Clear Sky Science · en
Deciphering novel genotypic biomarkers and functional alterations in double strand break repair pathways for Parkinson’s disease
Why tiny DNA glitches matter for brain health
Parkinson’s disease is best known for its shaking hands and slowed movements, but deep inside brain cells another drama may be unfolding: damage to the DNA that keeps those cells alive. This study asks a simple but powerful question: do inherited weaknesses in the way our cells repair serious DNA breaks make some people more likely to develop Parkinson’s disease? By probing the genes that patch up particularly dangerous breaks in DNA, the authors uncover new clues to who is at risk and why.
Parkinson’s disease and fragile brain cells
Parkinson’s disease is one of the fastest-growing brain disorders worldwide, especially in older adults. Its core problem is the gradual loss of dopamine-producing nerve cells and the buildup of clumps of a protein called alpha-synuclein. These neurons are under constant stress. They burn a lot of energy, generate reactive oxygen byproducts, and have very limited ability to regenerate. All of this makes their DNA especially vulnerable to damage. If breaks in the DNA strands are not fixed properly, the cell’s genetic blueprint becomes unstable, setting off self-destruct programs and contributing to slow but relentless neurodegeneration.
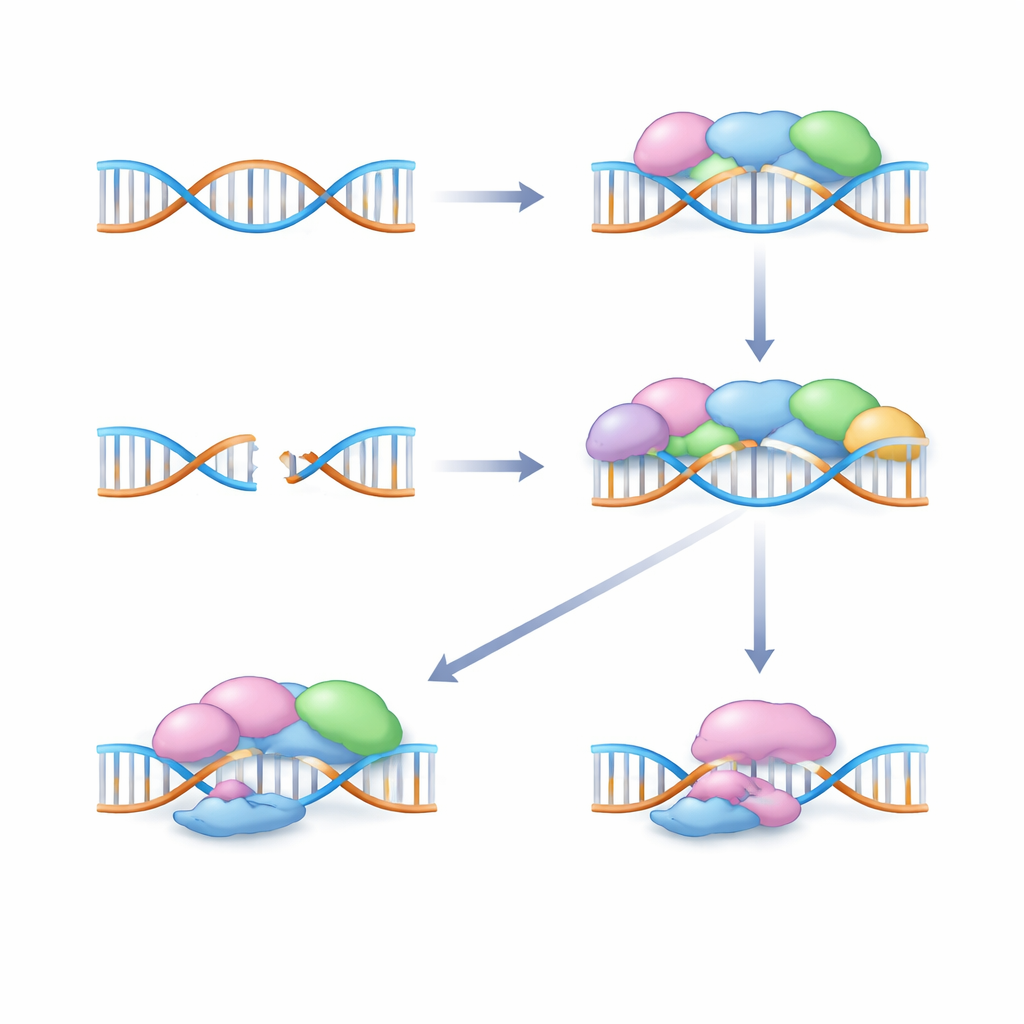
How cells mend dangerous DNA breaks
Among the worst forms of DNA damage are double-strand breaks, where both rails of the DNA ladder snap. Cells rely on specialized pathways to detect and repair these breaks. One major route, called non-homologous end-joining, quickly glues the broken ends together using a team of proteins encoded by genes such as XRCC4, XRCC5, XRCC6, XRCC7, and LIG4. Another crucial team is the MRN complex, made from the MRE11, RAD50, and NBN genes, which senses breaks and helps decide how they should be repaired. Subtle DNA spelling differences, or polymorphisms, in these genes can change how much of each repair protein is made or how well it works, potentially tipping the balance between efficient repair and accumulating damage.
Testing DNA repair genes in people with and without Parkinson’s
The researchers examined 123 Taiwanese patients with clinically diagnosed Parkinson’s disease and 492 healthy people of similar age and sex. They focused on specific genetic variants in eight key double-strand break repair genes. For the four most promising variants, they went a step further: in a subgroup of healthy volunteers, they measured how much messenger RNA each gene produced and how well the volunteers’ blood cells could repair DNA damage in the lab. These functional tests included a reporter assay for the main double-strand break repair pathway and a “comet” assay, which visually tracks how quickly broken DNA is restored over time.
Four risky gene variants and a standout culprit
Four genetic variants stood out as being linked to Parkinson’s risk: one each in XRCC6, XRCC4, RAD50, and NBN. People carrying the higher-risk forms of these variants were more likely to have Parkinson’s disease than those with the common forms. When the team counted how many of these risk variants each person carried, they saw a clear dose effect: the more risky combinations someone had, the higher their odds of Parkinson’s. One variant in particular, in the XRCC6 gene, emerged as a key player. Individuals with two copies of its rare version had much higher odds of having Parkinson’s. In blood cells from healthy carriers, this version produced less XRCC6 messenger RNA and showed weaker ability to mend double-strand breaks, pointing to a direct link between the inherited change and reduced repair power.
What weakened DNA repair may mean for the brain
Although some risk variants were uncommon and the functional experiments involved a relatively small number of people, the findings paint a consistent picture. Inherited changes in a core DNA repair gene, XRCC6, and in related partners XRCC4, RAD50, and NBN, appear to undermine the machinery that fixes the most dangerous DNA breaks. For neurons already stressed by aging, oxidative chemistry, and protein buildup, such lifelong repair weaknesses could push them closer to failure, increasing the chance of Parkinson’s disease. The work does not yet translate into a test your doctor can order, and it was conducted in a single ethnic group. Still, it suggests that measuring and strengthening double-strand break repair could one day help identify high-risk individuals earlier and guide more personalized approaches to preventing or slowing Parkinson’s disease.
Citation: Chen, CH., Tsai, CW., Chang, WS. et al. Deciphering novel genotypic biomarkers and functional alterations in double strand break repair pathways for Parkinson’s disease. Sci Rep 16, 13173 (2026). https://doi.org/10.1038/s41598-026-43533-5
Keywords: Parkinson’s disease, DNA repair, genetic risk, double-strand breaks, neurodegeneration