Clear Sky Science · en
Integrated proteomic and single-cell transcriptomic profiling elucidates immunomodulatory effects of L-serine in autism spectrum disorder
Why an amino acid matters for autism
Families affected by autism spectrum disorder often hear that there are no medicines for the core social and behavioral differences, only for associated problems such as anxiety or irritability. This study explores an unexpected candidate for helping with those core features: L-serine, a naturally occurring amino acid. By looking closely at the blood and immune cells of children with autism before and after 12 weeks of L-serine, the researchers ask a deceptively simple question: can changing body chemistry gently reset parts of the immune system that may be tangled up with brain development and behavior?
Looking at the immune system instead of the brain
Directly sampling brain tissue from children is neither ethical nor practical, so the team turned to blood as an accessible “mirror” of what might be happening in the body. They focused on two main actors in the bloodstream. The first were tiny membrane packages called extracellular vesicles, which carry proteins and other signals between cells. The second were individual immune cells, captured and analyzed one by one using single-cell RNA sequencing, a technique that reveals which genes are switched on in each cell. Together, these two approaches provide a wide-angle and close-up view of how the immune system behaves before and after L-serine treatment.
A small trial with measurable behavioral changes
Eleven children with autism, aged 2 to 11, received a carefully weight-adjusted dose of oral L-serine every day for 12 weeks. They were not taking other psychiatric medicines or amino acid supplements, so any changes could be more clearly linked to the study drug. Clinicians tracked overall functioning and autism-related behaviors using standard rating scales, including the Clinical Global Impression. Over the three months, scores generally moved in a favorable direction: children were rated as less severely affected and as showing meaningful improvement in day-to-day communication, socialization, and daily living skills. Although this was not a randomized, placebo-controlled trial, these clinical shifts motivated a deeper look into what was changing inside the blood.
Tuning key immune cells in the bloodstream
In the vesicles circulating in plasma, the researchers detected more than 900 different proteins, with over 200 showing large shifts after L-serine. Many of these proteins belonged to pathways that control T cell activity, especially CD4 T cells that coordinate immune responses. Using computational tools, they traced much of the vesicle cargo back to CD4 T cells and related immune cell types. When they zoomed in with single-cell sequencing, they found that children with autism had an unusually expanded subset of so-called naive CD4 T cells marked by low levels of a surface molecule called IL7R. After L-serine treatment, this skewed pattern softened, and the gene activity in these cells moved closer to that seen in healthy, age-matched children. A core set of about 20 genes that had stood out as abnormal shifted toward more typical levels, particularly genes involved in how T cells mature and communicate.
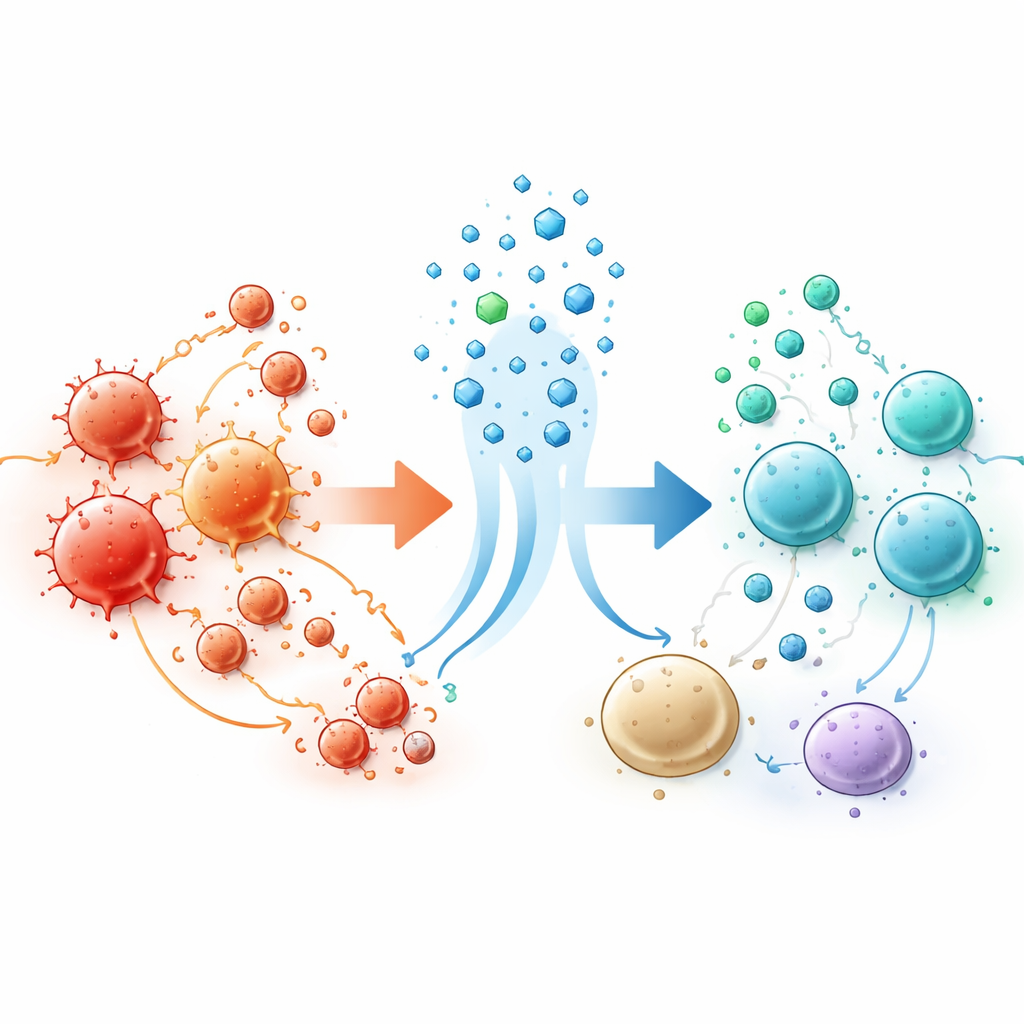
Rewiring how immune cells talk to each other
Beyond the identity of individual cells, the team examined how different immune cell types appeared to “talk” to each other through matching pairs of signaling molecules on their surfaces. Before treatment, CD4 T cells formed dense networks of predicted interactions with other immune cells, including monocytes, natural killer cells, and CD8 T cells. After L-serine, some of these interactions quieted, while new patterns emerged that suggested a more balanced exchange, including changes in molecules carried by vesicles. Computational network analysis showed that groups of genes controlling T cell activation and metabolic state were dialed down or up in coordinated ways. These shifts support the idea that L-serine nudges naive CD4 T cells out of a developmental bottleneck and toward a more mature, stable role in the immune system, without ramping up harmful inflammation.
What this could mean for children and families
For non-specialists, the central message is that an everyday molecule like L-serine may help restore balance in the immune systems of some children with autism, and these immune changes track with measurable behavioral gains. The study does not prove that L-serine is a cure, nor does it show cause and effect in the way a large, blinded clinical trial would. But by combining protein profiling of circulating vesicles with single-cell gene readouts, the work offers a detailed, mechanistic picture: L-serine appears to reshape specific T cell populations and their communication networks in ways that could feed back on brain development and behavior. This immune “retuning” may become an important piece of the puzzle as researchers look for safer, biology-informed treatments that address the underlying systems involved in autism, rather than only its outward symptoms.
Citation: Jang, J., Yeo, S., Kim, J.P. et al. Integrated proteomic and single-cell transcriptomic profiling elucidates immunomodulatory effects of L-serine in autism spectrum disorder. Sci Rep 16, 14210 (2026). https://doi.org/10.1038/s41598-026-43467-y
Keywords: autism spectrum disorder, L-serine, immune system, T cells, single-cell sequencing