Clear Sky Science · en
Novel multi-metal ion-exchanged zeolite for oxidative-adsorptive desulfurization of model and real diesel fuel: a response surface methodology (RSM) study
Why Cleaner Diesel Matters
Burning diesel fuel may power trucks, ships, and generators, but it also releases sulfur compounds that form acid rain and fine particles harmful to human lungs. Governments now demand ultra‑low‑sulfur fuels, yet the most stubborn sulfur molecules in diesel resist conventional cleanup methods. This study explores a new type of porous material that can grab and hold on to these hard‑to‑remove sulfur compounds more efficiently, offering a potential route to cleaner air without massively increasing energy use or cost.
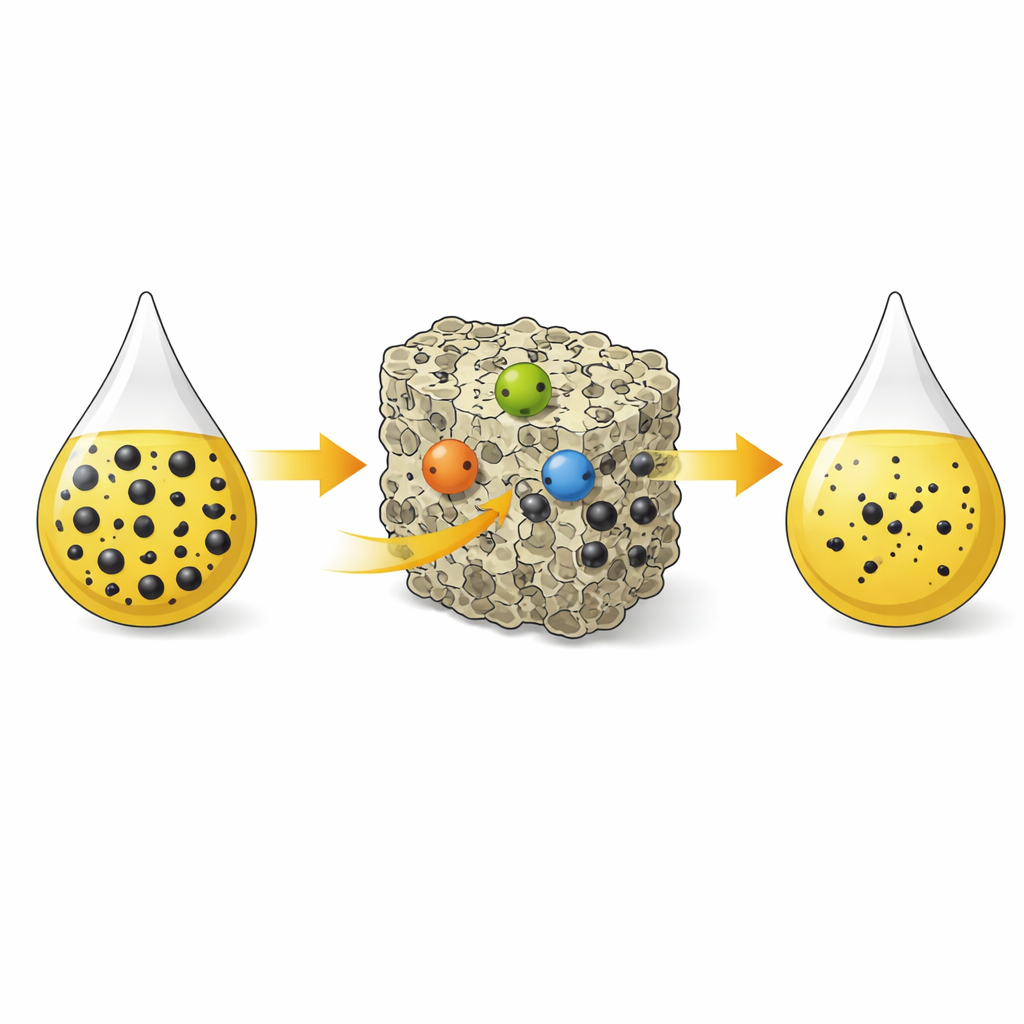
A New Sponge for Stubborn Sulfur
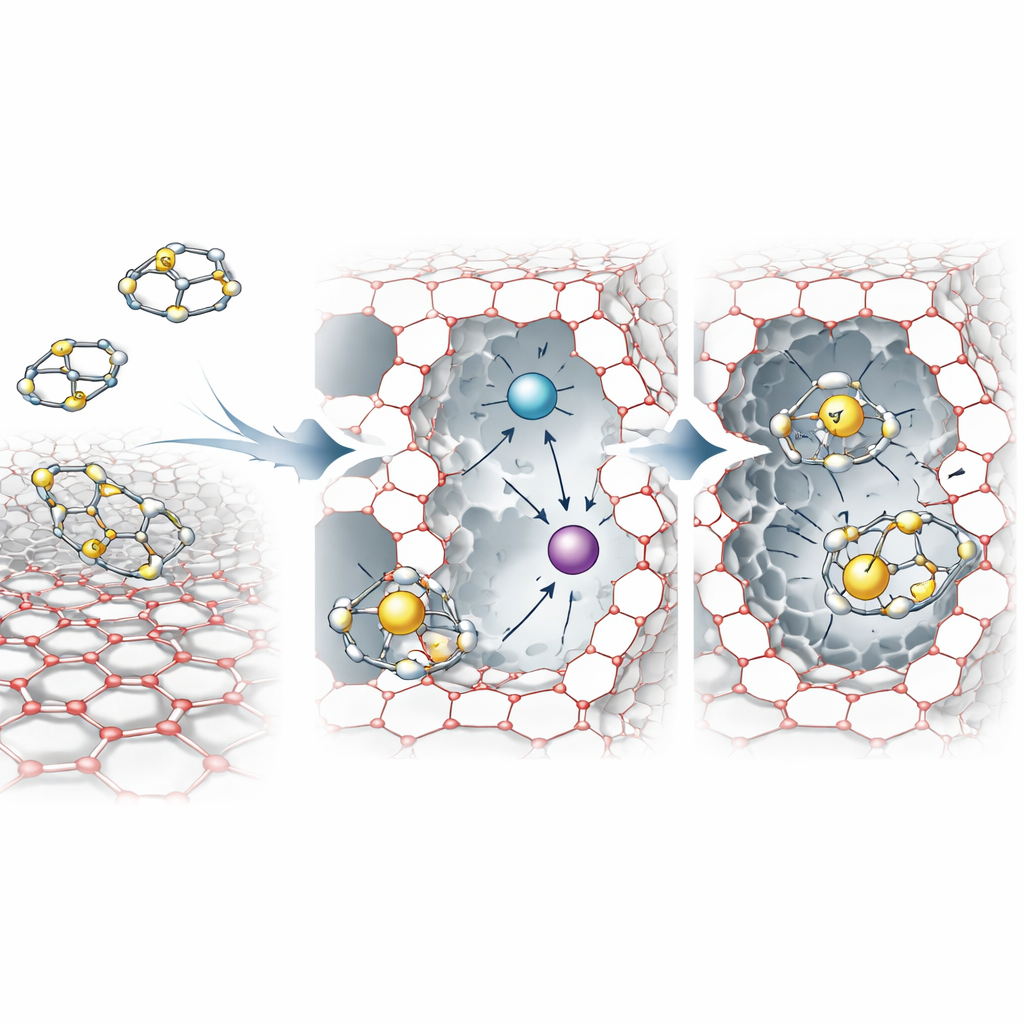
At the heart of the work is a material called a zeolite, a crystalline mineral full of tiny, uniform channels. The authors start with a common zeolite known as NaY and transform it into a more powerful “sponge” for sulfur by replacing some of its original sodium ions with three different metals: silver, nickel, and cerium. This carefully controlled sequence of ion exchanges produces a multi‑metal version, dubbed AgNiCeY. Each of the three metals offers a slightly different way to attract sulfur, so that together they create a richer landscape of active sites inside the pores than a single‑metal zeolite can provide.
Designing the Metal Mix for Maximum Effect
The team shows that the order in which metals are introduced is crucial. Silver goes in first and occupies the most accessible sites in the large pores, where it can form strong interactions with sulfur-containing rings. Nickel, with smaller ions, can then reach more tucked‑away positions. Cerium, which carries a higher positive charge, is added last so it does not crowd out the other metals too early. Structural tests confirm that, even after these exchanges and a high‑temperature treatment, the underlying zeolite framework remains intact, while the pore structure subtly shifts: very small pores shrink somewhat, but slightly larger ones become more prominent. This trade‑off actually favors the capture of bulky sulfur molecules like dibenzothiophene, which are among the hardest to remove from fuel.
How the Material Grabs Sulfur
To see how sulfur sticks to the new material, the researchers combine spectroscopy, electron microscopy, and electronic structure arguments. They find that the incorporated metal ions act as so‑called Lewis acids—positively charged centers that can accept pairs of electrons from sulfur atoms. When a sulfur‑bearing molecule approaches a metal site inside the zeolite, it can form a strong bond resembling a covalent connection between sulfur and the metal. Different metals favor slightly different bonding styles and strengths, leading to multiple distinct interaction patterns. Microscopy images before and after exposure to sulfur‑containing fuel show the zeolite crystals becoming visibly coated, consistent with a monolayer of adsorbed molecules lining the pore surfaces.
Finding the Sweet Spot in Realistic Conditions
The authors do not rely on trial‑and‑error alone. They use a statistical approach called response surface methodology to map how key operating variables—contact time, initial sulfur concentration, and the ratio of oil to adsorbent—affect performance. Working first with a simplified “model fuel” (a single sulfur compound dissolved in a clean solvent), they determine that the most important factor is how much fuel is treated per gram of zeolite, followed by sulfur concentration, while contact time beyond a moderate value adds little benefit. Under optimized conditions, the multi‑metal zeolite reaches an equilibrium capacity of about 33 milligrams of sulfur per gram of adsorbent, compared with about 13 milligrams for the original NaY, a 164% improvement. The adsorption behavior fits a classic monolayer model, reinforcing the picture of well‑defined, energetically similar binding sites.
From Model Fuel to Real Diesel
Crucially, the researchers then apply the conditions optimized on the model fuel to real diesel containing over 2500 parts per million of sulfur. In this much more complex mixture, the AgNiCeY zeolite still outperforms the parent NaY, removing about 61% of the sulfur compared with 58% for NaY. While the percentage gain looks modest, it occurs under demanding, realistic conditions where many other fuel components compete for space in the pores. The multi‑metal zeolite also shows better resilience when regenerated repeatedly with ethanol: even after five use‑and‑clean cycles, it retains close to 90% of its capacity, versus about 63% for the unmodified material.
What This Means for Cleaner Fuels
To a non‑specialist, the message is that by carefully decorating a porous mineral with a tailored mix of metals, it is possible to create a highly selective filter for the most troublesome sulfur molecules in diesel. Rather than relying solely on energy‑hungry, high‑pressure hydrogen treatment, refineries could, in principle, add an oxidative–adsorptive polishing step using such multi‑metal zeolites to push sulfur down to ultra‑low levels. This study shows both the design logic—tuning metal types, their locations, and operating conditions—and the practical promise, with strong performance on real diesel and good recyclability. It points toward more efficient, flexible approaches to cleaning up transportation fuels and reducing the environmental footprint of combustion engines while they remain in use.
Citation: Shafaghat, J., Movahedirad, S. & Sobati, M. Novel multi-metal ion-exchanged zeolite for oxidative-adsorptive desulfurization of model and real diesel fuel: a response surface methodology (RSM) study. Sci Rep 16, 11734 (2026). https://doi.org/10.1038/s41598-026-44172-6
Keywords: diesel desulfurization, zeolite adsorbent, multi-metal catalyst, sulfur pollution, clean fuels