Clear Sky Science · en
Design and implementation of Cs/GO/TiO2 nanocomposite for controlling sulfamethoxazole
Why this matters for our water
Traces of medicines are now routinely found in rivers, lakes, and even drinking water. One common antibiotic, sulfamethoxazole, is especially stubborn: it passes through typical treatment plants and lingers in the environment, where it can harm aquatic life and encourage drug‑resistant bacteria. This study explores, at the scale of atoms and electrons, how a specially designed nanomaterial made from chitosan (a sugar from shellfish), graphene oxide (a sheet‑like carbon material), and titanium dioxide might grab and hold onto sulfamethoxazole, helping to clean contaminated water.

A new cleanup helper made from familiar parts
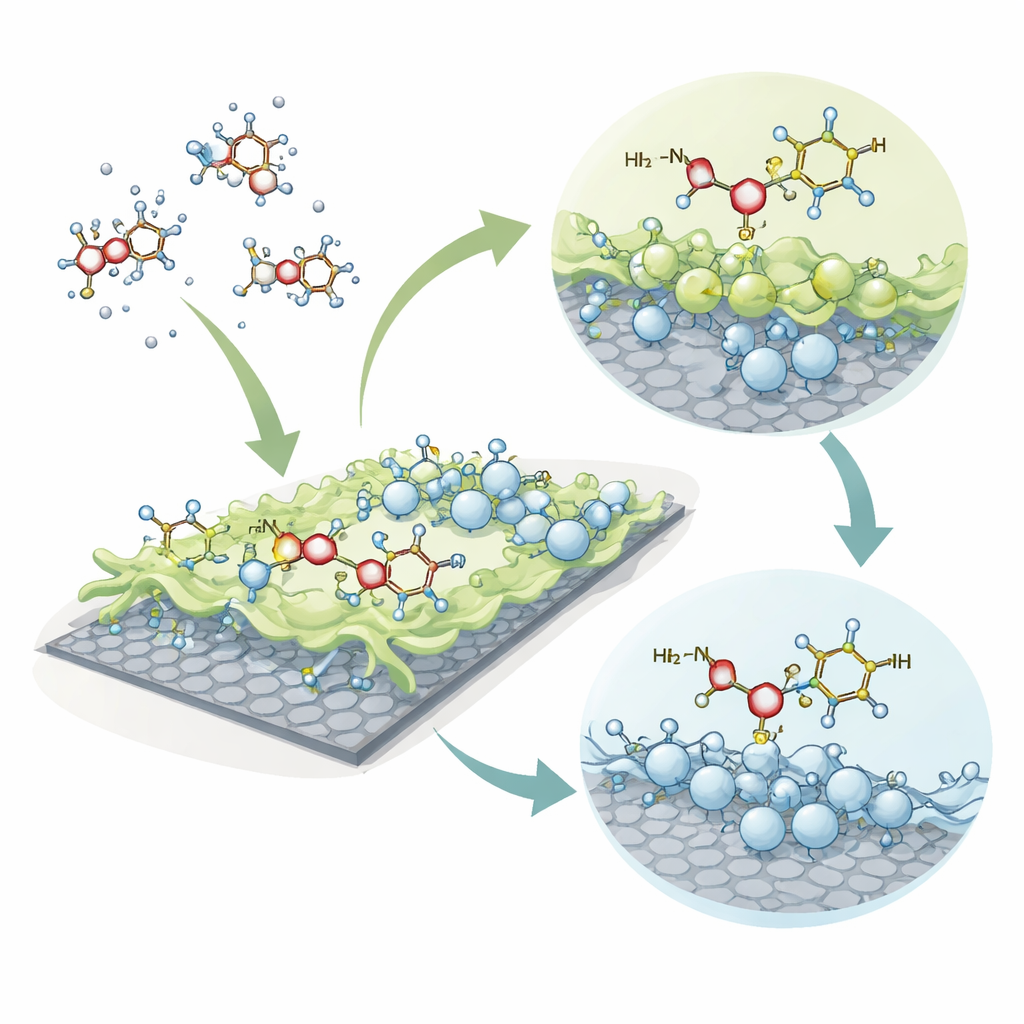
The researchers focus on a three‑component material that combines the strengths of its building blocks. Chitosan is biodegradable and naturally sticks to many pollutants. Graphene oxide is a thin, sturdy carbon sheet with a large surface and many oxygen‑bearing groups that can anchor molecules. Titanium dioxide is a well‑known photocatalyst that can help break down organic contaminants under light. When these three are joined into a single composite, they offer many different spots where antibiotic molecules can attach, and they may even support their eventual breakdown.
Looking at pollution control atom by atom
Instead of running lab filtration tests, the team used advanced computer simulations known as density functional theory. These calculations follow how electrons are arranged in the composite and in the antibiotic, and how that arrangement changes when they come together. The authors built detailed molecular models of the composite and a hydrated sulfamethoxazole molecule surrounded by a few water molecules, then tested two key ways the drug could attach: by interacting with the amine group on chitosan, or by forming a coordination bond between a nitrogen atom in the drug and a titanium atom in the titanium dioxide portion.
Two main docking spots for the antibiotic
The simulations show that both attachment routes are energetically favorable, meaning the antibiotic naturally "wants" to stick to the composite surface rather than remain free in water. When sulfamethoxazole binds through chitosan, the overall electrical polarity of the system increases and the energy gap between its filled and empty electron states shrinks. This pattern points to strong charge rearrangement and chemical reactivity, potentially useful for later light‑driven breakdown. When the antibiotic binds directly to titanium, the calculated binding energy is even stronger, signaling a very stable complex where the nitrogen in the drug coordinates closely to the metal center.
How electron shifts stabilize the capture
To understand why the bonding is so strong, the authors examined several electronic fingerprints. Maps of electrostatic potential highlight electron‑rich and electron‑poor patches, showing where attraction is likely. Charge‑distribution analyses reveal that, upon binding, electrons shift from the antibiotic toward titanium sites or around the chitosan groups, confirming significant charge transfer. Additional tools that track the density of available electron states and the nature of bonds indicate that the interaction is not merely a loose physical sticking, but involves a mix of coordination bonds, hydrogen bonds, and gentle van der Waals forces spread across the interface. Together, these effects lock the antibiotic into place on the composite surface.
What this means for cleaner water
Overall, the study shows that a chitosan/graphene oxide/titanium dioxide composite can grab sulfamethoxazole strongly at specific atomic sites, especially through titanium and certain chitosan groups. While the work is theoretical and based on simplified models of the material and its surroundings, it explains why such composites perform well in experiments and how their design could be tuned for even better removal. For non‑specialists, the key takeaway is that by understanding exactly how an antibiotic molecule latches onto a cleanup material, scientists can engineer smarter, more efficient filters and catalysts to help keep traces of pharmaceuticals out of our water.
Citation: Amin, K.S., Ghanem, M.S., Mahmoud, M.M. et al. Design and implementation of Cs/GO/TiO2 nanocomposite for controlling sulfamethoxazole. Sci Rep 16, 12033 (2026). https://doi.org/10.1038/s41598-026-44482-9
Keywords: antibiotic removal, water treatment, nanocomposite adsorbent, sulfamethoxazole, density functional theory