Clear Sky Science · en
Persistence of vestibular function in the absence of glutamatergic transmission from hair cells
Why balance without hearing is surprising
Most of us think that if the inner ear is badly damaged, both hearing and balance should fail. This study explores a striking exception: mice that are completely deaf because a key molecule for chemical signaling in their inner ears is missing, yet they still walk, climb, and right themselves almost as well as normal mice. By probing these animals from behavior down to individual synapses, the authors reveal that the balance system has a powerful backup mode of communication that does not rely on the usual chemical packets of neurotransmitter.
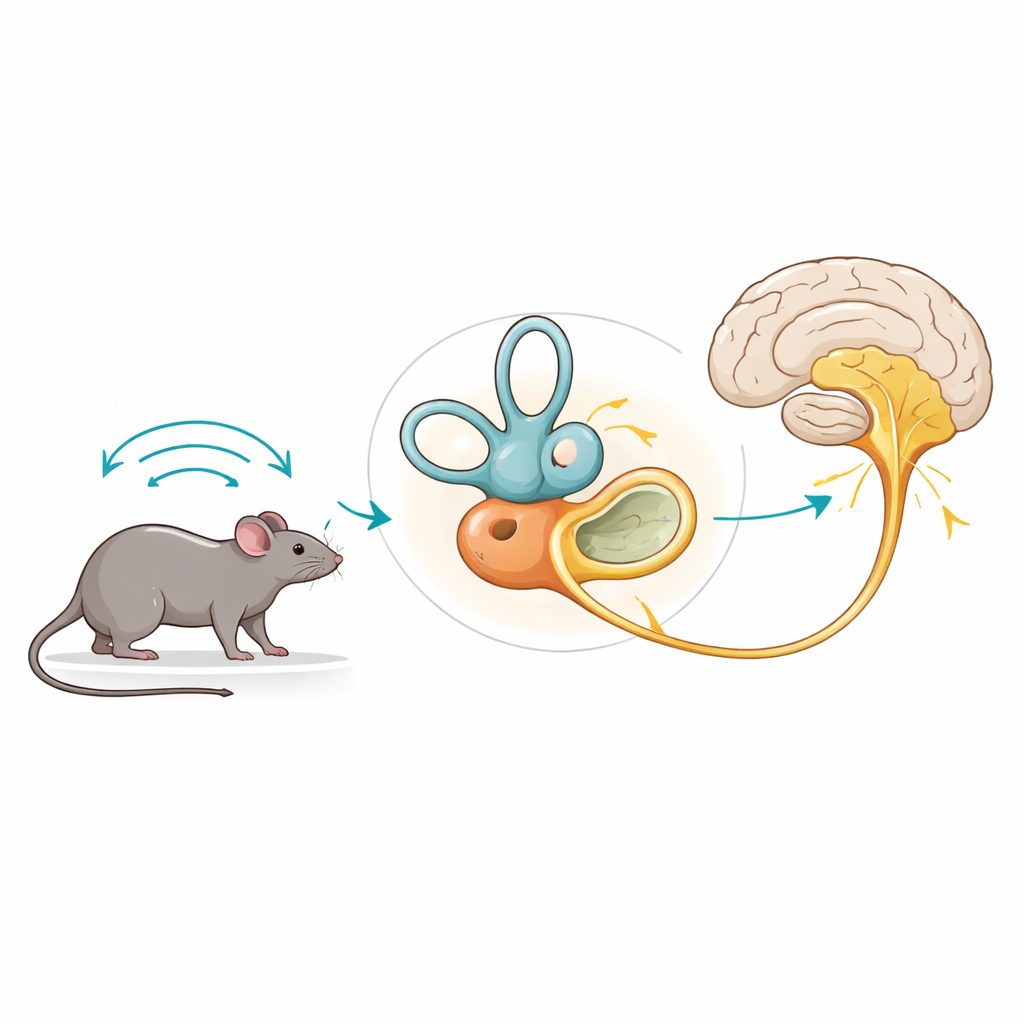
How the inner ear keeps us upright
The organs of balance sit deep in the skull and sense head tilt, gravity, and rotation. They contain tiny sensory cells, called hair cells, that signal to neighboring nerve endings when the head moves. In most parts of the brain and ear, this signaling relies on glutamate, a chemical messenger that is packed into microscopic vesicles by transporters known as VGLUTs. When a hair cell is activated, it releases glutamate-filled vesicles at specialized ribbon synapses, which in turn excite the fibers of the balance nerve that inform the brain about head motion.
A deaf mouse with nearly normal balance
The researchers studied mice lacking VGLUT3, a transporter that is essential for loading glutamate into vesicles in inner hair cells of the cochlea, the hearing organ. As expected, these mice were profoundly deaf. Yet when put through a wide range of balance and coordination tests—walking on rotating rods, climbing screens and poles, swimming, turning upright in mid-air or on a surface—they showed only mild and inconsistent problems compared with normal littermates. Their body weights, general activity, and brain electrical activity were also similar. Even when visual cues were reduced using red light, most balance-related performance remained close to normal, suggesting that the inner ear’s balance organs were still contributing strongly.
Microscopic checks on the wiring
To understand how signaling might persist, the team mapped where different VGLUT proteins and related molecules are expressed in the balance organs. They found that VGLUT3 is strongly present in one class of hair cells (type II) and more weakly or variably in another (type I), particularly in central regions of the utricle. The neighboring nerve endings, shaped like cups called calyces, mainly carried other VGLUT types, VGLUT1 and VGLUT2, which are typical for neurons. Removing VGLUT3 did not cause a wholesale loss of these nerve endings or obvious reshaping of the balance epithelium. There was only a modest reduction in the number of presynaptic ribbons in certain hair cells, far from the severe degeneration seen in the hearing part of the ear when the same transporter is absent.
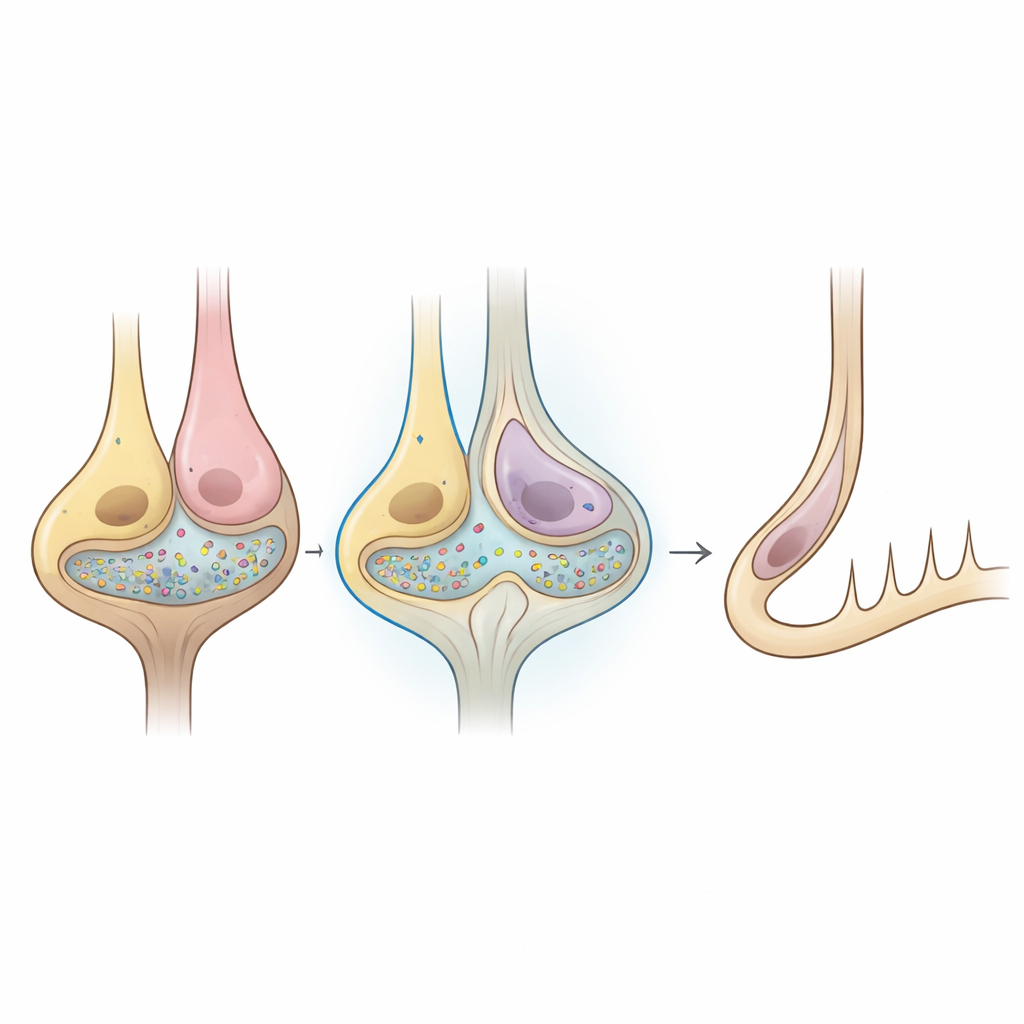
When chemical packets vanish but signals remain
Recordings from individual calyx endings in isolated balance organs showed that the usual glutamate-driven “quantal” events—tiny, fast currents produced by vesicle release—were reduced by more than 95 percent in the mutant mice. Yet these same nerve endings could still fire normal-looking action potentials when injected with current, and in live animals, balance nerve fibers continued to discharge spontaneous spikes at rates and timing regularity similar to controls. In sharp contrast, auditory nerve fibers in the deaf mice were almost completely silent and did not respond to sound. Additional measurements pointed to subtle shifts in ion channels such as HCN and KCNQ in hair cells and calyces, changes that could favor a different mode of communication across the narrow space between them.
A backup line that keeps balance working
The overall picture is that, in these mice, the conventional glutamate vesicle pathway from hair cells to balance nerve endings is largely shut down, but another, “non-quantal” mode of signaling carries enough information to preserve most balance functions. This backup likely relies on rapid changes in voltage and ion concentrations in the tiny gap between the hair cell and calyx, allowing the nerve ending to track head motion without needing clearly defined packets of neurotransmitter. In everyday terms, the balance system appears to have a hard-wired, analog safety line that can keep the brain informed about movement even when the usual chemical messaging is crippled—helping explain why balance can be surprisingly robust when hearing is lost.
Citation: Mukhopadhyay, M., Modgekar, R., Yang-Hood, A. et al. Persistence of vestibular function in the absence of glutamatergic transmission from hair cells. Sci Rep 16, 14550 (2026). https://doi.org/10.1038/s41598-026-43836-7
Keywords: vestibular system, balance, hair cells, synaptic transmission, VGLUT3