Clear Sky Science · en
Mechanistic role of gut microbiota metabolites in hypertension-insomnia comorbidity via integrated network pharmacology and molecular dynamics
Why the Gut Matters for Blood Pressure and Sleep
High blood pressure and poor sleep are two problems that often strike together, raising the risk of heart attacks, strokes and reduced quality of life. Yet doctors still struggle to explain why these conditions are so closely linked. This study explores an unexpected player in this story: the trillions of microbes living in our intestines and the tiny chemicals they produce. By combining large-scale data analysis with computer simulations of molecules, the researchers map out how gut-derived substances may help drive – or ease – both hypertension and insomnia.
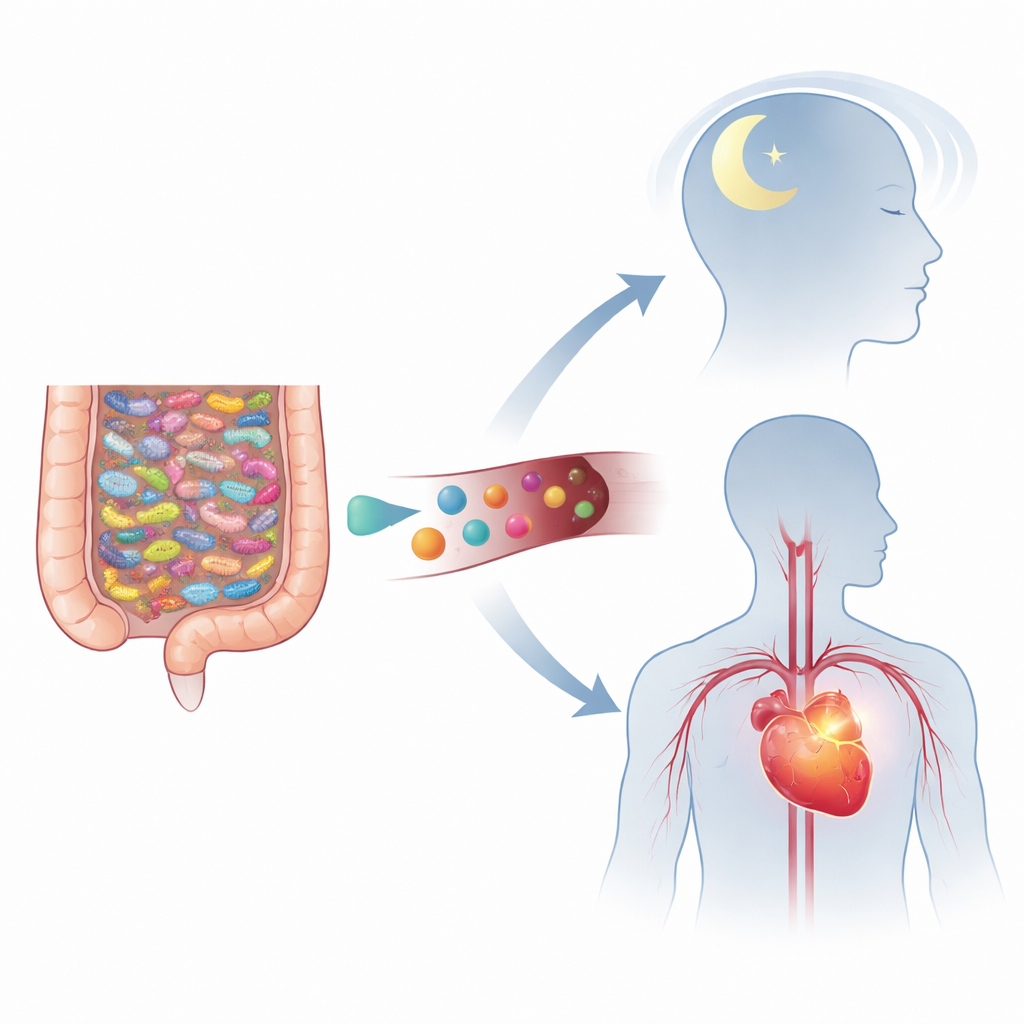
Two Common Problems with a Hidden Connection
Hypertension affects more than a billion people worldwide, and chronic insomnia is also widespread. People with high blood pressure are far more likely to suffer from trouble falling asleep, frequent awakenings and restless nights than those with normal pressure. Long-term studies show that insomnia raises the risk of later developing hypertension, and having both together greatly worsens cardiovascular danger. Blood tests in such patients often reveal higher levels of inflammatory molecules, suggesting that chronic, low-grade inflammation might be a shared thread between the two conditions.
Tiny Gut Chemicals with Big Effects
The human gut is home to countless microbes that break down food into short-chain fatty acids, tryptophan by-products and many other small molecules. These substances do more than aid digestion: they can influence blood vessel tone, brain signaling and immune responses. For example, butyrate and acetate can lower blood pressure and calm overactive immune cells, while tryptophan derivatives help shape brain chemistry linked to mood and sleep. The authors propose that changes in this chemical output – due to diet, stress or altered gut communities – might simultaneously disturb blood pressure control and sleep regulation.
Building a Map of Shared Molecular Targets
To test this idea, the team first pulled together 278 known gut microbial metabolites and their human protein targets from specialized databases. They also assembled more than 14,000 genes and proteins associated with hypertension or insomnia from major biomedical resources, then looked for overlap. This filtering process yielded 18 core molecular targets that sit at the crossroads of gut chemistry, blood pressure and sleep. Network analysis showed that two proteins, the inflammatory messenger IL-6 and the metabolism regulator PPARG, were especially central, connecting to many other components. Pathway analysis highlighted the IL-17 signaling route – a key driver of immune activation – as a major hub that bridges inflammation, fat handling and brain-related processes.
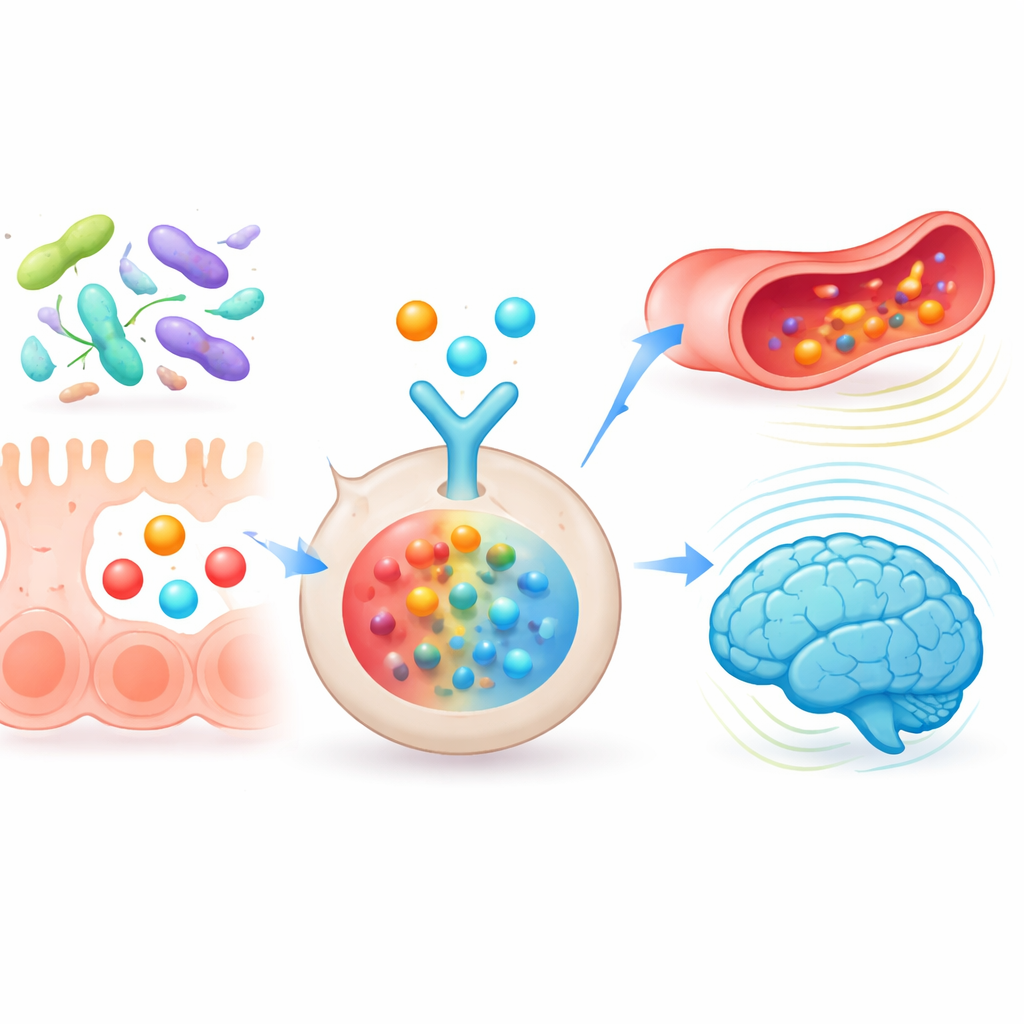
Zooming In on Key Metabolites and Pathways
The researchers then focused on a subset of gut-derived molecules, including short-chain fatty acids like butyrate and propionate and the tryptophan product 3-indolepropionic acid. Using computer docking and molecular dynamics, they simulated how strongly these metabolites could bind to IL-6 and how stable those interactions were over time. Several metabolites showed robust, energetically favorable binding, especially 3-indolepropionic acid and butyrate, supporting the idea that they could dampen IL-6-driven inflammation. Further analysis of metabolic pathways pointed to butyrate metabolism and tryptophan breakdown as central routes tying together energy use, vascular health and brain function. A broader network that linked microbes, metabolites, targets and pathways singled out the probiotic species Lacticaseibacillus paracasei as a particularly influential microbe, capable of boosting beneficial metabolites and toning down IL-17–related inflammatory signals.
From Computational Clues to Future Therapies
The study concludes that a "microbiota–metabolite–inflammation" axis likely underlies much of the overlap between hypertension and insomnia. In simple terms, when gut microbes and their products are in balance, they help keep immune responses, blood vessels and brain activity in a healthy range. When this balance is disturbed, inflammatory pathways such as IL-17 and IL-6 become overactive, contributing both to raised blood pressure and disrupted sleep. While the findings come from computational models rather than clinical trials, they point toward promising strategies: targeting IL-17 signaling, supplementing specific gut metabolites like butyrate or 3-indolepropionic acid, and using carefully selected probiotics such as L. paracasei to restore a healthier gut ecosystem and, in turn, improve both heart health and sleep.
Citation: Ma, RL., Kou, YS., Wang, YY. et al. Mechanistic role of gut microbiota metabolites in hypertension-insomnia comorbidity via integrated network pharmacology and molecular dynamics. Sci Rep 16, 14486 (2026). https://doi.org/10.1038/s41598-026-44114-2
Keywords: gut microbiome, hypertension, insomnia, inflammation, short-chain fatty acids