Clear Sky Science · en
Cross-linked PVA/PSSA-CNTs based polyelectrolyte membranes with enhanced proton conductivity for fuel cell applications
Cleaner Power from Better Plastics
Imagine running your phone, laptop, or even your car on a small, silent box that turns liquid fuel directly into electricity, releasing almost no pollution. That is the promise of fuel cells. But the heart of these devices—a thin plastic sheet that lets certain particles through while blocking others—still has drawbacks: it can be expensive, fragile, and leaky. This study presents a new kind of plastic membrane designed to move electrical charge efficiently while wasting as little fuel as possible, potentially making cleaner power sources more practical and affordable.
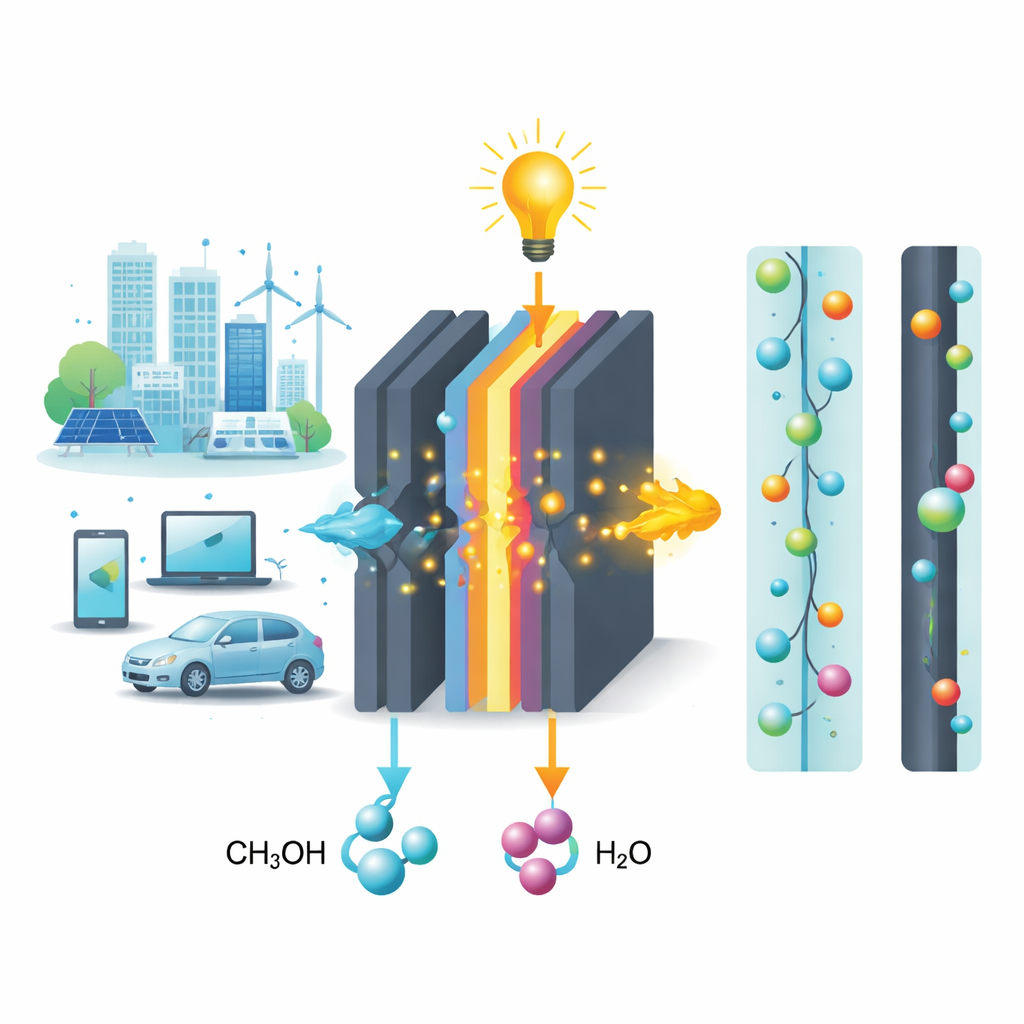
Why Fuel Cells Need Smart Filters
Direct methanol fuel cells use a simple alcohol, similar to what is found in some fuels and solvents, mixed with water to generate electricity. Between the two sides of the cell sits a membrane that has to do two jobs at once: it must let positively charged particles (protons) move freely to carry current, but it must stop methanol from slipping through, which wastes fuel and lowers performance. Traditional commercial membranes, such as widely used fluorinated plastics, conduct protons well but allow too much methanol to cross and are costly to make. The challenge is to design a membrane that strikes a better balance between strength, conductivity, and fuel-tightness, using cheaper and more environmentally friendly ingredients.
Building a Tough, Conductive Film
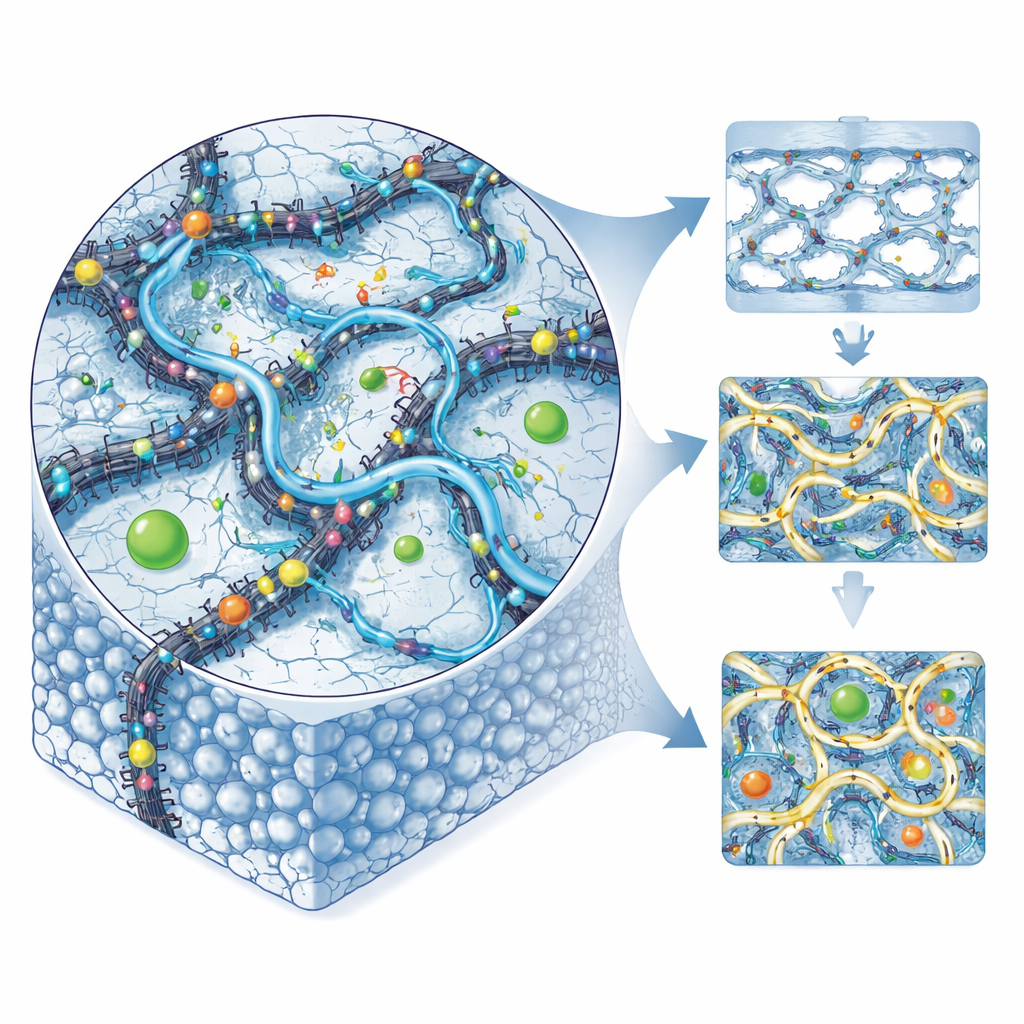
The researchers started from polyvinyl alcohol, a common, water‑loving plastic already known for forming smooth, flexible films. By itself, however, this material softens too much in water and does not carry protons very efficiently. To upgrade it, the team blended in carbon nanotubes—tiny hollow cylinders of carbon—whose surfaces were carefully treated with an acid-containing polymer. This coating adds many sites that can shuttle protons while helping the nanotubes spread evenly through the plastic instead of clumping. The scientists then "locked" the whole mixture into a three‑dimensional network using small organic acids as connectors, tightening the structure so it would resist swelling and remain mechanically strong during operation.
Peeking Inside the New Membranes
Using a combination of imaging and chemical probes, the authors showed that the nanotubes remained tube‑like and became well dispersed throughout the plastic matrix. Electron microscope images revealed that adding the treated nanotubes and crosslinkers transformed the originally smooth, waxy film into a denser, sponge‑like material with tiny surface pores that do not run all the way through. This kind of structure helps water and protons find connected pathways while forcing methanol to take a more winding route, making it harder for the fuel to leak. Heat‑testing experiments also indicated that the modified films stay stable at temperatures far above those typically used in low‑temperature fuel cells, while measurements of stretching and breaking showed that tensile strength could rise by roughly 60 percent or more compared with the unmodified plastic.
Moving Charge Without Wasting Fuel
The team then measured how much water and methanol the membranes absorb, how easily they swell, and how well they ferry ions. Adding more of the functionalized nanotubes increased both the number of charge‑carrying sites and the membrane’s ability to hold water, both of which boost proton transport. At the same time, the cross‑linked network limited swelling and narrowed the channels that larger methanol molecules must navigate. One particular recipe, based on polyvinyl alcohol combined with one percent of the treated nanotubes and linked with succinic acid, stood out. It showed relatively high proton conductivity, several times that of the commercial benchmark Nafion‑117, yet its methanol permeability was about three orders of magnitude lower, meaning much less fuel slipped through. When the researchers combined these two measures into a single "efficiency" factor, the best new membrane vastly outperformed the commercial film.
What This Could Mean for Everyday Energy
In plain terms, the study demonstrates a plastic film that is strong, heat‑resistant, and able to move the particles that carry electrical current while strongly resisting fuel loss. Because it is built from relatively simple polymers, small organic acids, and carbon nanotubes that can be processed by common casting methods, it points toward more affordable and sustainable membranes for direct methanol fuel cells. If scaled up and integrated into real devices, such membranes could help make compact, quiet, low‑emission power sources more efficient and practical for portable electronics, backup power, and possibly vehicles, bringing cleaner energy technology a step closer to everyday use.
Citation: El-Desouky, E.A., Soliman, E.A., El-Bardan, A.A. et al. Cross-linked PVA/PSSA-CNTs based polyelectrolyte membranes with enhanced proton conductivity for fuel cell applications. Sci Rep 16, 10921 (2026). https://doi.org/10.1038/s41598-026-43521-9
Keywords: fuel cells, proton exchange membranes, carbon nanotubes, polyvinyl alcohol, methanol crossover