Clear Sky Science · en
Mechanistic insights and by-product analysis in sonocatalytic degradation of Orange-G dye molecule via potassium persulfate
Cleaning Up Colorful Wastewater
From brightly dyed clothes to printed packaging, modern life relies on synthetic dyes. But the same vivid colors that brighten our world can linger as stubborn pollutants in rivers and lakes. This article explores a promising way to scrub one such dye, called Orange G, from water by using high-frequency sound waves and a common chemical oxidant. The work shows not only that the color disappears, but that the dye is broken down almost completely into harmless substances.
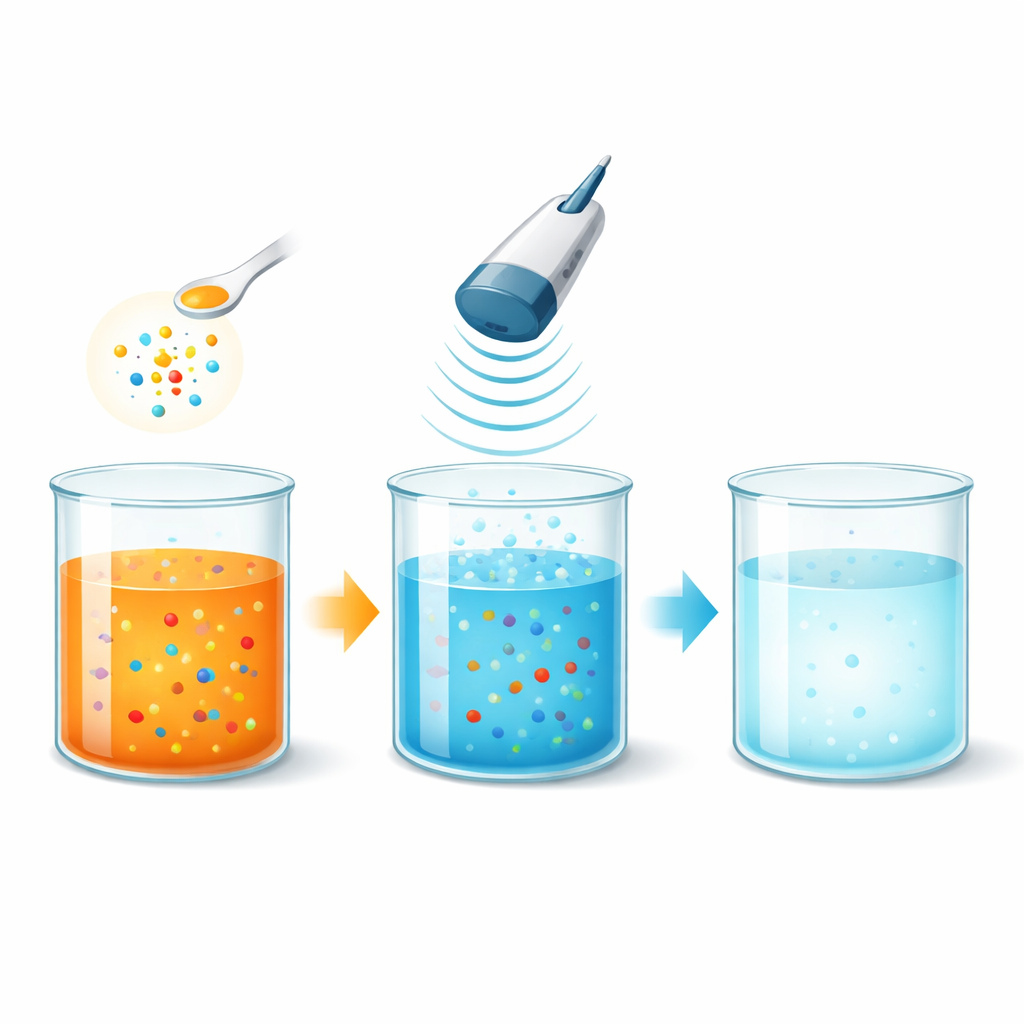
Why Colored Wastewater Is a Problem
Textile, leather, paper, and many other industries use huge amounts of water and release it colored with complex dyes. Many of these dyes are hard to break down naturally, can block sunlight in waterways, and may be toxic to fish, plants, and even humans. Orange G, a widely used orange dye that also appears in laboratory tests, is one such compound. Conventional treatment methods often struggle to remove these molecules fully, sometimes leaving behind smaller but still harmful by-products.
Using Sound and a Helper Chemical
The researchers tested a treatment that combines ultrasound – sound waves at frequencies far above what humans can hear – with potassium persulfate, a relatively inexpensive oxidizing salt. When ultrasound passes through water, it creates tiny bubbles that grow and violently collapse, a process called cavitation. These microscopic “implosions” generate brief, localized zones of very high temperature and pressure. Under these conditions, water and persulfate molecules are shaken apart into extremely reactive fragments known as radicals, which eagerly attack the dye molecules.
Finding the Sweet Spot for Fast Cleaning
To see how well this approach works, the team varied several practical factors: how acidic or basic the water was, how much persulfate they added, how concentrated the dye was, and how much ultrasonic energy they used. They found that near-neutral pH gave the best overall performance, likely because the most effective radicals form and survive well under these conditions. With an optimized persulfate dose, more than 95 percent of Orange G was removed across a broad range of starting concentrations, from lightly colored to strongly dyed water. Increasing the ultrasonic power sped up the process by generating more bubbles and, in turn, more radicals, allowing similar cleaning results in shorter times.
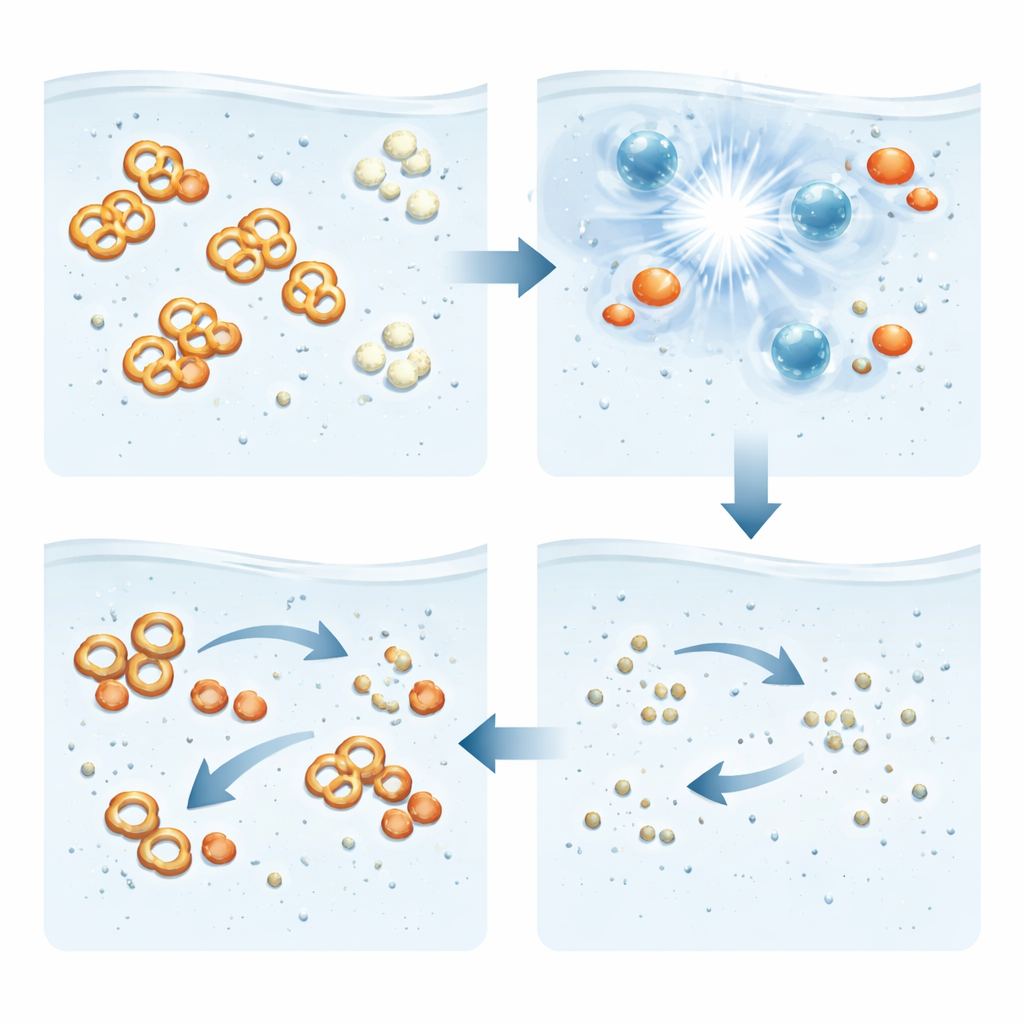
Proving the Dye Is Truly Destroyed
Losing the visible color is not enough; the remaining water must also be free of hidden toxic fragments. To test this, the team measured chemical oxygen demand and total organic carbon, two standard indicators of how much organic material is left. Both dropped sharply, showing that the dye’s carbon-rich structure was largely converted to simple end products like carbon dioxide. Advanced tools such as electron paramagnetic resonance confirmed that the key radicals were indeed being formed in the liquid, while chromatography and mass spectrometry revealed that the large Orange G molecules were chopped into smaller pieces and then further broken down, leaving only minor traces of short-lived intermediates.
Robust Performance in Realistic Conditions
Industrial waste streams often contain salts such as sodium chloride or magnesium sulfate, which can sometimes interfere with treatment processes. The authors tested several common salts at realistic levels and found that they had very little effect on the overall removal of Orange G, which consistently stayed above 95 percent. This robustness suggests that the ultrasound–persulfate combination could be applied to a variety of real-world wastewaters without extensive pre-treatment. The process also followed predictable “pseudo-first-order” behavior, meaning its speed can be described with a simple mathematical relationship, which is useful for designing and scaling up reactors.
What This Means for Cleaner Water
For a non-specialist, the key message is that there is a practical way to strip out stubborn dyes from water using sound and a relatively simple chemical. The method does more than just bleach the color: it almost completely mineralizes the dye, leaving behind mainly carbon dioxide, water, and harmless salts. Because it works well over a wide range of dye levels, tolerates dissolved salts, and responds predictably to changes in energy input, this approach could be adapted for industrial treatment plants. While further work is needed on cost and large-scale engineering, the study points toward a cleaner future in which even the most persistent artificial colors can be safely removed from our water.
Citation: R, B., D, V., S, R. et al. Mechanistic insights and by-product analysis in sonocatalytic degradation of Orange-G dye molecule via potassium persulfate. Sci Rep 16, 14333 (2026). https://doi.org/10.1038/s41598-026-43751-x
Keywords: wastewater treatment, ultrasound oxidation, azo dye removal, potassium persulfate, advanced oxidation