Clear Sky Science · en
Sentinel lymph node detection in gastric cancer using a dual tracer (Superparamagnetic iron oxide and methylene blue): a prospective study with histological and OSNA validation
Why this research matters
Stomach cancer is common and deadly, largely because it often spreads through tiny filters in the body called lymph nodes. Surgeons usually remove many of these nodes to be safe, but that can mean longer operations and more complications. This study explores a gentler, more targeted way to find the few lymph nodes that matter most, using a magnetic dye and a blue stain together, plus a rapid genetic test, to check whether cancer has truly spread.
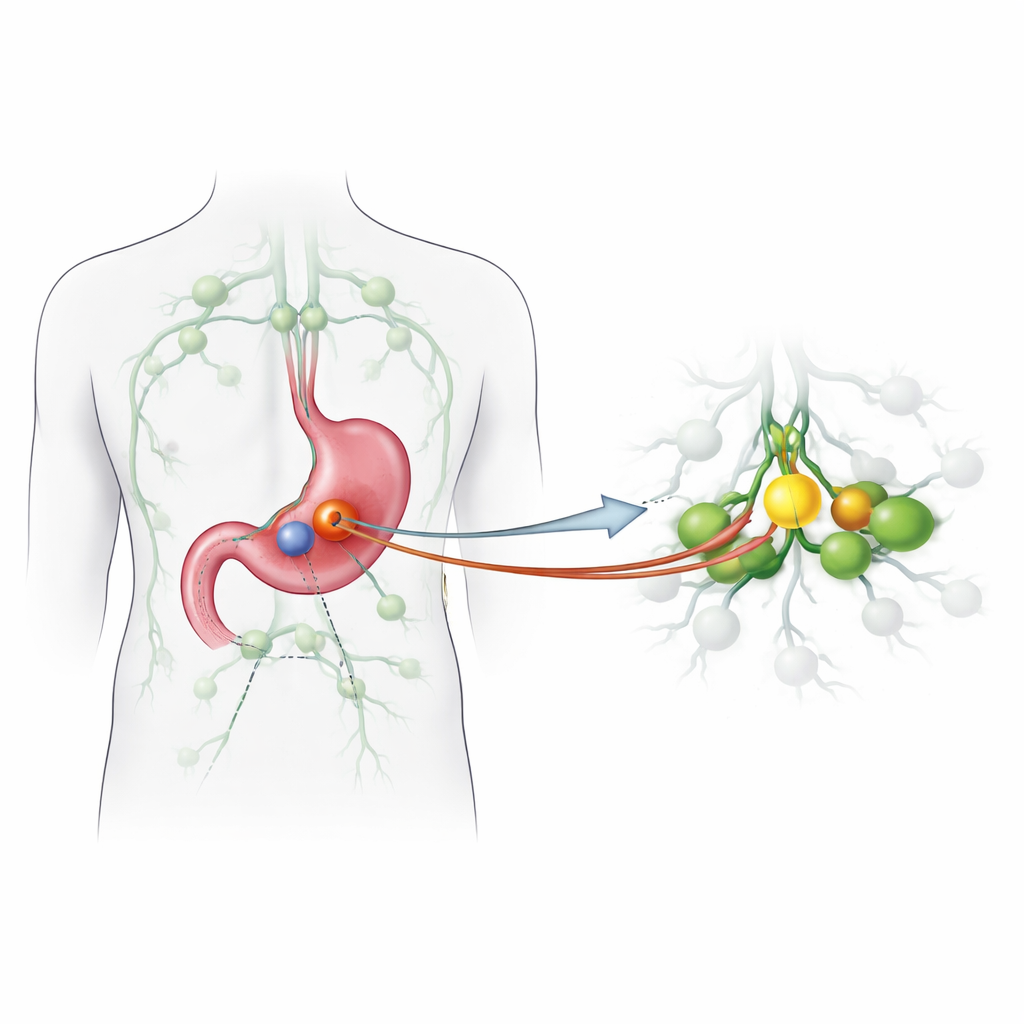
Finding the first stop on cancer’s journey
When cancer cells leave a tumor, they usually travel first to a “sentinel” lymph node, the body’s earliest warning station. If this first node is clean, the remaining nodes are often cancer-free; if it is involved, more extensive spread is likely. This idea has transformed surgery for breast cancer and melanoma, but has been harder to apply in stomach cancer because drainage pathways are complex and sometimes unpredictable. The team behind this study wanted to test whether they could reliably spot the true sentinel nodes in stomach cancer using a new pairing of tracers: superparamagnetic iron oxide particles, detected with a handheld magnetic probe, and a classic blue dye that colors the draining nodes.
A careful test outside the operating room
The researchers enrolled 38 adults with stomach adenocarcinoma who were already scheduled for standard cancer surgery with removal of the stomach (total or partial) and a wide lymph node dissection. During surgery, they injected the magnetic tracer around the tumor, then, once the specimen was removed, injected blue dye into the opened stomach. Any node that either turned blue or gave a magnetic signal was labeled as a candidate sentinel node and taken for detailed examination. To judge how well this method worked, they used a strict definition of “true” sentinel nodes based on whether these nodes correctly reflected the cancer status of the rest of the lymph nodes.
Pairing classic microscopes with rapid molecular testing
Each sentinel node was cut in half. One half went to the traditional microscope route, where pathologists looked for cancer cells on stained slides. The other half went to a rapid molecular assay called one-step nucleic acid amplification (OSNA), which measures the amount of a specific messenger molecule made by stomach cancer cells. For the remaining, non-sentinel nodes, the team tried a pooling strategy: small pieces from many nodes in the same region were blended and tested together by OSNA, while separate slices were checked under the microscope. This approach aims to screen a large number of nodes quickly while still being able to detect even tiny deposits of cancer.
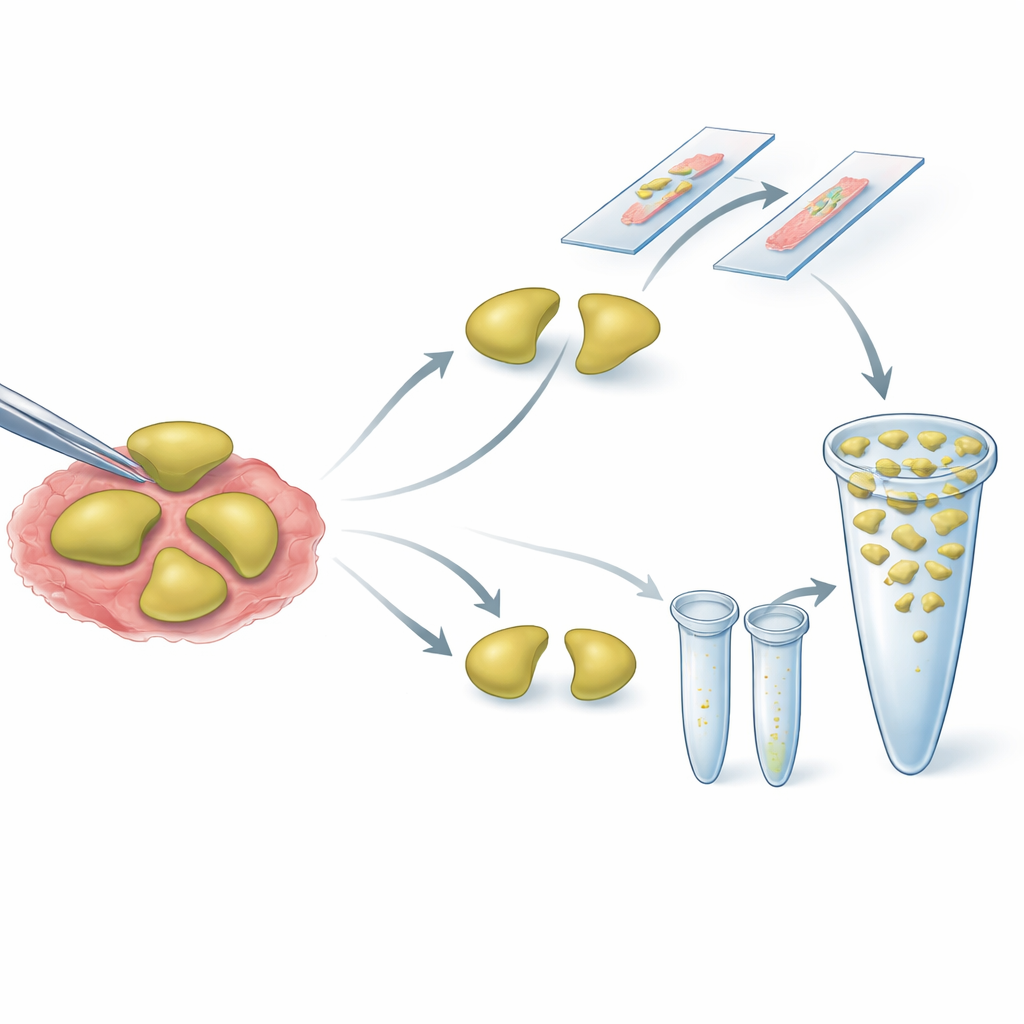
What the study found
The dual tracer method was able to identify sentinel nodes in just over 84% of patients, with an average of about two nodes per person. In most cases, these nodes correctly represented whether the rest of the lymph nodes were involved, giving the technique high sensitivity but only moderate specificity under the strict criteria used. The magnetic and blue tracers overlapped in many, but not all, nodes, suggesting that using both together adds reliability. The OSNA test showed good agreement with standard pathology overall, and it was particularly helpful in picking up very small clusters of cancer cells that might be missed on a few thin microscope slices. Pooling of non-sentinel nodes allowed more than a thousand nodes to be evaluated efficiently, with generally good concordance between pooled molecular results and conventional histology across many nodal regions.
Early signs for patient outcomes
Patients were followed for a median of more than four years. As expected, those whose lymph nodes were negative—whether judged by standard slides or by the molecular test—tended to live longer than those with nodal spread. However, when the researchers accounted for the overall tumor stage, the molecular result alone did not independently predict survival, suggesting it largely mirrors the information already captured by existing staging systems. Because the study was relatively small and performed on tissue outside the body rather than during live decision-making in the operating room, these outcome findings are considered exploratory clues rather than firm proof.
What this means for future care
For now, this work is a technical proof-of-concept rather than a new standard of care. It shows that combining a magnetic tracer with blue dye to map sentinel nodes in stomach cancer is feasible and reasonably accurate in an experimental setting, and that pairing traditional microscopy with rapid molecular testing can give a richer, more sensitive picture of whether cancer has reached the nodes. If future, larger in-body trials confirm these results, surgeons may one day be able to tailor stomach cancer operations more precisely—removing fewer lymph nodes and preserving more normal tissue in carefully selected patients, without sacrificing the accuracy of cancer staging.
Citation: Escalera-Pérez, R., Medina-Achirica, C., García-Molina, F. et al. Sentinel lymph node detection in gastric cancer using a dual tracer (Superparamagnetic iron oxide and methylene blue): a prospective study with histological and OSNA validation. Sci Rep 16, 14202 (2026). https://doi.org/10.1038/s41598-026-43345-7
Keywords: gastric cancer, sentinel lymph node, dual tracer, OSNA, lymph node staging