Clear Sky Science · en
Development of head-to-head and longitudinal CycleGAN algorithm for MRI harmonization: validation in follow-up MRI evaluation in patients with brain metastasis
Why Matching Brain Scans Matters
When people with cancer develop tumors that spread to the brain, doctors rely on repeated MRI scans to see whether treatment is working. But there is a hidden problem: follow-up scans are often done on different MRI machines, which can make the same tumor look larger, smaller, brighter, or dimmer even when it has not changed at all. This study introduces a new artificial intelligence (AI) method designed to "harmonize" MRI images from different scanners so that doctors can focus on real changes in the tumor rather than on quirks of the machines.
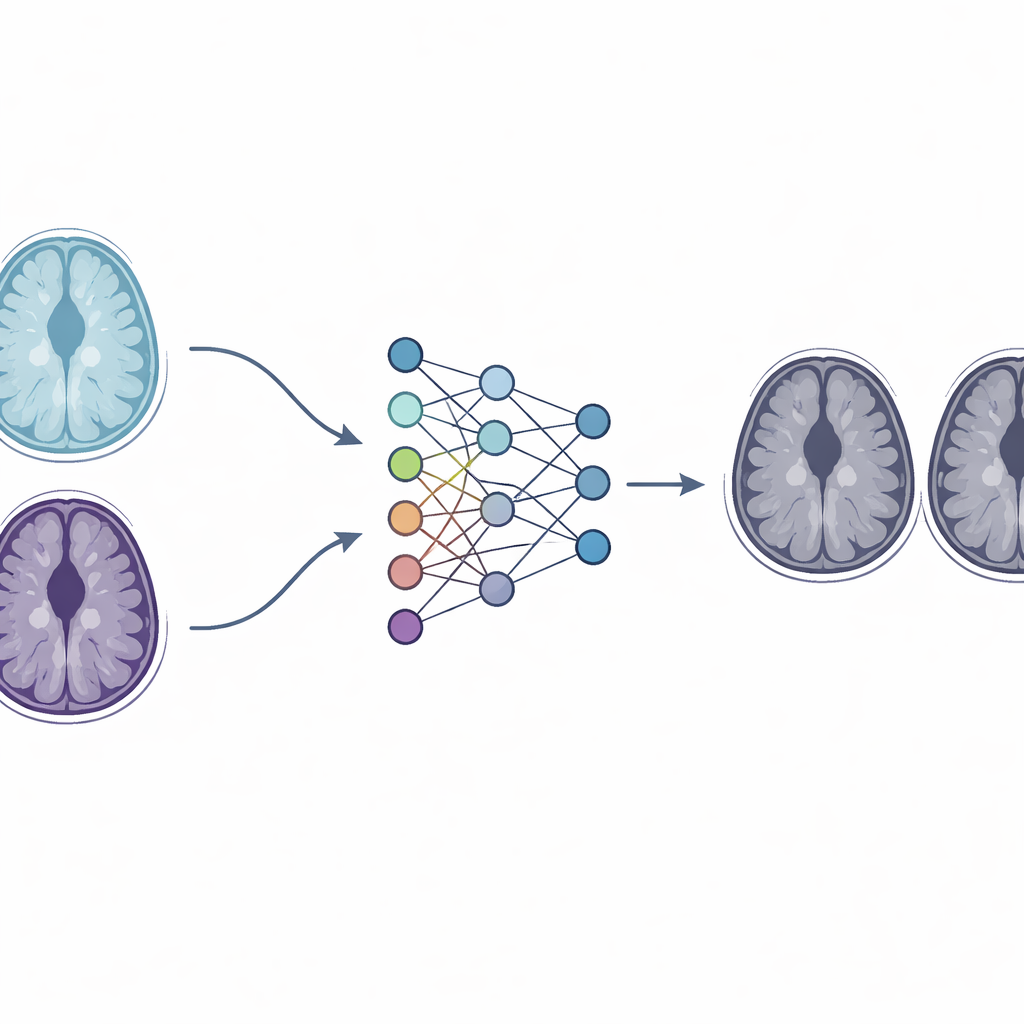
The Challenge of Different MRI Machines
Brain metastases—tumors that have spread to the brain from cancers such as lung or breast cancer—are a major cause of illness and death. Patients often undergo frequent follow-up MRI scans to check whether lesions have grown, shrunk, or stayed the same. Ideally, every scan would be done on the same type of MRI machine using the same protocol. In reality, busy hospitals use multiple scanners from different manufacturers, each with its own settings and contrast behavior. As a result, two scans of the same patient can show the same lesion with different brightness or sharpness, making a stable tumor look suspiciously different. This variability complicates care and can also undermine large research studies that combine data from many centers.
Using Artificial Intelligence to Standardize Images
To tackle this, the researchers built a deep learning system based on a family of neural networks called generative adversarial networks, and specifically a design known as CycleGAN. Instead of trying to change the hardware, they ask software to learn how images from one scanner should look if they had been acquired on another scanner. Their modified "paired" CycleGAN is trained with baseline and follow-up MRI scans from the same patient, where the first and third scans come from the same machine while the middle scan comes from a different one. By comparing these linked images, the network learns to adjust the style of the follow-up scan from the second machine so that it matches the look of the first machine, while preserving the actual anatomy of the brain and tumors.
Keeping the Tumor’s Shape While Fixing the Style
Standard CycleGAN models are good at changing the style of an image, but they risk altering fine details—dangerous in medical imaging. The team therefore added a special "original matching" term to the network’s training objective. This extra constraint pushes the AI to keep every voxel, or 3D pixel, as close as possible to the original scan’s structure, even as it shifts overall contrast and brightness to match the baseline scanner. The authors tested different strengths of this constraint and found a setting that best balanced style correction with faithful preservation of the brain’s anatomy. They then compared their method against conventional tricks like histogram matching and other deep learning approaches, such as Pix2Pix, STGAN, and unmodified CycleGAN.
Sharper Agreement and More Confident Reading
In a group of patients whose brain metastases were known to be stable over time, the harmonized follow-up images looked more similar to the baseline scans than the original, uncorrected follow-ups did, according to several technical image-quality measures. Differences in contrast between various brain regions were also smaller after harmonization, indicating that the scanner-related shifts had been reduced. Two neuroradiologists then reviewed baseline scans alongside either the original or the harmonized follow-up scans, without being told that the lesions were unchanged. With the harmonized images, they more often judged the tumor’s border, size, and contrast to be stable and reported higher confidence in their assessments. Importantly, the AI also preserved real disease changes in a separate test group where tumors truly progressed or regressed, suggesting it was not simply smoothing away clinically important details.

What This Means for Patients
For patients with brain metastases, the new harmonization method could reduce false alarms about tumor growth that are due only to different MRI machines, helping avoid unnecessary worry and potentially unneeded treatment changes. By making follow-up scans visually consistent across scanners, the paired CycleGAN approach gives radiologists a clearer view of what is really happening in the brain. While the study was done in a single center and on a limited set of scanner types, it points toward a future in which smart software routinely normalizes medical images behind the scenes, so that doctors and patients can trust that apparent changes on the screen reflect true changes in disease.
Citation: Hwang, H., Choi, HU., Jeong, H. et al. Development of head-to-head and longitudinal CycleGAN algorithm for MRI harmonization: validation in follow-up MRI evaluation in patients with brain metastasis. Sci Rep 16, 13163 (2026). https://doi.org/10.1038/s41598-026-43755-7
Keywords: brain metastasis, MRI harmonization, deep learning, CycleGAN, medical imaging