Clear Sky Science · en
Portable AI-powered scanning slit-light device for low-cost eye disease screening
A Pocket Tool to Protect Sight
Many of the world’s two billion people with poor vision lose sight because eye diseases are found too late. The front part of the eye—where the clear cornea, iris, and lens sit—holds early clues to problems such as cataract, glaucoma, and corneal warping, yet today’s best measuring machines are bulky and cost tens of thousands of dollars. This study introduces a battery‑powered, AI‑equipped handheld scanner that shines a thin beam of light into the eye and measures key structures with near‑clinic‑grade accuracy, at a parts cost under 500 dollars. If widely deployed, such a device could bring advanced eye screening from big hospitals to neighborhood clinics and community events.
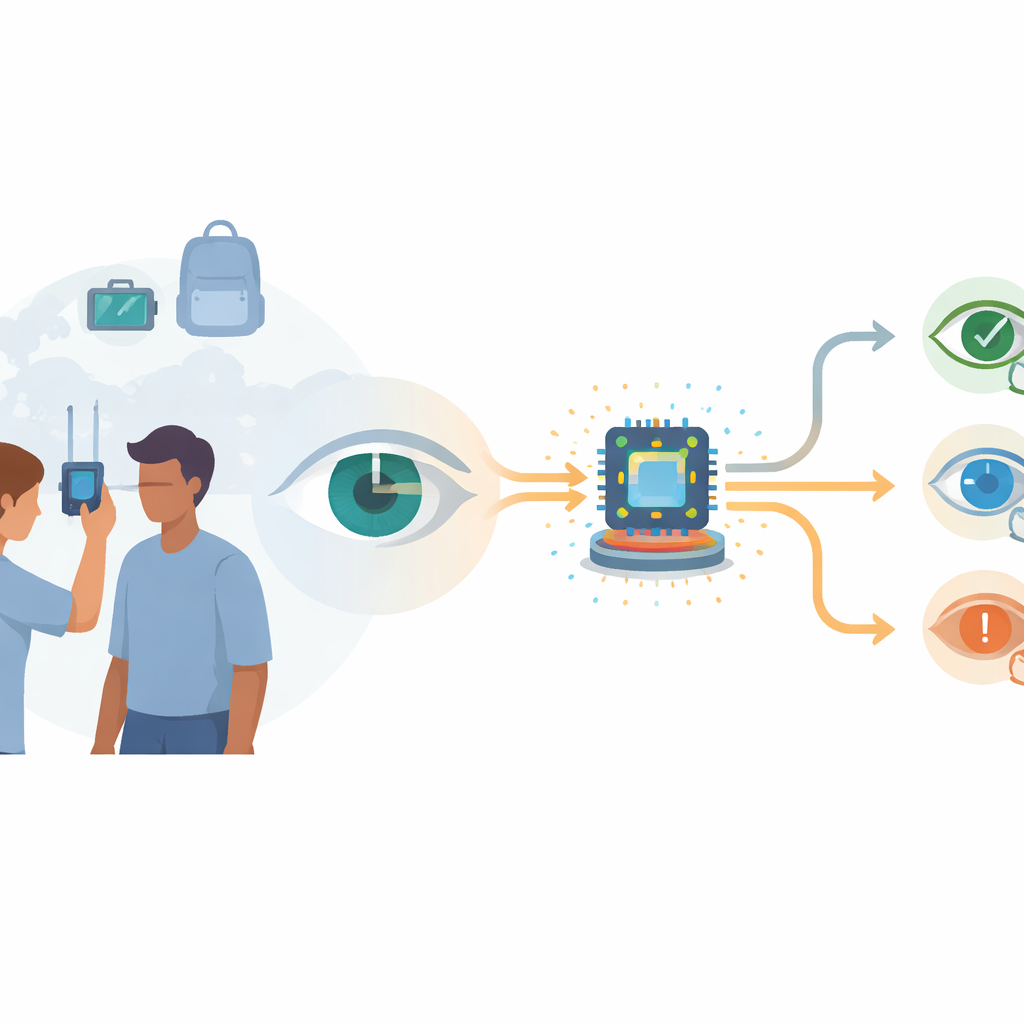
Why Eye Screening Is Hard to Reach
Vision loss is not spread evenly around the globe. People in low‑ and middle‑income countries, and even underserved groups in richer nations, often have limited access to eye doctors and advanced imaging tools. Standard slit‑lamp microscopes let specialists examine the eye’s front surface in detail, but they are heavy, expensive, and depend on the examiner’s skill and judgment. More advanced machines, such as anterior segment optical coherence tomography (AS‑OCT) scanners, can measure eye structures precisely, but their price tags above 50,000 dollars keep them locked in large clinics. As a result, basic measurements like how deep the front chamber of the eye is, or how thick the cornea is, are rarely collected during routine community screenings—even though these numbers are vital for spotting glaucoma risk and other conditions early.
A Handheld Scanner with Built‑In Intelligence
The research team built a compact scanning device that works a bit like a miniaturized, motorized slit‑lamp. A white LED shines through a narrow opening to create a thin “sheet” of light, which a tiny mirror sweeps across the eye. A small camera, fixed at an angle, records 50 to 70 images over about 15 seconds as the light moves. To keep patients comfortable, the system collects several dim images at each mirror position and averages them, boosting image quality without using harsh light. Inside the device, a lightweight deep‑learning model called LWBNA‑unet automatically traces key features in each frame: the bright reflections on the cornea and iris, the outline of the pupil, and the front and back surfaces of the cornea. Frames in which these landmarks are unclear—because of blinks or motion—are discarded automatically, serving as built‑in quality control.
Turning Pictures into Measurements
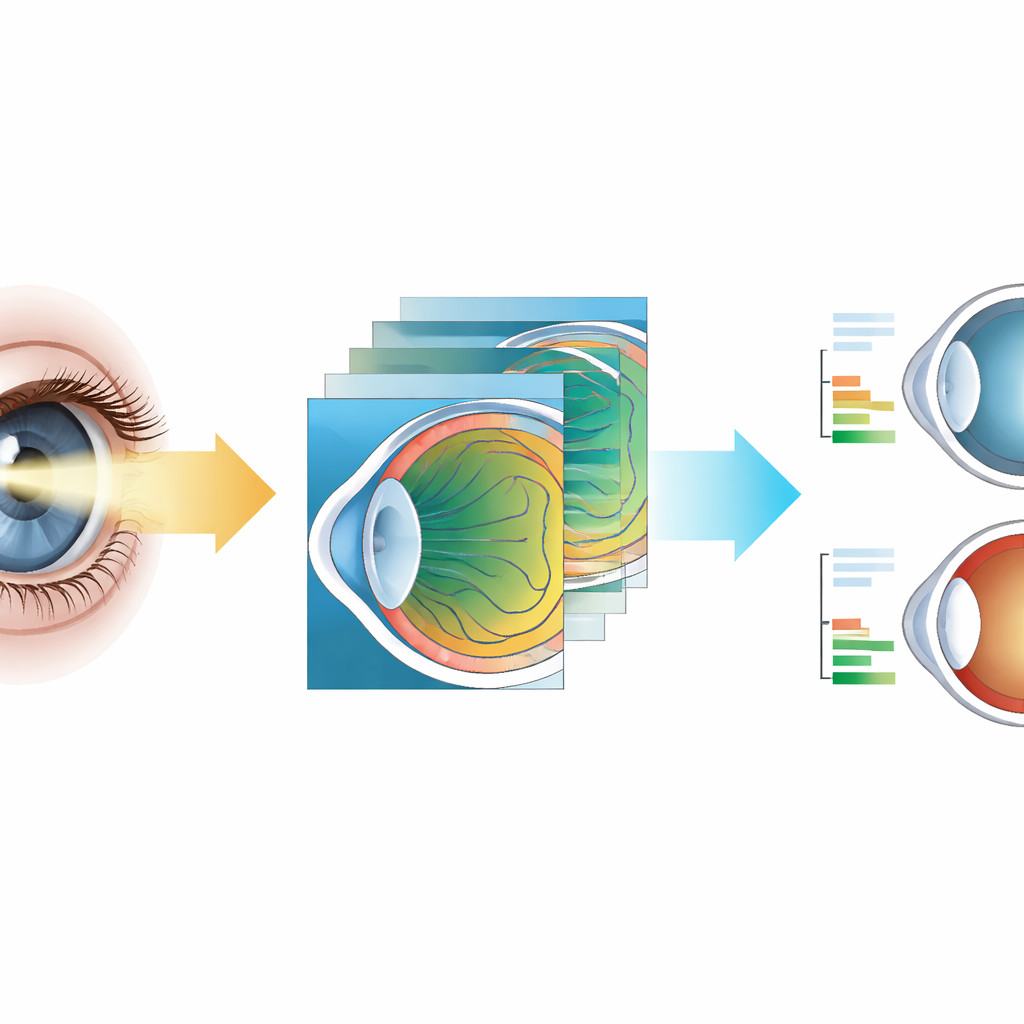
Once the eye structures are outlined, geometry‑aware software converts the raw images into real‑world distances. Because the light beam strikes the cornea at a slant, the reflected band appears artificially wide; the system corrects this using the known angles of illumination at each position. It then uses the visible width of the cornea in each frame as a personal ruler, assuming a typical real‑world diameter of 12 millimeters, to convert pixels into millimeters. With these steps, the device estimates the depth of the front chamber (the gap between the inner corneal surface and the iris) and the thickness of the central cornea, and also computes related measures such as the angle where the iris meets the cornea and the area of the chamber. Because the images are in natural color, the same scan can highlight cloudy lenses (cataract), hazy patches in the cornea, and abnormal steepening seen in keratoconus.
How Well It Matches Hospital Machines
To check accuracy, the researchers tested the prototype on about 170 adults and compared detailed measurements in 50 eyes with those from a commercial AS‑OCT system. For the depth of the front chamber, the handheld device tracked the reference machine very closely: the typical difference was near zero, with most eyes agreeing within about three‑tenths of a millimeter—small enough for screening and risk‑stratification. Statistical tests showed that, in practice, the two methods can be used almost interchangeably for this measurement. Corneal thickness estimates were less precise: on average, the portable unit read 20 to 30 micrometers thinner, and individual results could differ by roughly one to two camera pixels. The authors attribute this mainly to the current image resolution rather than the underlying principle, and they label these thickness readings as exploratory rather than clinic‑grade. Still, clinical examples demonstrated that the system clearly distinguishes deep, open angles from crowded, narrow ones and captures features of cataract, corneal opacity, and keratoconus that match standard slit‑lamp or OCT views.
What This Could Mean for Everyday Eye Care
By combining low‑cost optics, a motorized light beam, and on‑board AI, this handheld scanner delivers quantitative measurements that once required large, expensive machines, while also producing color images rich in disease clues. Its ability to run entirely on a small edge‑computing module, without an internet connection, makes it suitable for battery‑powered outreach in remote or crowded settings. The study shows that front‑chamber depth can be measured with near‑clinical reliability, positioning the device as a promising tool for early detection of angle‑closure glaucoma and related disorders. With further refinement and larger real‑world trials, the authors envision this platform evolving into a comprehensive, AI‑driven front‑of‑the‑eye screening system that could help prevent avoidable blindness on a global scale.
Citation: Kaushik, N., Sharma, P., Miya, T. et al. Portable AI-powered scanning slit-light device for low-cost eye disease screening. Sci Rep 16, 13862 (2026). https://doi.org/10.1038/s41598-026-44392-w
Keywords: eye screening, portable imaging, artificial intelligence, glaucoma risk, low-cost medical device