Clear Sky Science · en
Synthesis of rare earth doped MoS2 by the co-pyrolysis of molecular precursors
Why tiny magnetic layers matter
Imagine a material as thin as a few stacked sheets of paper, but made of atoms, that can both guide light and respond strongly to magnets even at extremely low temperatures. This study explores such a material: molybdenum disulfide, a well-known ultra-thin crystal, into which the researchers have carefully mixed small amounts of rare-earth elements. By developing a simple way to make these new powders, they open doors to cooler-running electronics, quantum technologies, and even ultra-low-temperature refrigeration techniques.
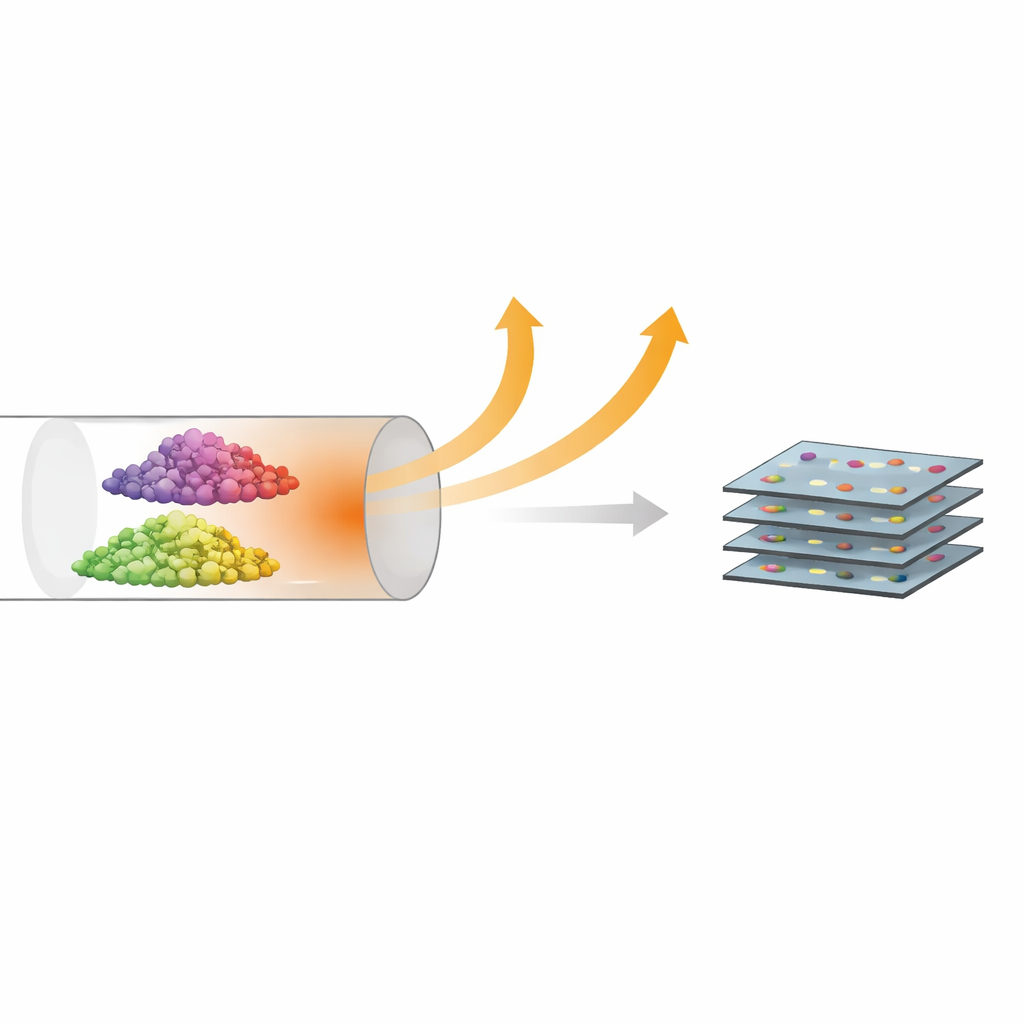
Building designer powders from simple ingredients
The team focused on MoS2, a layered compound sometimes called a cousin of graphene. To change its properties, they “doped” it with rare-earth atoms—erbium (Er) and neodymium (Nd)—which carry strong magnetic moments and can also influence light emission. Rather than using complex high-temperature, high-vacuum growth methods, they took a more practical route: mixing powdered metal–organic molecules that already contain molybdenum, sulfur, and the chosen rare-earth elements. When this mixture is heated to about 500 °C in an inert gas, the molecules break apart and reorganize into tiny MoS2 crystals with the rare-earth atoms built in from the start.
Checking that the recipe really works
To see what they had made, the researchers used a suite of microscopes and scattering techniques. X-ray diffraction and Raman measurements showed that the powders consist of very small crystallites—only a few nanometers across—of the usual hexagonal form of MoS2, and typically only two to three atomic layers thick. Electron microscopy combined with elemental mapping revealed that the rare-earth atoms are spread evenly throughout the samples rather than clumping into separate particles. Importantly, the relative amounts of molybdenum and dopant measured in the final powders closely matched the ratios in the starting mixture, meaning the method offers straightforward control over how much rare-earth is added.
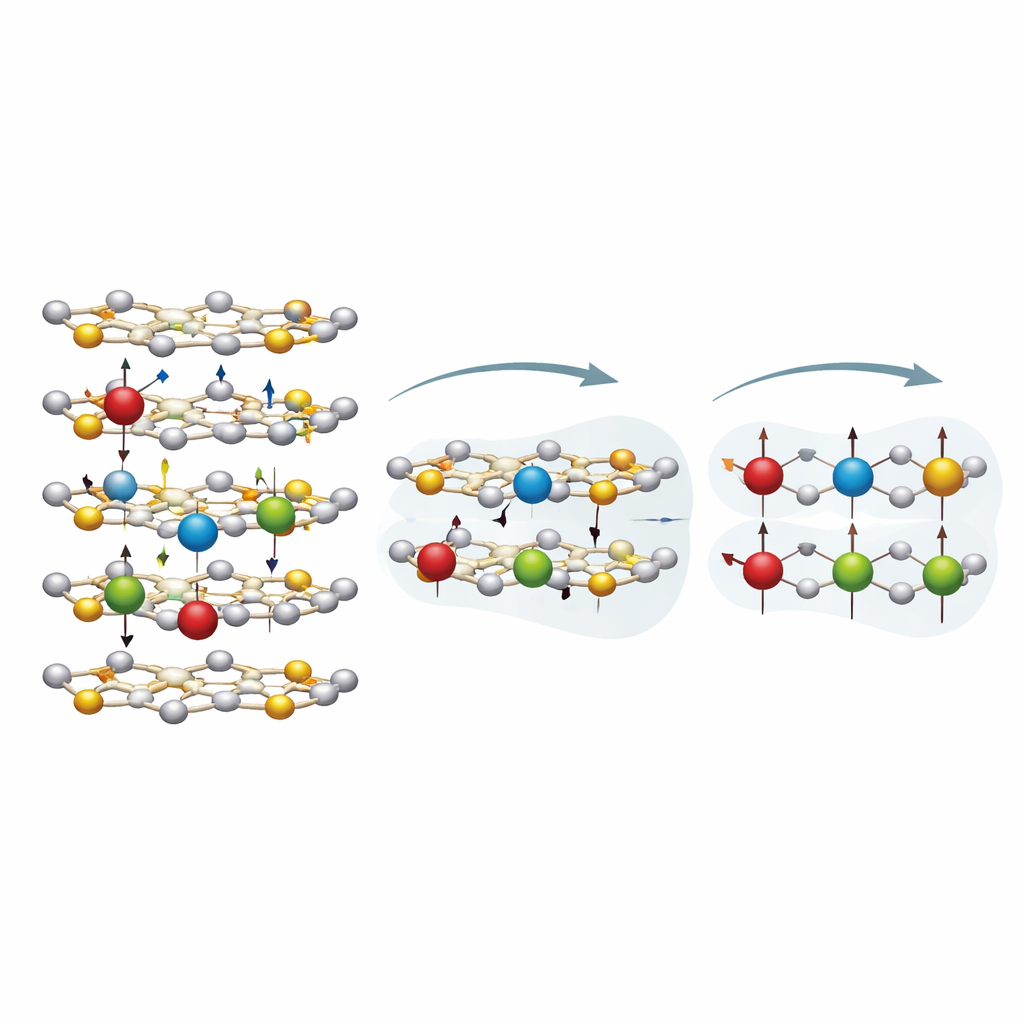
Where the rare-earth atoms sit
A key question is whether the Er and Nd atoms squeeze between the MoS2 layers or actually replace some of the molybdenum atoms inside those layers. To answer this, the team turned to synchrotron-based x-ray absorption measurements, which sense how an atom is surrounded by its neighbors. By comparing the experimental data with computer-built structural models, they found that the best match occurs when the rare-earth atoms occupy molybdenum sites within the sulfur–molybdenum–sulfur sandwiches, rather than being trapped in the gaps between layers. This picture agrees with earlier theoretical work suggesting that such “intralayer” substitution is energetically more favorable and mirrors what has been seen for other metals doped into MoS2.
From quiet layers to strong magnetic responders
While light emission from the doped powders is weak—consistent with the fact that few-layer MoS2 tends to emit poorly—their magnetic behavior changes dramatically. Measurements with a sensitive magnetometer show that Er- and Nd-doped samples respond strongly to an applied magnetic field, far more than undoped MoS2. At very low temperatures, the spins associated with the rare-earth atoms nearly line up completely in strong fields, a hallmark of a robust paramagnetic response. Crucially, there is no sign of the collective, permanent magnetization (ferromagnetism) that other groups have seen in more defect-rich samples, even down to 2 kelvin. Analysis using the Curie–Weiss law confirms that the magnetic moments per rare-earth ion closely match their expected free-ion values.
Silencing defects to keep spins independent
Electron paramagnetic resonance measurements add another piece to the puzzle. Undoped MoS2 shows a sharp signal associated with magnetic defects, likely linked to missing atoms or unwanted adsorbed species. In the doped samples, this defect signal is strongly reduced, while new broad features appear that can be traced to the Er and Nd ions themselves. This suggests that the rare-earth atoms not only introduce their own magnetic moments, but also “quiet” many of the pre-existing magnetic defects that might otherwise cause the spins to couple and freeze into an ordered state. By keeping the spins relatively isolated, the material maintains paramagnetism instead of becoming ferromagnetic.
What this means for future technologies
In simple terms, the authors have shown a scalable, low-temperature route to make ultra-thin MoS2 powders whose magnetic strength can be dialed in with rare-earth atoms, while keeping those atoms neatly embedded within the crystal layers and largely free from disruptive defects. Because the material stays paramagnetic down to at least 2 kelvin, it could eventually serve as a platform for techniques that cool devices by magnetizing and demagnetizing a solid, or as a host for rare-earth spins in quantum applications. The work also hints that the same chemical strategy could be extended to many other rare-earth elements, offering a toolbox for designing custom magnetic and optical properties in two-dimensional materials.
Citation: Cao, Y., Alfredsson, M., Chadwick, A.V. et al. Synthesis of rare earth doped MoS2 by the co-pyrolysis of molecular precursors. Sci Rep 16, 14252 (2026). https://doi.org/10.1038/s41598-026-44301-1
Keywords: rare earth doped MoS2, two-dimensional materials, paramagnetism, molecular precursors, magnetic nanomaterials