Clear Sky Science · en
Dual functional rGO/Fe3O4/PANI nanocomposite anodes for enhanced performance of microbial fuel cells
Turning Waste Into Power and Cleaner Water
Modern life produces two big problems: mountains of organic waste and streams of water laced with toxic metals such as chromium and lead. Treating that water usually costs energy instead of producing it. This study explores a different path: using living microbes and a smartly engineered electrode to clean heavy-metal-contaminated wastewater while generating electricity at the same time. The work shows how a new, low-cost material made from agricultural leftovers can boost the performance of tiny biological power plants called microbial fuel cells.
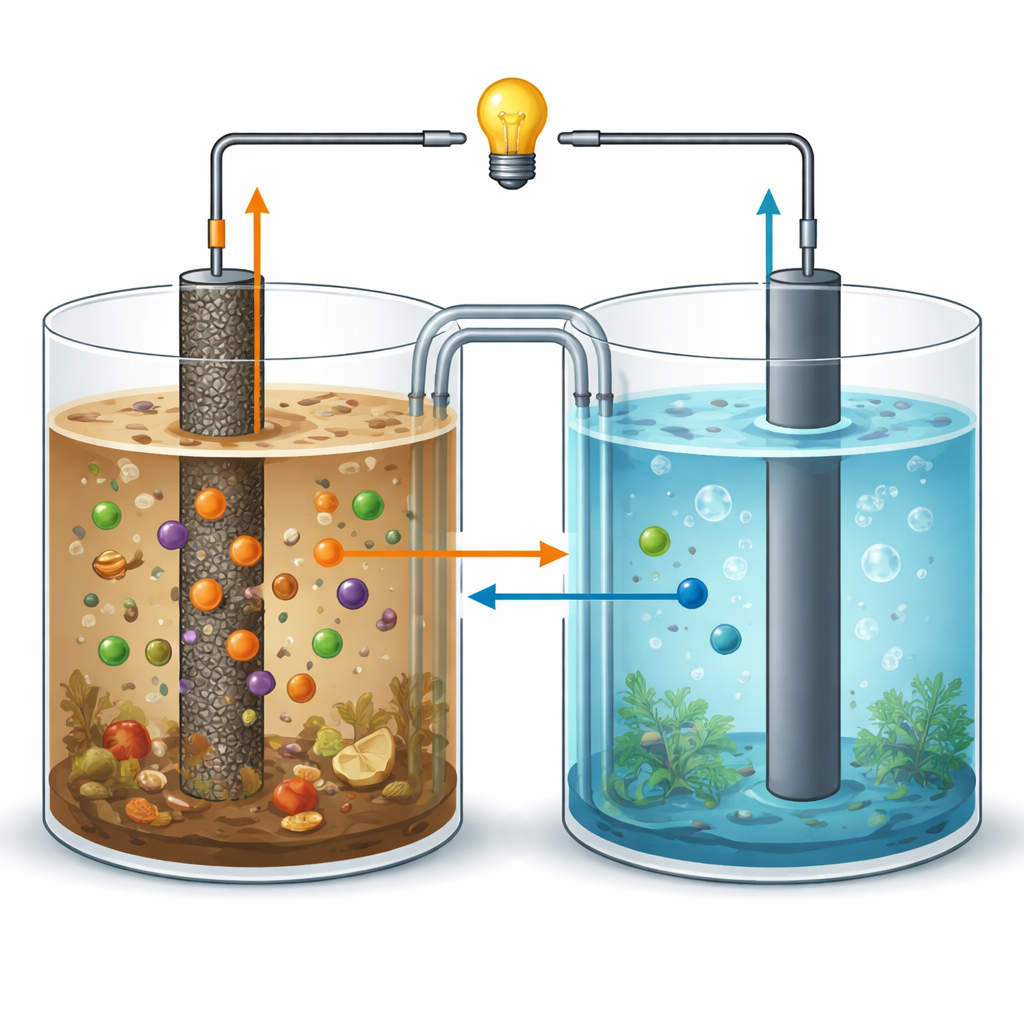
A Tiny Power Plant Run by Microbes
Microbial fuel cells act a bit like living batteries. In a sealed chamber, naturally occurring bacteria feed on organic matter, such as chopped sweet potato waste suspended in pond water. As they digest this food, the microbes release electrons and protons. The electrons flow to a solid surface called the anode, travel through an external wire to a second electrode (the cathode), and create an electric current. At the same time, the water around the microbes can be cleaned as pollutants are broken down or transformed into safer forms. In principle, this lets us recover energy from waste while purifying water, but in practice most devices produce very little power and only modest pollutant removal.
Building a Better Electrode From Plant Waste
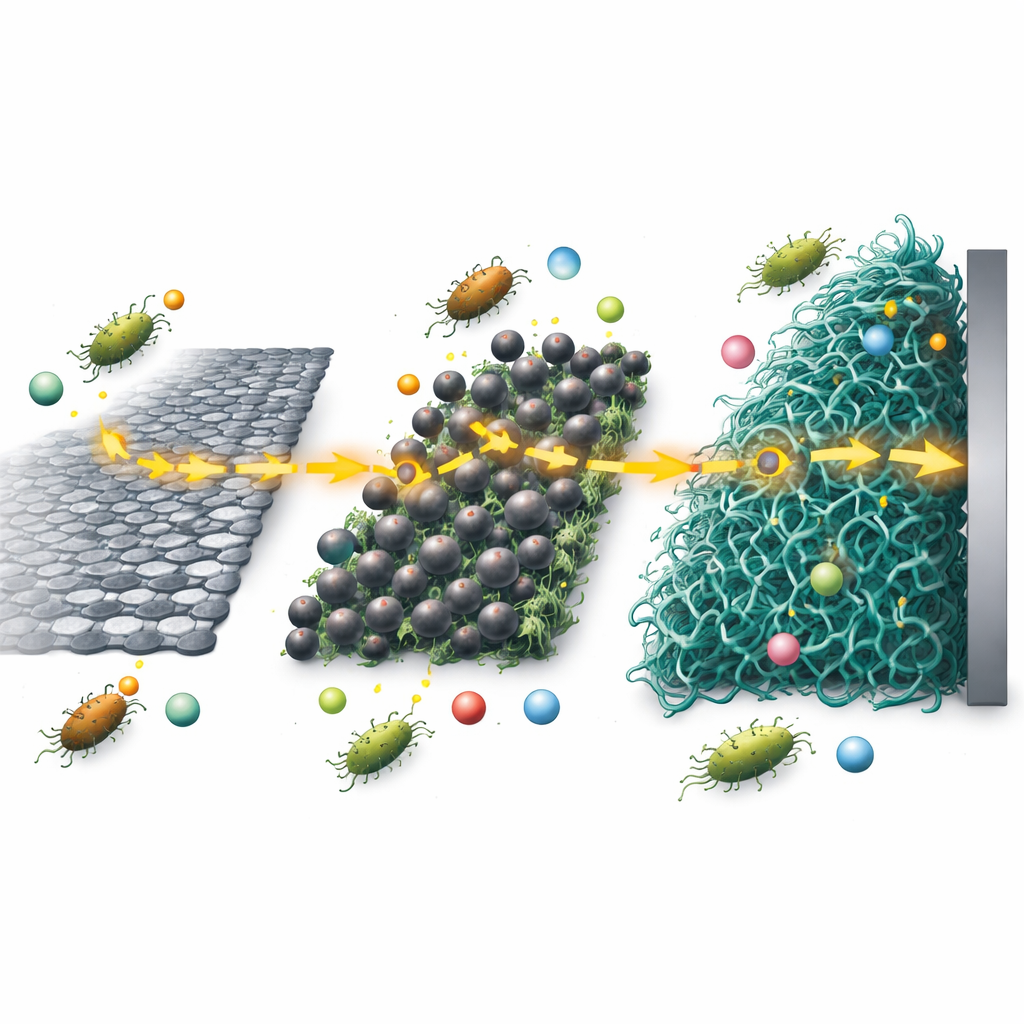
The weak point in many microbial fuel cells is the anode surface where microbes hand off their electrons. Common carbon materials are often too smooth, not conductive enough, or unfriendly to microbial growth. The researchers tackled this by turning discarded corms from the enset plant—a staple crop in Ethiopia—into a high-performance carbon material. They first converted the plant waste into graphene oxide, then chemically reduced it to form thin, wrinkled sheets of reduced graphene oxide with plenty of surface area for microbes to cling to. To further improve performance, they added tiny iron oxide particles and coated the whole structure with a thin layer of a conducting plastic called polyaniline, creating a three-part (ternary) nanocomposite anode.
How the New Material Helps Microbes and Grabs Metals
Microscope images and spectroscopy showed that the iron oxide particles and polyaniline spread evenly over the graphene sheets, creating a rough, porous, and strongly connected network. This structure offers many nooks and crannies for microbes to attach, while the conducting pathways help electrons move quickly from the cells into the circuit. Electrical tests in a simple liquid environment revealed that the new anode had much stronger redox activity and much lower resistance to charge flow than bare graphite or graphene alone. In the fuel cell, this translated into a higher open-circuit voltage, more current over a month of operation, and a significant drop in internal energy losses.
Powering Up While Pulling Out Toxic Metals
To test real-world usefulness, the team filled a two-chamber device with sweet potato waste and pond water spiked with high levels of chromium(VI) and lead(II), two particularly harmful metal ions. Using the new nanocomposite as the anode, the microbial fuel cell reached a peak power density of about 65 milliwatts per square meter—around eight times higher than with the simpler graphene anode—and more than doubled the current density. Just as important, the system removed 88% of the chromium and 86% of the lead over 30 days, clearly outperforming both plain graphite and unmodified graphene electrodes. The metals are either converted into less harmful forms or trapped as insoluble compounds on or near the anode surface.
Steps Toward Practical Green Treatment Systems
In everyday terms, this work shows that a carefully designed electrode made from plant waste and common chemicals can help microbes do two jobs at once: generate electricity and clean up toxic metals from water. While the power output is still modest compared with the most advanced lab-scale devices, the gains in both energy recovery and metal removal are substantial for a system aimed at real wastewater. The authors note that future work must test long-term stability, scale up the design, and study which microbial communities are most active. Even so, the study offers a promising blueprint for low-cost, sustainable devices that turn agricultural leftovers and polluted water into a useful source of clean energy and cleaner effluent.
Citation: Weldegrum, G.S., Zemedagegnehu, D.A. Dual functional rGO/Fe3O4/PANI nanocomposite anodes for enhanced performance of microbial fuel cells. Sci Rep 16, 14000 (2026). https://doi.org/10.1038/s41598-026-43694-3
Keywords: microbial fuel cells, heavy metal removal, biomass-derived graphene, wastewater treatment, waste-to-energy