Clear Sky Science · en
Evodiamine alleviates MPTP-induced Parkinson’s disease in mice by regulating gut microbiota and suppressing TLR4/MyD88/NF-kB pathway
A New Clue in the Parkinson’s Puzzle
Parkinson’s disease is best known for its tremors and stiffness, but scientists are increasingly looking to a surprising place for answers: the gut. This study explores whether a natural compound called evodiamine, extracted from a traditional medicinal plant, can ease Parkinson-like symptoms in mice by calming inflammation in the intestines and the brain. For readers, it offers a glimpse of how treating the gut—rather than targeting the brain alone—might someday help slow a devastating brain disorder.
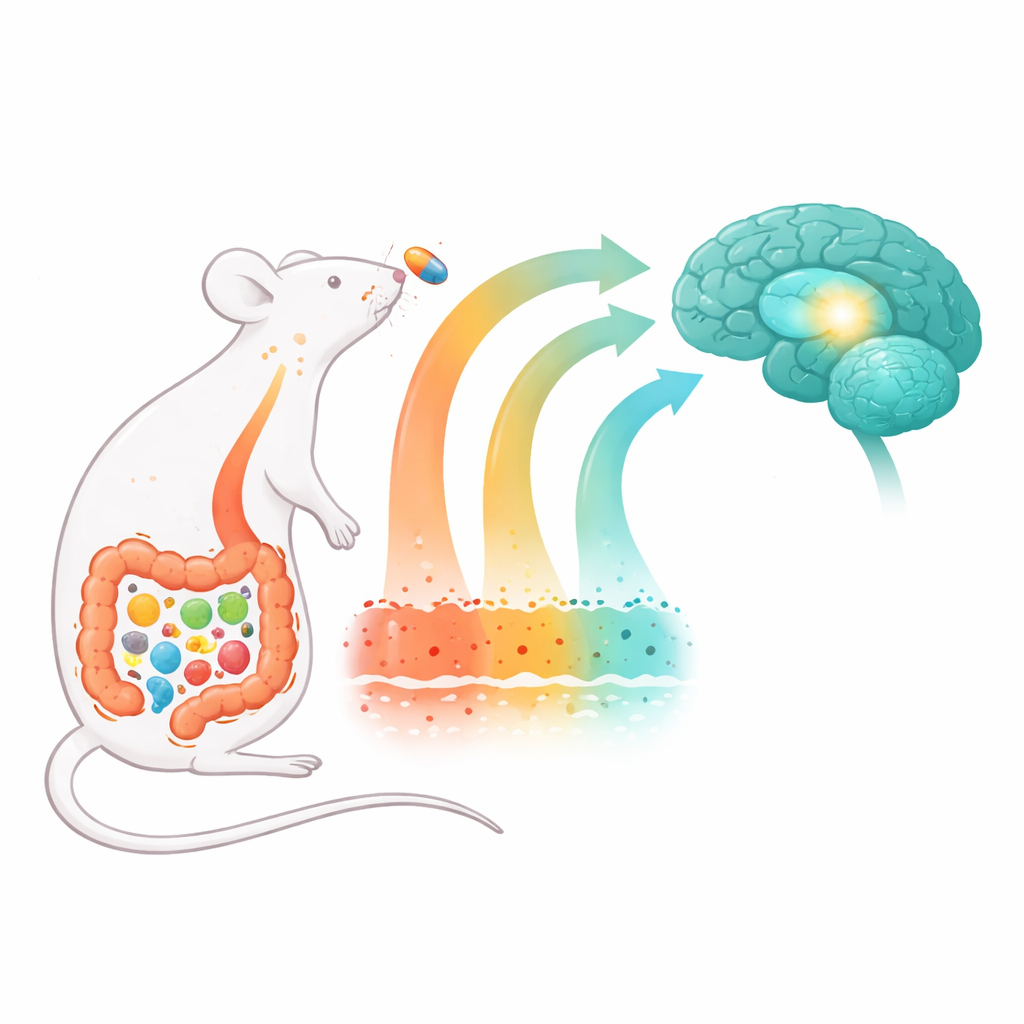
From Shaky Movement to Hidden Inflammation
Parkinson’s disease gradually damages nerve cells that produce dopamine, a chemical that helps control movement. The loss of these cells in a deep brain region known as the substantia nigra leads to the slowness, rigidity, and balance problems seen in patients. Many current drugs boost dopamine temporarily but do not stop the underlying damage, and often lose effectiveness over time. Mounting evidence suggests that long-lasting inflammation—both in the brain and throughout the body—drives this nerve cell loss, making it crucial to find treatments that can cool this chronic "brain-on-fire" state.
Why the Gut Matters for the Brain
In recent years, researchers have uncovered an intimate relationship between gut microbes and brain health, sometimes called the microbiota–gut–brain axis. People with Parkinson’s often experience constipation and other digestive problems years before movement symptoms appear, and their gut bacteria differ from those of healthy individuals. An imbalanced community of microbes can inflame the intestinal wall, weaken its barrier, and allow bacterial products to leak into the bloodstream. These circulating signals can then fuel widespread inflammation and may even help harmful proteins travel from the gut to the brain along nerve pathways.
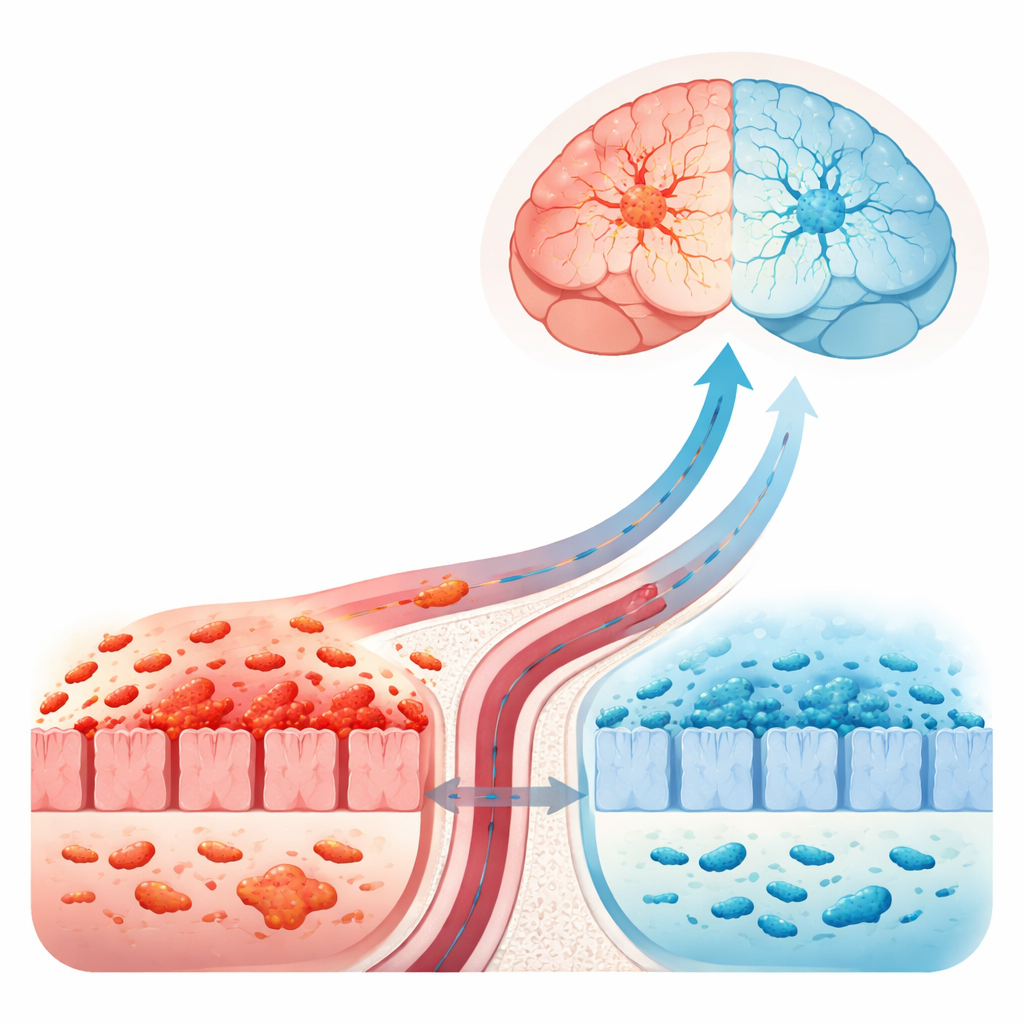
A Plant Compound Put to the Test
To probe this gut–brain connection, the researchers used a well-established mouse model of Parkinson’s created by a chemical called MPTP, which selectively injures dopamine-producing neurons and causes movement problems. Mice were divided into three groups: healthy controls, MPTP-treated animals, and MPTP-treated animals that also received evodiamine by mouth for ten days. The team then evaluated the animals’ motor skills, examined their brains and colons under the microscope, measured inflammatory molecules in their blood, and analyzed the mix of bacteria living in their intestines using genetic sequencing.
Calmer Immune Cells and Stronger Barriers
Mice exposed to MPTP moved more slowly and clumsily, and their brains showed the expected loss of dopamine-producing neurons. The supporting immune cells in the brain—microglia and astrocytes—were highly activated, a sign of neuroinflammation. Their gut tissue and blood also carried high levels of inflammatory markers, and the tight protein “zippers” that normally seal the gut and blood–brain barriers were weakened. Evodiamine treatment improved the animals’ performance on movement tests, preserved more dopamine-producing neurons, and quieted overactive brain immune cells. At the same time, it reduced inflammatory molecules in the brain, gut, and bloodstream, and boosted the proteins that help keep both the intestinal wall and the blood–brain barrier intact, suggesting fewer inflammatory signals were leaking into the body and brain.
Resetting the Microbial Neighborhood
One of the most striking effects of evodiamine was on the gut microbiota itself. MPTP disturbed the normal balance of microbes, decreasing groups of bacteria known to produce short-chain fatty acids—compounds that nourish gut cells and have anti-inflammatory effects—and increasing bacteria linked to gut lining damage and inflammation, such as Akkermansia. Evodiamine partly reversed these changes: beneficial genera like Butyricicoccus, Oscillospira, Ruminococcus, and Coprococcus rebounded, while potentially harmful or overabundant groups declined. Statistical analyses showed that mice harboring more of the helpful bacteria had less systemic inflammation and better motor function, strengthening the idea that microbe shifts are tied to disease severity rather than being a side effect.
What This Could Mean for People
Together, the findings suggest that evodiamine protects vulnerable brain cells in this mouse model not by acting only in the brain, but by orchestrating a chain reaction that begins in the gut. By reshaping the microbial community, reinforcing barrier defenses, and damping a key inflammatory signaling route (the TLR4–MyD88–NF-κB pathway), the compound appears to reduce the inflammatory burden reaching the brain and slow neuronal loss. While these results are early and limited to animals, they add weight to the idea that future Parkinson’s therapies may work best when they treat the gut and the immune system alongside the brain, potentially using safe, plant-based molecules as part of a multi-pronged strategy.
Citation: Wang, S., Zhu, Y., Wang, J. et al. Evodiamine alleviates MPTP-induced Parkinson’s disease in mice by regulating gut microbiota and suppressing TLR4/MyD88/NF-kB pathway. Sci Rep 16, 13353 (2026). https://doi.org/10.1038/s41598-026-43865-2
Keywords: Parkinson’s disease, gut microbiota, neuroinflammation, evodiamine, microbiota–gut–brain axis