Clear Sky Science · en
Scalable electrochemical CO2 reduction to oxalate in a continuous flow reactor
Turning a climate problem into a useful product
Carbon dioxide is usually seen as a waste gas that drives climate change, but it is also a raw material waiting to be reused. This study explores a way to turn CO2 into oxalate, a solid chemical widely used in industry, using electricity in a compact flow reactor. The work shows that thoughtful reactor design can make this conversion efficient, scalable, and compatible with cleaner energy systems.
From waste gas to valuable solid
The heart of the research is a process that feeds CO2 into a liquid and uses an electric current to stitch pairs of CO2 molecules together into oxalate. Oxalate and its acid form, oxalic acid, are already important in textiles, metals, pharmaceuticals, and advanced materials. Each oxalate molecule locks in two carbon atoms in a stable form, so making it from CO2 not only supplies a useful commodity but can also help remove carbon from the atmosphere, especially in regions where electricity comes mainly from renewable sources.
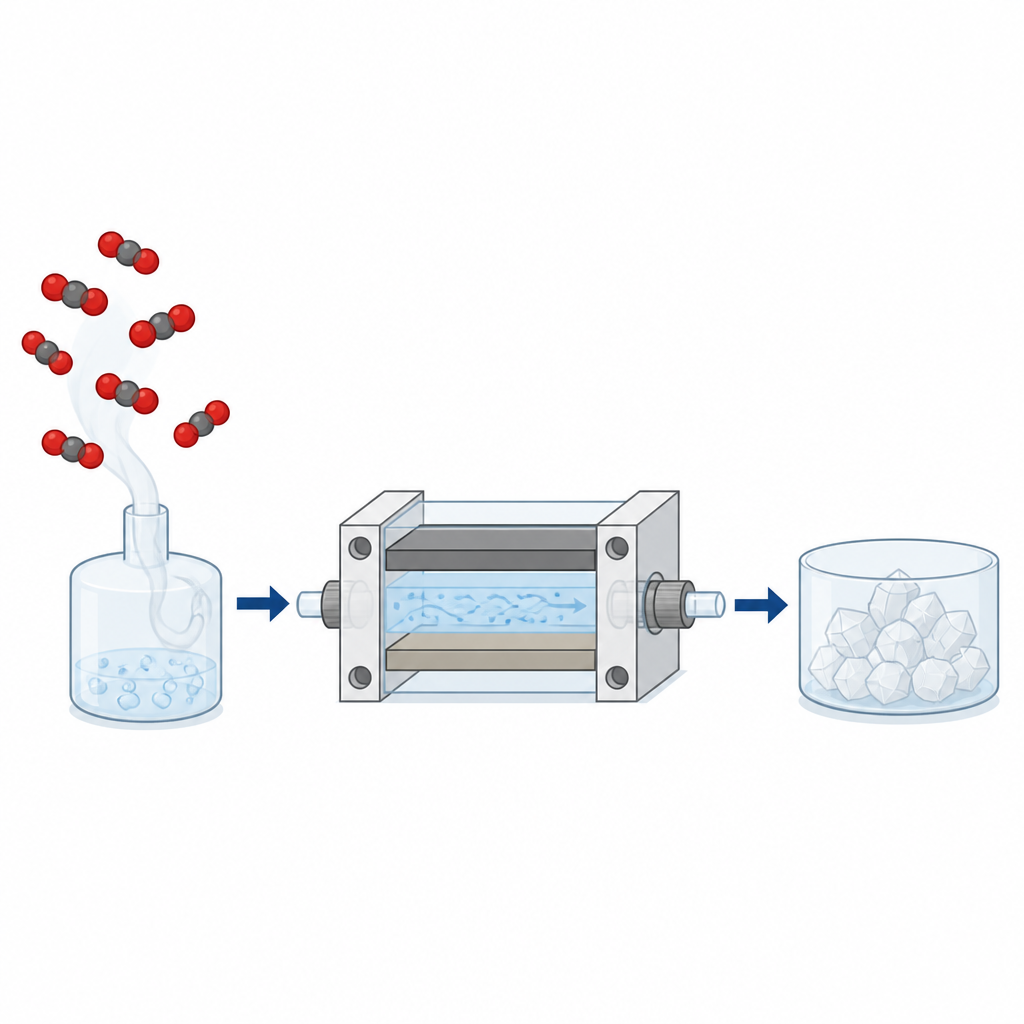
A new type of flowing reaction cell
Many earlier experiments used simple, still reactors and often relied on lead electrodes, which are effective but toxic and unsuitable for large scale use. In contrast, this work uses a stainless steel cathode paired with a zinc anode inside a small, 3D printed flow reactor. The liquid containing dissolved CO2 is pumped continuously through a narrow channel between two flat metal plates. By varying how far apart the plates are and later by enlarging their area, the team could test how geometry affects performance while the system keeps running in a steady, production like mode.
Balancing distance, flow, and performance
The researchers carefully mapped how the reactor behaves at different voltages and electrode gaps of 2, 1, and 0.5 millimeters. A smaller gap lowers electrical losses and improves the supply of CO2 to the metal surface, which boosts the current and makes the process more energy efficient. However, pushing the gap down to 0.5 millimeters led to clogging, because the solid zinc oxalate crystals formed so quickly that they began to block the narrow channel. The best overall tradeoff came from a 1 millimeter spacing, which reached a Faradaic efficiency of 72 percent and current densities above 130 milliamps per square centimeter at about 4 volts, outperforming previous flow based systems and rivaling many still reactors that operate more slowly.
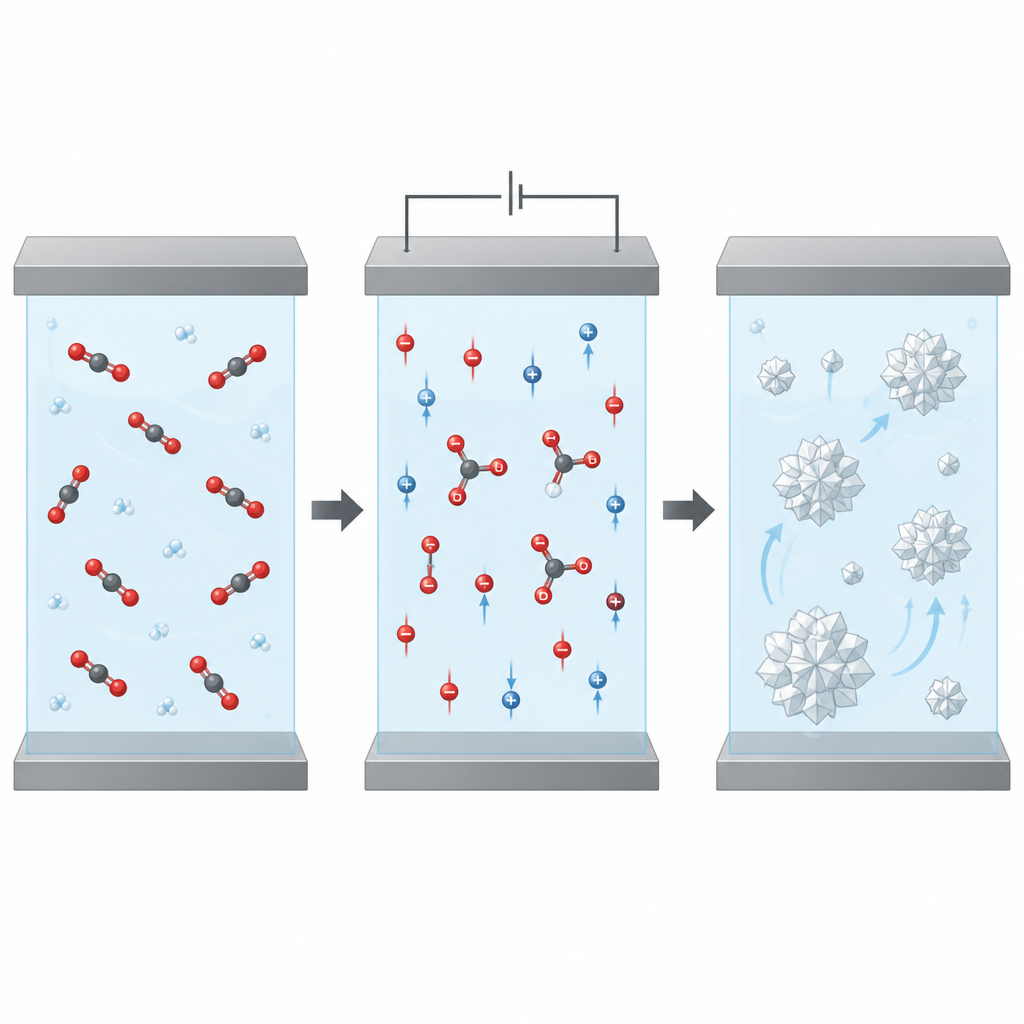
Looking inside the process
Beyond raw performance, the team studied how different kinds of electrical losses add up in the reactor. They measured how much voltage is wasted simply pushing current through the liquid and how much comes from limits in how fast CO2 can reach the electrode. Impedance tests, which record how the system responds to small electrical signals over a wide range of frequencies, helped separate these effects. The results showed that shrinking the gap mainly cuts ohmic losses, while mass transport limitations only become important at higher driving voltages. This insight supports the idea that careful shaping of the reactor can be as important as discovering new catalysts.
Scaling up without losing efficiency
To probe real world potential, the authors scaled the electrode area from 10 square millimeters to 656 square millimeters in new reactor versions while keeping similar gaps. The larger devices preserved competitive efficiencies but greatly increased oxalate production in much shorter operating times. Energy use for many operating points fell between about 5 and 15 kilowatt hours per kilogram of oxalate, which compares favorably with values reported for several other electrochemical CO2 conversion routes and is far lower than that of mature water splitting technologies used to make hydrogen.
Why this matters for cleaner industry
In simple terms, the study shows that a well designed flow reactor made from practical materials can turn CO2 into a useful solid at high rates without extreme energy costs. While more work is needed to fine tune efficiency, prevent clogging, and move toward continuous operation, the approach points toward future plants where waste CO2 is directly converted into oxalate as both an industrial feedstock and a form of stored carbon. Instead of venting CO2 into the air, factories could route it through reactors like these, helping to close the carbon loop.
Citation: Dionisio, D., Narváez-Romo, B., Ribeiro, L.N.B.S. et al. Scalable electrochemical CO2 reduction to oxalate in a continuous flow reactor. Sci Rep 16, 14913 (2026). https://doi.org/10.1038/s41598-026-43540-6
Keywords: CO2 utilization, electrochemical conversion, oxalate production, flow reactor, carbon storage