Clear Sky Science · en
Preparation of new modified CuFe₂O₄ nanoparticles by benzalkonium chloride as enhancer of natural gas hydrate formation
Turning Gas into Ice-Like Fuel Bricks
Suppose you could store natural gas not in heavy steel cylinders or long pipelines, but as compact, ice-like blocks that are stable and safe to handle. This study explores exactly that idea. The researchers show how specially engineered nanoparticles can help natural gas quickly freeze into “gas hydrates” – crystalline solids where gas molecules are trapped in cages of water – making it easier to store and transport cleaner-burning fuel around the world.
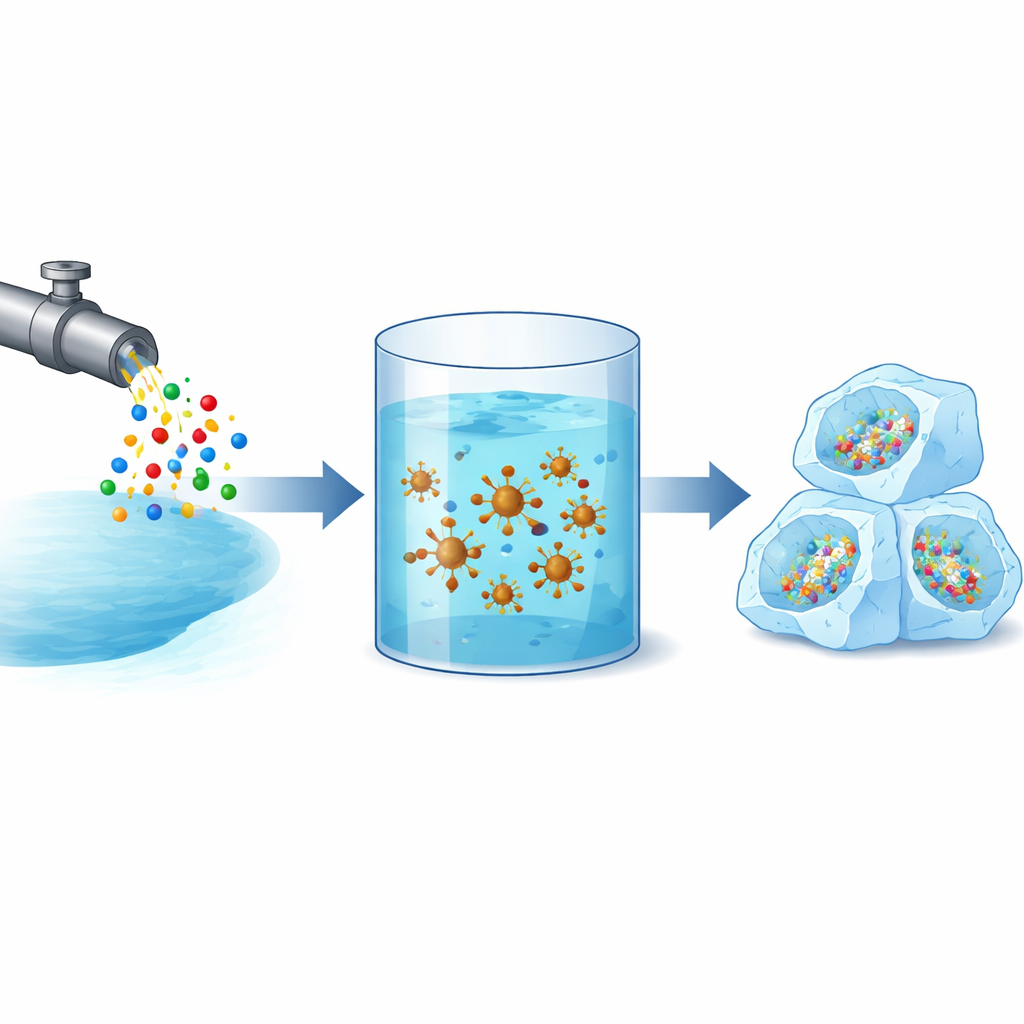
Why Storing Gas as Ice Matters
Natural gas is a key part of today’s energy mix because it burns more cleanly than coal or oil. But getting it from remote fields to cities usually requires either long pipelines or energy-hungry liquefaction plants. Gas hydrates offer an appealing alternative: under the right pressure and temperature, water forms solid cages that trap natural gas, creating dense, solid “fuel bricks.” The catch is that these hydrates often form slowly and do not always pack in as much gas as engineers would like. Finding ways to speed up hydrate formation and increase how much gas they store could make this technology far more practical for large-scale energy use.
Building Smarter Nanoparticles
The team focused on tiny particles of copper ferrite (CuFe₂O₄), a magnetic material that can be suspended in water. On their own, these nanoparticles already provide extra surfaces where hydrate crystals can begin to grow. The researchers went further by modifying the particles with benzalkonium chloride, a common disinfectant that also acts like a soap, helping particles disperse in water and interact with gas and water molecules. They prepared three systems: plain copper ferrite in water, copper ferrite physically mixed with the surfactant, and copper ferrite chemically bonded to the surfactant. Advanced tools, including infrared spectroscopy, X‑ray diffraction, electron microscopy, and surface-area measurements, confirmed how the surfactant coats and restructures the particles, creating more pores, larger surface area, and a distinctive flower-like texture ideal for holding gas and water together.
Speeding Up Formation and Packing in More Gas
To test performance, the researchers formed natural gas hydrates in a high‑pressure cell at conditions similar to cold deep‑sea environments. They measured how long it took before hydrates started forming (the induction time), how fast gas was consumed, and how much gas ended up stored in the solid. Plain copper ferrite at its best concentration needed about 12 minutes before hydrates appeared and stored only about 0.12 moles of gas per mole of water. Adding benzalkonium chloride as a simple mixture already cut the waiting time in half and more than doubled gas uptake. The chemically bonded version performed best: at an ultra‑low dose of 0.005 weight percent, induction time fell to about 5 minutes and gas storage climbed to roughly 0.35 moles per mole of water, almost a three‑fold improvement over unmodified particles. Gas could also be recovered more completely during warm‑up, with recovery rising from about 82 percent for plain particles to about 95 percent for the chemically modified ones.
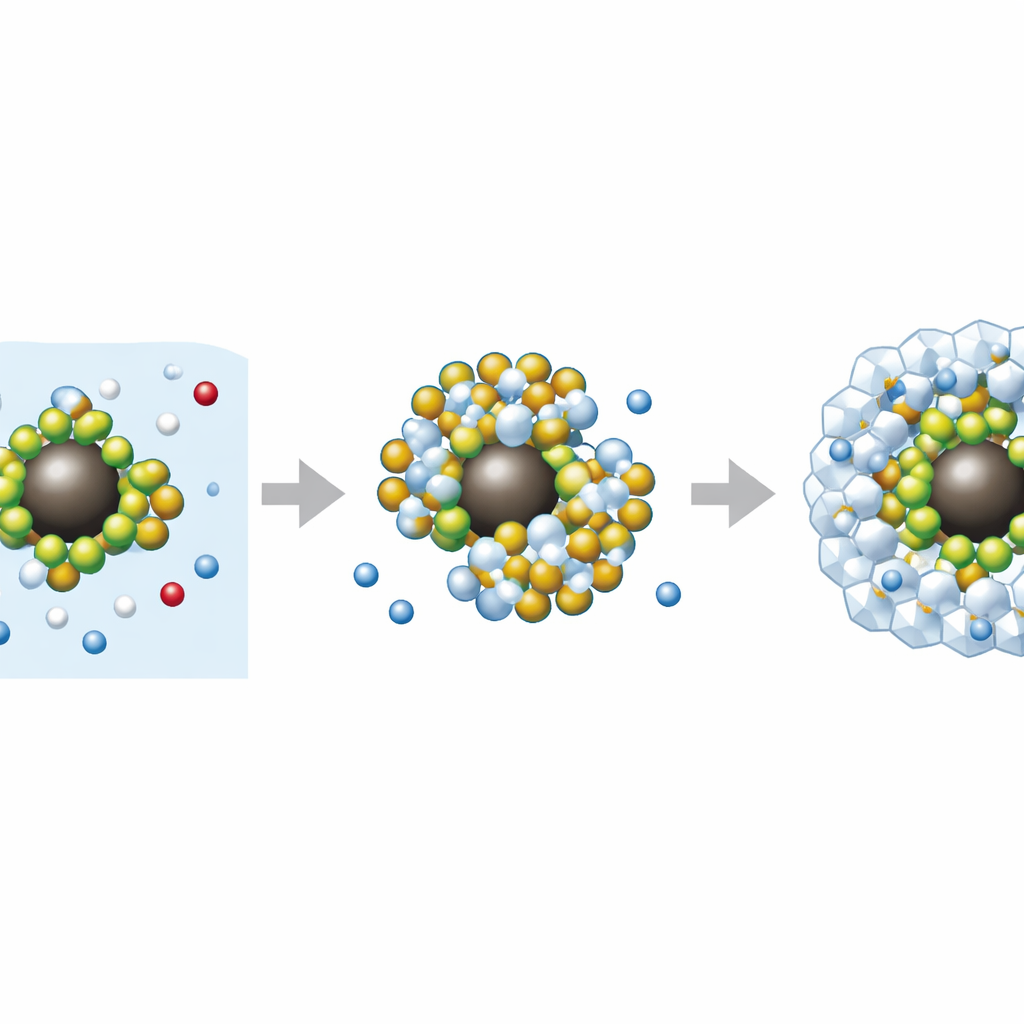
How the Tiny Helpers Do Their Job
The enhanced behavior traces back to how the modified nanoparticles reshape the microscopic environment where hydrates form. Chemically attaching benzalkonium chloride spreads the particles evenly through the water and prevents them from clumping, so many more active sites are available. The surfactant tail groups and the enlarged, mesoporous surface help gather gas molecules near the particle surface while simultaneously organizing surrounding water. Electronic calculations show that the chemical coating changes how charge is distributed on the particle, strengthening interactions with both gas and water. Together, these effects lower the energy barrier for the first hydrate cages to appear, then support orderly, rapid crystal growth and more efficient gas packing. During dissociation, the same improved structure allows the trapped gas to be released more completely when the solid is warmed.
From Lab Concept to Future Fuel Blocks
In everyday terms, the study shows how giving nanoparticles a soap-like coat can turn them into powerful “seeds” that help natural gas freeze quickly and densely into solid, ice‑like blocks. By sharply reducing the time needed to form hydrates and nearly tripling the storage capacity, the benzalkonium‑modified copper ferrite system points toward more compact, energy‑efficient ways to store and move natural gas. While further engineering is needed before such materials are used in real tanks or ships, the work outlines a promising route to safer, cleaner, and more flexible fuel logistics.
Citation: Alsabagh, A.M., Shoaib, A.M., Awad, M. et al. Preparation of new modified CuFe₂O₄ nanoparticles by benzalkonium chloride as enhancer of natural gas hydrate formation. Sci Rep 16, 14634 (2026). https://doi.org/10.1038/s41598-026-44211-2
Keywords: natural gas hydrate storage, nanoparticle promoters, benzalkonium chloride, copper ferrite nanofluids, gas transport technology