Clear Sky Science · en
Topotactic engineering of high-entropy (oxy) hydroxide nanotubes for enhanced photocatalysis
Cleaning Water with Tiny Tubes
Antibiotic residues in rivers and lakes are a growing concern because they can harm aquatic life and promote drug‑resistant bacteria. This study explores a new kind of ultra‑small tube, built from a mix of five metals, that can break down the antibiotic ciprofloxacin in water when exposed to ultraviolet (UV) light. By carefully guiding how the material forms at the atomic level, the researchers created a highly efficient "self‑cleaning" surface that could help treat contaminated water.
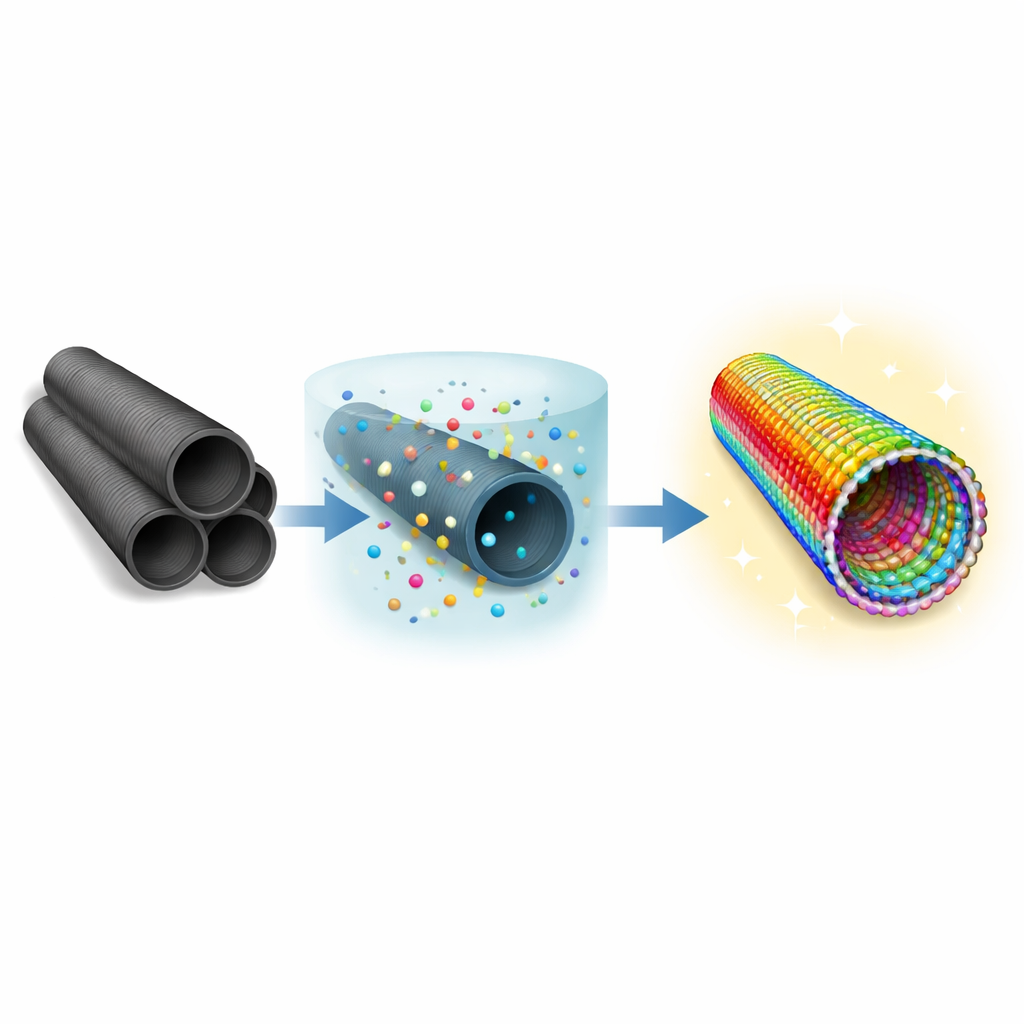
Turning Carbon Tubes into Metal Tubes
The researchers started from multiwalled carbon nanotubes—microscopic hollow fibers made of rolled‑up graphene sheets. Instead of building a new structure from scratch, they used these tubes as a scaffold and gradually replaced their interior with a new material through a process called topotactic transformation. In this solid‑state route, the original shape and orientation of the carbon tubes are largely preserved while their internal chemistry changes. Metal ions of cerium, cobalt, nickel, aluminum, and gallium were inserted between the carbon layers and, under mild heating, re‑organized into a new multiwalled structure made of metal (oxy)hydroxides.
Building a High‑Entropy Shell
The resulting tubes are called high‑entropy oxyhydroxide nanotubes, meaning that many different metal elements share the same crystal framework. Here, a distorted structure related to the mineral fluorite is stabilized by cerium and crowded with other metals. Because these metals carry different charges and sizes, the crystal naturally develops many missing‑oxygen sites and surface hydroxyl (OH) groups. Detailed measurements using X‑ray diffraction, Raman spectroscopy, electron microscopy, and photoelectron spectroscopy showed that the tubes have concentric walls, a fluorite‑like atomic arrangement, and a high concentration of structural defects that are spread evenly through the material.
Tuning the Tubes with Heat
The team then gently heated the nanotubes from 80 to 600 °C to see how their structure and activity evolved. Up to about 500 °C, the overall tubular shape and fluorite‑like phase remained intact, while water‑related groups were gradually removed in a process called dehydroxylation. This heating step increased the number of oxygen vacancies and rearranged the charges among the metals, creating a more interconnected, disordered network typical of high‑entropy oxides. However, at the highest temperature the tubes began to collapse into clumps of particles and a secondary phase appeared, showing that too much heat undermines the carefully engineered structure.
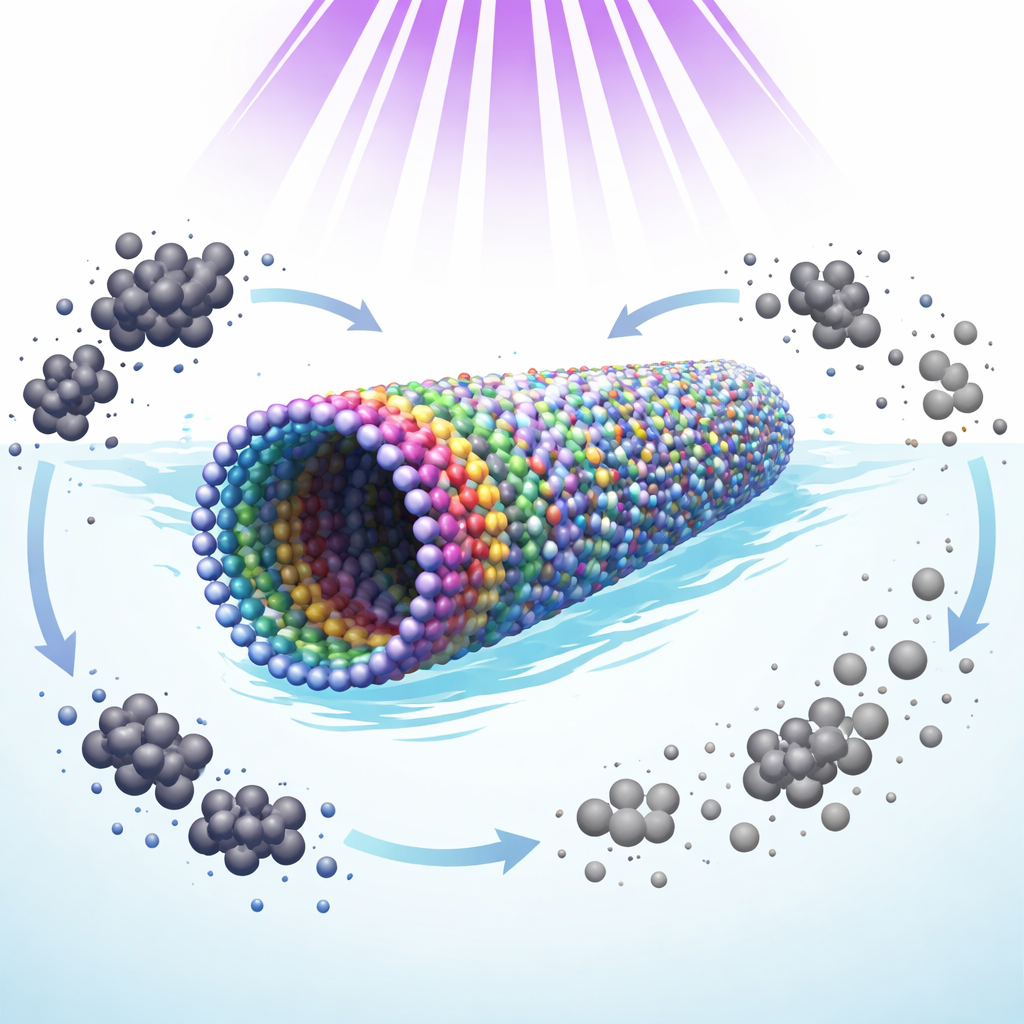
How the Tubes Break Down Antibiotics
To test their practical value, the researchers used the nanotubes to degrade ciprofloxacin in water under UV light. The best performer was the material treated only at 80 °C, which removed 96% of the antibiotic in just 45 minutes and followed a fast surface‑controlled reaction. Chemical "scavenger" tests revealed that positively charged holes generated in the material under light are the main agents attacking the antibiotic, while free radicals and electrons play secondary roles. The 80 °C sample has many surface hydroxyl groups that act as active sites: they help capture these holes, pass charges to the surrounding water, and form highly reactive species right where ciprofloxacin molecules are adsorbed. Hotter‑treated samples had fewer hydroxyls and more deep defects that trap charges without using them, which reduced their cleaning efficiency.
Why This Matters for Cleaner Water
Overall, the study shows that carefully reshaping carbon nanotubes into multi‑metal oxyhydroxide tubes, while preserving their form, creates a powerful photocatalyst for breaking down stubborn antibiotics in water. The key lies in balancing structural order and disorder: enough defects and hydroxyl groups to boost reactivity, but not so many that they waste charges. This topotactic approach offers a precise way to design next‑generation water‑cleaning materials by controlling both the shape and electronic structure of high‑entropy compounds.
Citation: Pacheco-Espinoza, S., Hernández-Pérez, M.Á., Cuesta-Balderas, A.I. et al. Topotactic engineering of high-entropy (oxy) hydroxide nanotubes for enhanced photocatalysis. Sci Rep 16, 14136 (2026). https://doi.org/10.1038/s41598-026-44418-3
Keywords: photocatalysis, water purification, nanotubes, high-entropy oxides, ciprofloxacin