, COVID-19 metabolism, endothelial health, amino acid pathways, oxidative stress