Clear Sky Science · en
Photocatalytic degradation of Congo red dye using innovative cerium titanate nanorods embedded in a cellulose-based hydrogel
Cleaning Up Colorful Water
Brightly colored dyes make our clothes and fabrics appealing, but when they wash out of factories into rivers, they can linger for years and harm living things. This study explores a new, sponge-like material that can both soak up and break down one especially stubborn red dye, offering a simple way to clean polluted water using ordinary light.

Why Stubborn Dyes Are a Problem
Textile plants release a small but significant fraction of the dyes they use into wastewater. These dyes are designed to resist fading, which also makes them hard to remove once they reach the environment. Congo red, the dye examined here, has a complex structure that is toxic, potentially cancer-causing, and very resistant to natural breakdown. Common treatment methods—such as filtering on charcoal, adding chemicals to clump the dye, or relying on microbes—tend to move the dye from water into another waste stream rather than destroying it. That means extra treatment steps, higher cost, and a risk that the pollutant returns to the environment.
Turning Light Into a Cleaning Tool
In recent years, scientists have turned to “photocatalysts,” materials that use light to trigger reactions that tear apart dye molecules. Titanium dioxide is one of the best-known examples: when lit, it can generate highly reactive forms of oxygen that attack pollutants. However, tiny titanium particles are hard to separate from water after treatment, and they mostly absorb ultraviolet light, which is only a small slice of sunlight. To overcome these limits, the authors combined titanium with cerium, a rare-earth element that helps keep electric charges separated inside the material and improves its performance under visible light. They shaped this mixture into rod-like structures a few billionths of a meter wide, providing straight pathways for charges to travel and react instead of quickly cancelling each other out.
A Soft Net for Hard Work
Simply sprinkling these nanorods into water would again make them difficult to recover. Instead, the team locked them inside a soft, three-dimensional hydrogel made from a modified plant fiber (carboxymethyl cellulose) and a common water-absorbing plastic (polyacrylamide). This gel behaves like a wet sponge filled with tiny channels. Its chemical groups attract negatively charged dye molecules, pulling them out of the surrounding water and concentrating them near the nanorods. At the same time, the gel keeps the rods from clumping or washing away. Careful imaging and surface measurements showed that the rods remained well spread throughout the gel, and that the material had a large internal surface area where reactions could occur.
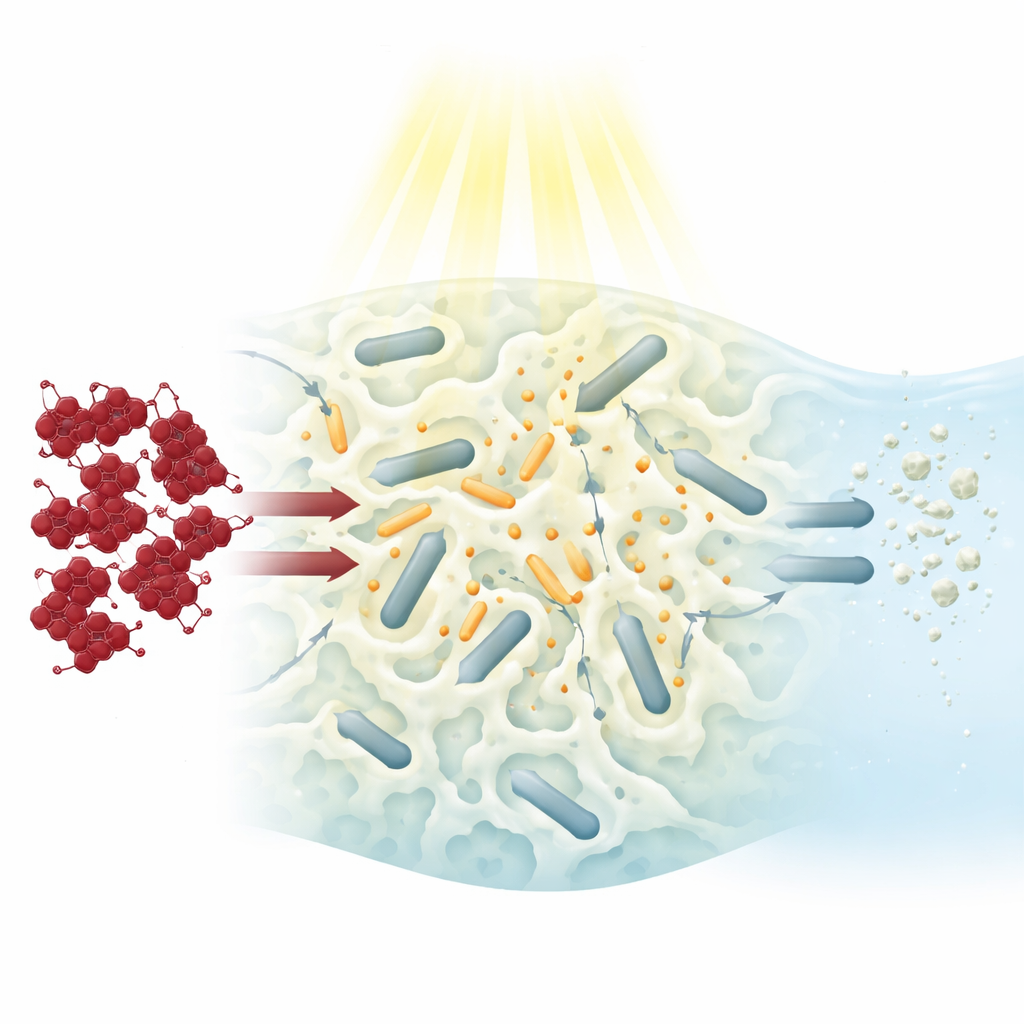
How Well the New Sponge Works
To test performance, the researchers placed pieces of the composite gel into Congo red solutions and illuminated them with visible light from a lamp or direct sunlight. Even without light, the gel alone could remove about 40% of the dye by simple attraction. When light was added, however, removal jumped to about 92% within just 90 minutes, with most of the improvement coming from true chemical breakdown rather than mere capture. The team varied many conditions—dye concentration, contact time, acidity, temperature, stirring speed, and gel amount—and found that the material maintained high efficiency over a fairly wide range. Calculations of how quickly the dye disappeared suggested that the process follows a simple first-order pattern, and that both movement through the surrounding water and diffusion inside the gel’s pores play roles in getting dye molecules to the active sites.
What This Means for Real-World Water
Compared with similar dye-cleaning systems reported in the literature, this new gel removes Congo red faster while still reaching around 92% degradation using visible light or sunlight. In plain terms, the material acts as a reusable, light-activated sponge: it pulls harmful dye molecules close, then uses light energy to chop them into smaller pieces that are far less troublesome. The authors note that longer-term tests of stability and possible metal leakage are still needed, but the combination of plant-based ingredients, strong performance, and operation under mild conditions suggests a promising route toward simpler and more sustainable treatment of colored wastewater.
Citation: Khalil, A.M., Kamel, S. & Mohy-Eldin, M.S. Photocatalytic degradation of Congo red dye using innovative cerium titanate nanorods embedded in a cellulose-based hydrogel. Sci Rep 16, 12476 (2026). https://doi.org/10.1038/s41598-026-43425-8
Keywords: wastewater treatment, dye pollution, photocatalytic hydrogel, nanomaterials, Congo red