Clear Sky Science · en
Rapid neurological recovery in Guillain-Barré syndrome treated with efgartigimod
Why this nerve illness and new treatment matter
Guillain–Barré syndrome is a sudden nerve disorder that can turn a healthy person into someone who can barely move or even breathe on their own within days. Current treatments help many patients, but recovery is often slow and some people are left with lasting weakness. This study looks at a newer medicine, efgartigimod, that works by quickly clearing harmful antibodies from the blood, and asks a simple but crucial question: can it help people with Guillain–Barré syndrome get better faster and more safely than standard therapies?
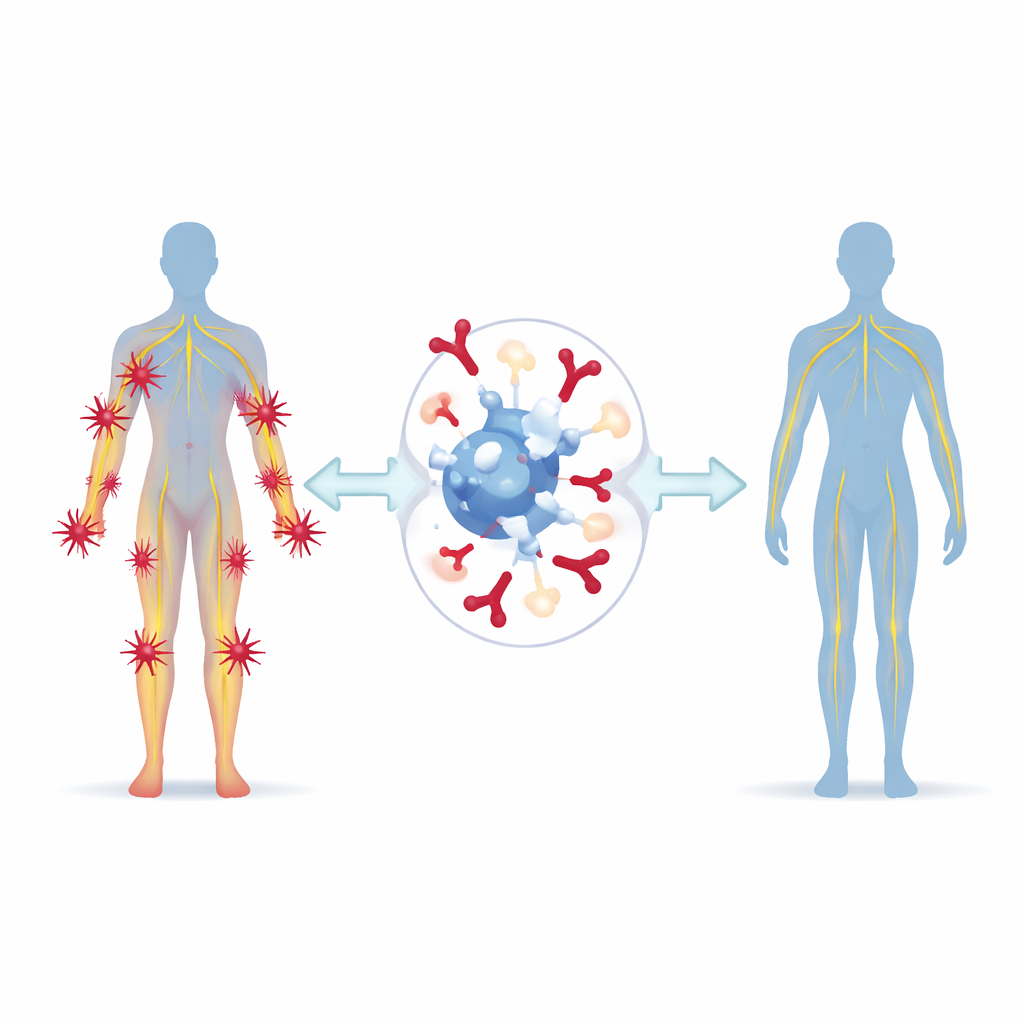
The illness that turns the body against itself
Guillain–Barré syndrome is an autoimmune attack on the nerves that run outside the brain and spinal cord. Often triggered by an infection or, less commonly, a vaccination, it causes the immune system to mistake nerve coverings for invaders. People can develop rapidly rising weakness in all four limbs, lose the ability to walk, swallow, or speak clearly, and in severe cases need a ventilator to breathe. Around the world, about 100,000 new cases occur each year. For decades, doctors have relied on two main treatments: intravenous immunoglobulin (IVIg), a pooled antibody preparation from donors, and plasma exchange, a procedure that filters harmful substances out of the blood. Both can help, but they are slow to act in some patients, hard to access in many hospitals, and can cause serious side effects such as blood clots, heart rhythm problems, or kidney injury.
A drug designed to sweep out harmful antibodies
Efgartigimod was created to tackle autoimmune diseases by speeding up the removal of IgG antibodies from the body. It binds tightly to the neonatal Fc receptor, a protein that normally rescues antibodies from being broken down and returns them to the circulation. By blocking this recycling system, efgartigimod causes antibody levels—including the misdirected ones that attack nerves—to fall more quickly. The drug is already approved or under study for other autoimmune conditions, such as myasthenia gravis and chronic inflammatory nerve disorders, and early case reports hinted that it might rapidly ease symptoms in Guillain–Barré patients who did not respond well to standard care. 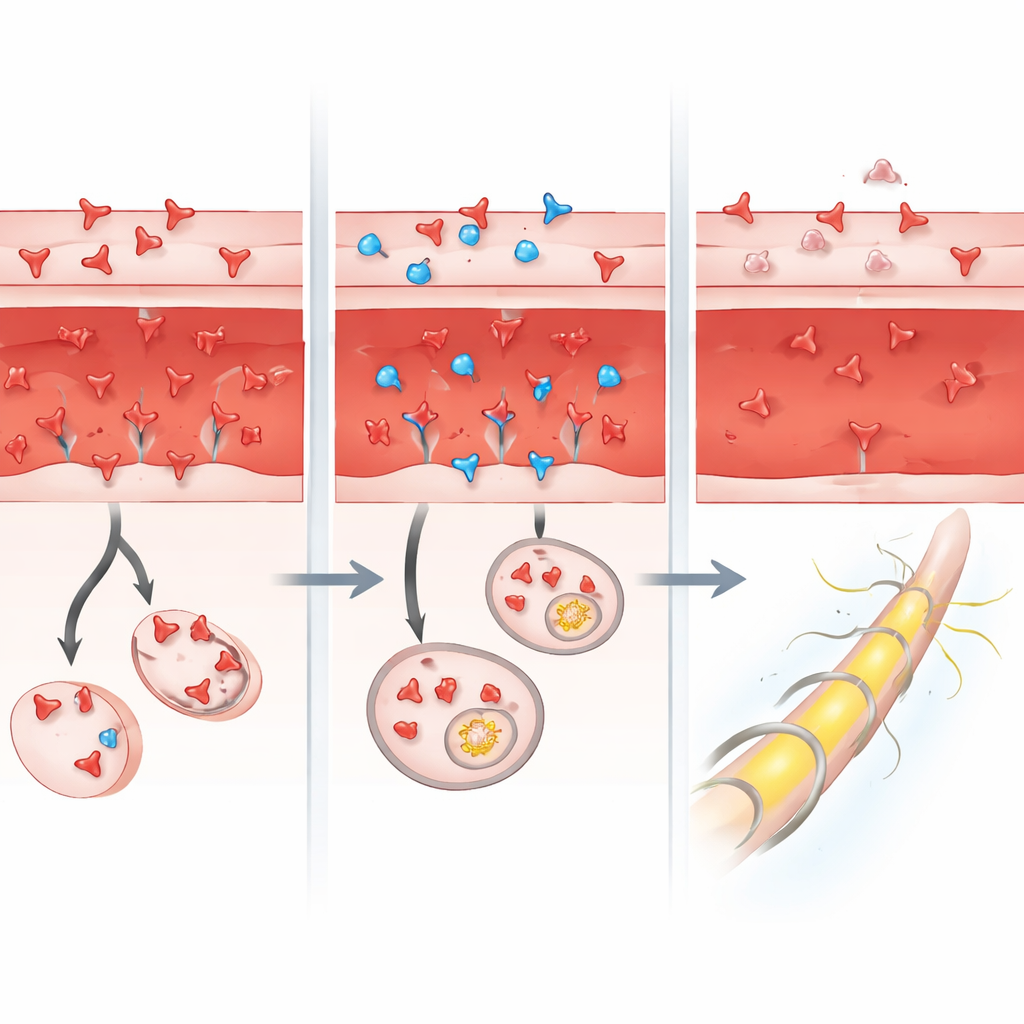
How the study was done and who was treated
The researchers reviewed the medical records of adults treated for Guillain–Barré syndrome at two hospitals in China between late 2022 and mid‑2024. All had significant disability at the time treatment began; they could not walk independently and some needed breathing support. Seventeen patients fit the criteria and were divided into three groups based on the therapy they actually received: eight got IVIg, four had plasma exchange, and five were treated with efgartigimod, sometimes after an initial plasma exchange. Everyone started immunotherapy within three days of diagnosis, and the teams tracked how quickly their nerve function improved using widely used disability and muscle strength scores, as well as the time it took to walk again without help.
Faster early gains with the new approach
Patients who received efgartigimod improved noticeably sooner than those given IVIg or plasma exchange alone. On average, they gained one step on the standard disability scale in about four days, compared with seven days for the IVIg group and more than eleven days for the plasma‑exchange group. Within the first week, four out of five people in the efgartigimod group had mild or no disability by the INCAT scale, and four out of five had normal total muscle strength scores. In contrast, only about one in eight IVIg‑treated patients and one in four plasma‑exchange patients reached that same level of early recovery. Many efgartigimod patients were walking without assistance within a week, and troublesome symptoms such as breathing difficulty, facial weakness, double vision, and swallowing problems often cleared earlier than in those on standard therapies.
Safety and what this means for future care
The new treatment did not appear to add safety concerns in this small series. One efgartigimod‑treated patient developed a mild rash, while half of those who underwent plasma exchange had low clotting protein levels, a known risk of that procedure. No serious complications linked to efgartigimod were reported. By one and three months after treatment, however, overall outcomes looked similar across all three groups, suggesting that the main advantage of efgartigimod lies in how quickly it acts rather than in a dramatically better long‑term result.
What this could mean for patients and families
For a condition that can steal movement and independence in a matter of days, even a few days’ faster turnaround can mean less time in intensive care, fewer complications, and a quicker return to everyday life. This study offers early but encouraging evidence that efgartigimod can bring on that early improvement more rapidly than long‑standing treatments, while remaining relatively safe. Because only 17 patients were included and treatment choice was not randomized, the findings are not final proof. Larger, carefully controlled trials will be needed to confirm whether this antibody‑clearing strategy should become a routine option. Still, the results point to a future in which people with Guillain–Barré syndrome might have more targeted, faster‑acting therapies that shorten the most frightening phase of the illness.
Citation: Cheng, Y., Li, W., Xie, S. et al. Rapid neurological recovery in Guillain-Barré syndrome treated with efgartigimod. Sci Rep 16, 14128 (2026). https://doi.org/10.1038/s41598-026-44163-7
Keywords: Guillain-Barré syndrome, autoimmune neuropathy, efgartigimod, antibody clearance, neurological recovery