Clear Sky Science · en
Enhancement of CO2 capture in post combustion process using actived carbon modified by amino acids
Cleaning the Air After Fuel Is Burned
Carbon dioxide from power plants is a major driver of global warming, yet most of our electricity still comes from burning fossil fuels. One practical way to cut these emissions is to trap CO2 from the hot exhaust gases before they escape into the sky. This study explores how to make a common, relatively cheap material—activated carbon—better at grabbing CO2 by gently coating its surface with simple building blocks of proteins called amino acids.
Turning Everyday Carbon into a Better Sponge
Activated carbon is already used in water filters and air purifiers because it is full of tiny pores that act like a sponge for molecules. For post‑combustion capture in power plants, an ideal material must hold a lot of CO2 even when it is present at low levels and at elevated temperatures, all while remaining stable and easy to reuse. The researchers started with commercial activated carbons and compared their natural ability to adsorb CO2—"adsorb" here meaning molecules sticking to the surface rather than reacting chemically. They confirmed that at room temperature the material can hold several millimoles of CO2 per gram, but this capacity drops sharply as the gas gets hotter, a typical signature of physical sticking rather than strong chemical bonding.
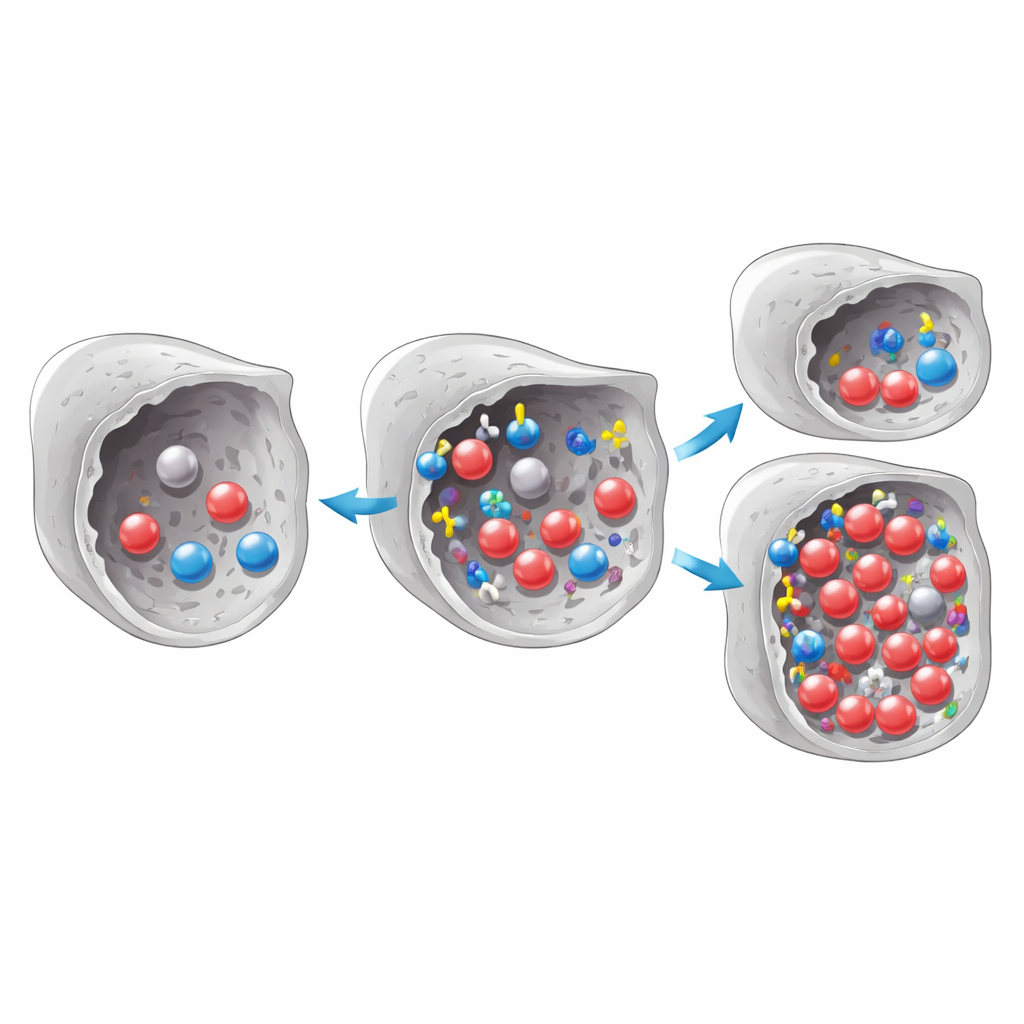
Adding Gentle Grips for Carbon Dioxide
To boost performance, the team modified the carbon surface using three amino acids: glycine, serine, and lysine. These are small organic molecules rich in nitrogen, a type of atom known to interact more strongly with CO2. The activated carbon was soaked in solutions containing each amino acid, sometimes together with simple alkaline salts, then washed and dried. When the treated samples were tested, those coated with glycine and serine generally captured more CO2 than the original material, while lysine often made things worse, especially when combined with added salts. Glycine stood out: despite being the smallest of the three, it increased CO2 uptake by up to about 25 percent without sacrificing the material’s ability to take up nitrogen, a key background gas in flue streams.
Finding the Sweet Spot in Treatment Conditions
Because overloading the carbon with modifiers can clog its pores, the researchers carefully varied the treatment temperature, time, and glycine concentration, and analyzed the results using a statistical design approach. They found that an intermediate recipe—moderate temperature, a few hours of treatment, and a medium glycine level—gave the best CO2 capture. Microscopy and gas‑adsorption measurements showed that the modified carbon kept almost the same surface area and pore size distribution as the original, indicating that glycine mostly decorated the inner walls instead of blocking passages. Infrared and X‑ray tests confirmed that new nitrogen‑ and oxygen‑bearing groups had appeared on the surface, while the underlying carbon framework remained largely unchanged.
How the Improved Material Behaves
By measuring how CO2 and nitrogen stick to the material at several temperatures, the team estimated the heat released when each gas is adsorbed. These values fell in the range typical for physical interactions, not permanent chemical reactions, but were noticeably stronger for the glycine‑treated carbon than for the raw sample. This means CO2 is held more tightly, which explains the higher capacity, yet it should still be possible to release the gas again by modest heating or pressure changes. The modified carbon also showed good thermal stability up to temperatures well above those normally found in post‑combustion capture systems, suggesting it could survive many capture–release cycles.
What This Could Mean for Power Plant Emissions
In everyday terms, the work shows that lightly decorating activated carbon with a simple amino acid can turn an off‑the‑shelf filter material into a more selective and stronger "magnet" for CO2. Glycine offers the best balance: its small size lets it line the pore walls with extra gripping points without clogging them, so more CO2 molecules can be caught and released repeatedly. While the material still works best at lower temperatures, smart heat‑exchange strategies in real plants could help cool exhaust gases enough to take advantage of this improved sponge. Together, these findings point toward inexpensive, tunable sorbents that could be retrofitted to existing facilities to help cut greenhouse emissions without overhauling the entire energy system.
Citation: Houshmand, D., Rashidi, F., Amjad-Iranagh, S. et al. Enhancement of CO2 capture in post combustion process using actived carbon modified by amino acids. Sci Rep 16, 10569 (2026). https://doi.org/10.1038/s41598-026-44400-z
Keywords: carbon capture, activated carbon, amino acids, post-combustion, CO2 adsorption