Clear Sky Science · en
HIF3A-mediated aberrant activation of TXNIP promotes Alzheimer’s disease progression
Why Brain Support Cells Matter in Memory Loss
Alzheimer’s disease is usually described as a problem of dying nerve cells, but this study shines a spotlight on their lesser-known helpers: astrocytes, the star-shaped support cells of the brain. By zooming in on tens of thousands of individual cells, the researchers uncovered a damaging stress pathway inside astrocytes that appears to drive energy failure and cell damage in Alzheimer’s. Understanding this hidden machinery could open new paths to treatments that slow or soften the disease by protecting the brain’s internal chemistry.
Stress Inside the Aging Brain
Our brains constantly use oxygen to fuel thinking, but this process also produces reactive by-products often called “oxidative stress.” In healthy tissue, these chemical sparks are kept under tight control. In Alzheimer’s, however, they build up and damage cell membranes, proteins, and DNA. The team analyzed large genetic datasets from donated human brain tissue and used single-cell sequencing to score how strongly each cell type was experiencing oxidative stress. They found that overall stress levels were clearly higher in brains with Alzheimer’s compared to healthy brains of similar age.
The Surprising Role of Astrocytes
Not all brain cells were affected equally. When the scientists grouped more than 37,000 individual cells into major cell types—such as neurons, microglia, and astrocytes—they discovered that astrocytes carried the heaviest oxidative burden. These cells, which normally nourish neurons, help maintain brain chemistry, and manage blood flow, showed the highest stress scores and were actually less abundant in Alzheimer’s tissue. This suggests that the very cells meant to shield neurons from harm are themselves overwhelmed, potentially leaving nerve cells more vulnerable to damage and death.
A Hidden Switch: The HIF3A–TXNIP Pathway
To understand what was pushing astrocytes into this stressed state, the researchers sifted through more than a thousand stress-related genes and used several machine-learning approaches to narrow them down. One gene, called HIF3A, stood out. It was strongly linked to stress pathways and was especially active in astrocytes. Further experiments in mouse brains and cultured human astrocytes showed that HIF3A acts like a switch in the cell nucleus, turning on another gene called TXNIP. TXNIP interferes with one of the cell’s key antioxidant systems, allowing harmful reactive molecules to pile up, especially in the energy-producing compartments known as mitochondria.
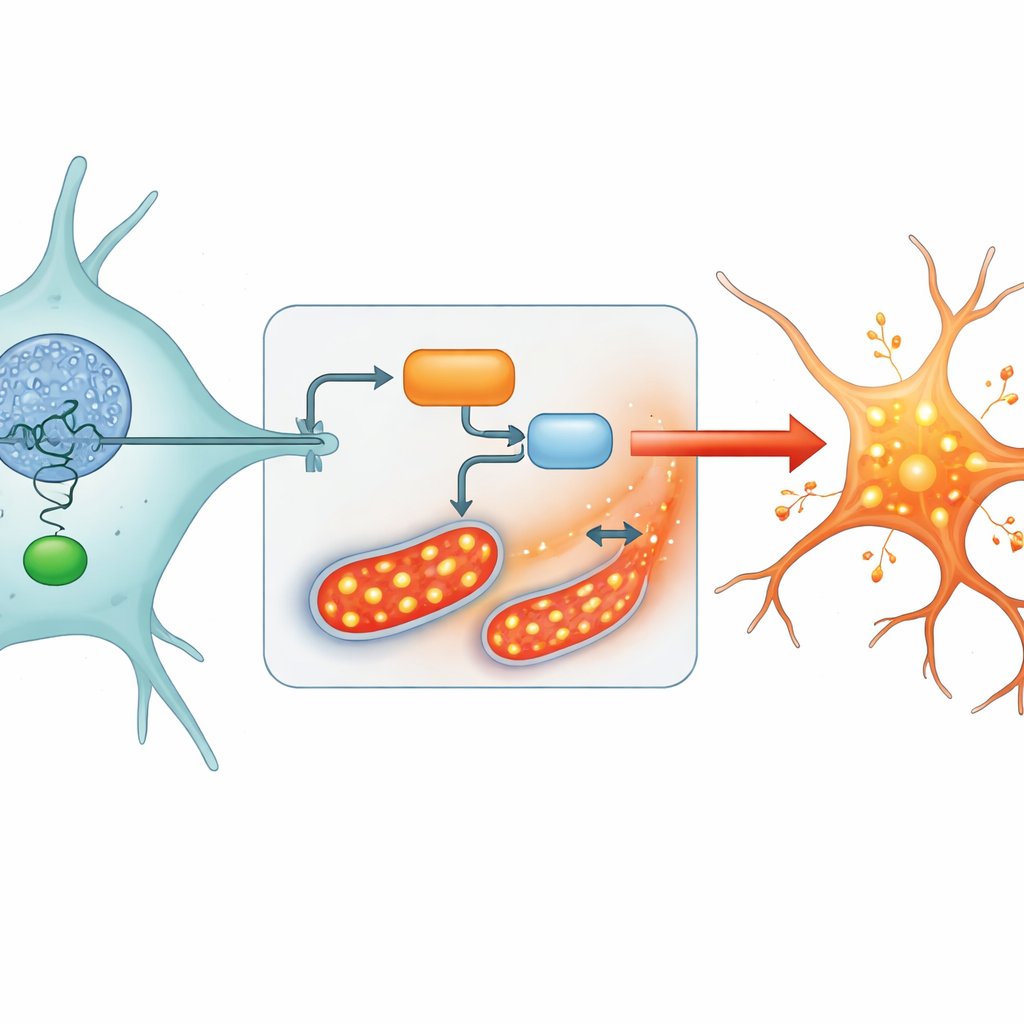
From Energy Failure to Worsening Disease
When TXNIP was boosted by HIF3A, astrocyte mitochondria produced more reactive molecules and less energy. Measures of cellular antioxidants dropped, while mitochondrial stress and oxidative damage climbed. In an Alzheimer’s-like mouse model, both HIF3A and TXNIP were elevated in the brain, and animals showed memory problems in maze tests. Knocking down HIF3A in astrocytes reversed many of these changes: oxidative stress decreased, mitochondrial activity and energy production improved, and the cells’ internal balance shifted back toward a healthier state. These findings support a chain of events in which overactive HIF3A drives TXNIP, which in turn disrupts energy and redox balance, helping to push Alzheimer’s disease forward.
What This Means for Future Treatments
For non-specialists, the message is that Alzheimer’s is not just a story of bad plaques and tangles in neurons. It is also a story of exhausted support cells and runaway chemical stress. This study identifies a specific control pathway inside astrocytes—the HIF3A–TXNIP axis—that links gene activity to oxidative damage and energy failure. Targeting this switch, for example by dampening HIF3A or blocking TXNIP, could provide a new therapeutic angle aimed at restoring the brain’s natural defense systems and slowing memory decline.
Citation: Xue, M., Zheng, W., Li, F. et al. HIF3A-mediated aberrant activation of TXNIP promotes Alzheimer’s disease progression. Sci Rep 16, 12912 (2026). https://doi.org/10.1038/s41598-026-43404-z
Keywords: Alzheimer’s disease, oxidative stress, astrocytes, HIF3A, TXNIP