Clear Sky Science · en
Tailoring electronic structure and magnetic anisotropy in spray-pyrolyzed NiFe2O4 thin films for spintronic applications
Why tiny magnetic films matter
From faster memory chips to ultra-sensitive sensors, tomorrow’s electronics will increasingly rely not just on the charge of electrons, but also on their spin—a field known as spintronics. For these devices to work reliably, engineers need magnetic materials that can be grown as ultra-thin films with carefully tuned properties. This study explores how a relatively simple manufacturing method, spray pyrolysis, can be used to fine‑tune the internal structure and magnetism of nickel ferrite (NiFe2O4) thin films, making them promising building blocks for future spin‑based technologies.
Making magnetic films with a spray
The researchers fabricated NiFe2O4 films by spraying a solution of nickel and iron salts onto heated glass substrates. As the fine droplets hit the hot surface, they decomposed and crystallized into a solid, nanometer‑grained layer. By varying the substrate temperature between 300 °C and 400 °C, the team could systematically change how the atoms arranged themselves and how the grains in the film grew. X‑ray diffraction measurements confirmed that all films formed the desired “spinel” crystal structure, but the degree of order and strain in that structure depended strongly on the growth temperature.
How growth temperature reshapes the film
High‑resolution electron microscopy revealed that films grown at the lowest temperature (300 °C) were thicker, smoother, and more uniform, with well‑developed grains and a very even distribution of nickel, iron, and oxygen. As the growth temperature increased, the films became thinner and rougher, with more agglomerated particles, islands, and subtle chemical inhomogeneities. Detailed analysis of the X‑ray diffraction peak shapes showed that higher temperatures led to smaller crystallites and more internal strain. These microstructural changes, caused by faster evaporation, re‑evaporation of material, and enhanced defect formation, set the stage for how the films behave magnetically.
Magnetism shaped by structure
Magnetization measurements at room temperature showed that all samples were soft ferrimagnets: they could be magnetized easily and had very low coercivity, a desirable trait for many device applications. However, the strength and character of the magnetism changed with growth conditions. The coolest, best‑ordered film had the highest saturation magnetization and the lowest coercive field, meaning its internal magnetic regions aligned readily and efficiently. As the growth temperature rose, magnetization steadily decreased and coercivity increased, signaling more defects, smaller grains, and more obstacles to the motion and rotation of magnetic domains. Advanced modeling of the magnetization curves suggested that, in addition to ordinary ferrimagnetism, localized regions where charge carriers couple to magnetic ions—so‑called bound magnetic polarons—also contribute to the overall behavior.
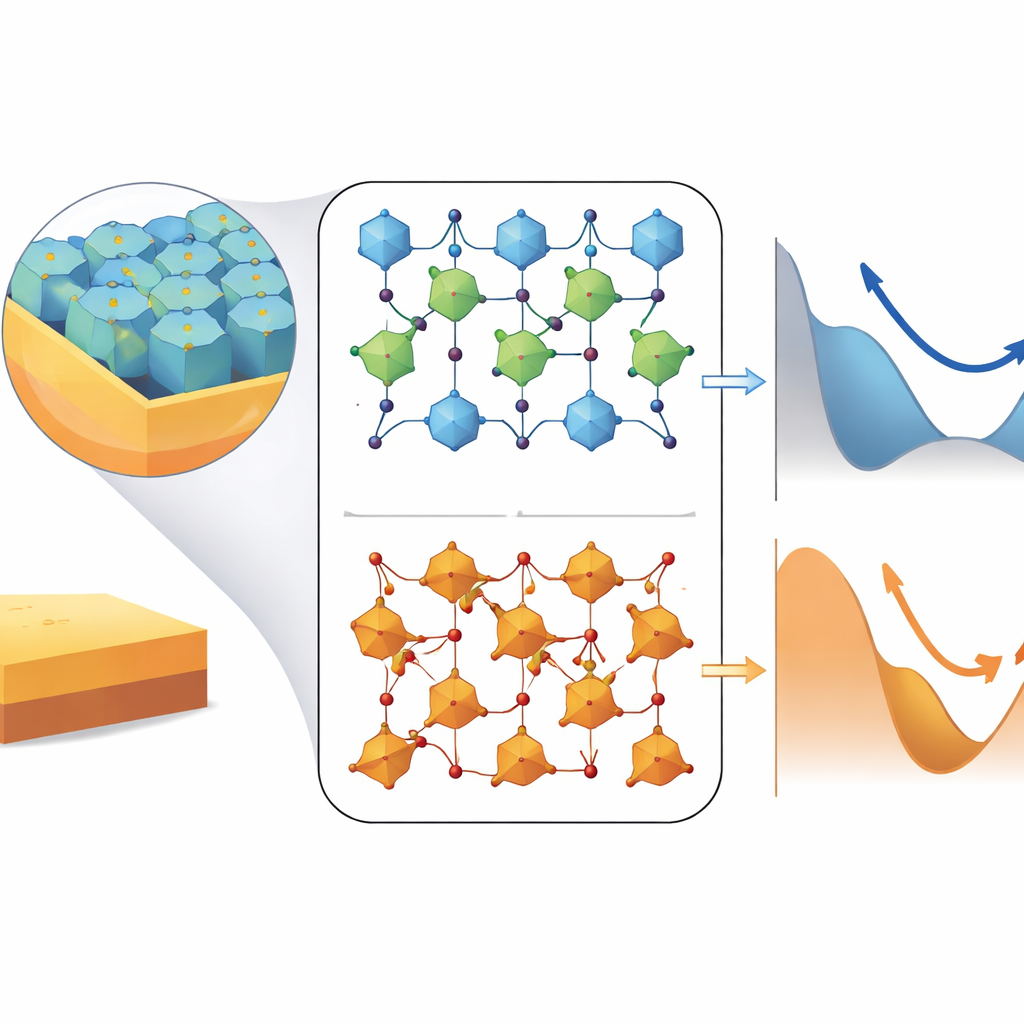
Peeking at atoms with X‑rays
To link these magnetic trends to the behavior of specific atoms, the team used X‑ray absorption spectroscopy and X‑ray magnetic circular dichroism. These techniques act like element‑sensitive magnetic fingerprints, revealing which atomic sites nickel and iron occupy, what charge states they have, and how their spins and orbital motions contribute to the total magnetism. The measurements confirmed that nickel mainly appears as Ni²⁺ on octahedral sites, while iron appears as Fe³⁺ on both octahedral and tetrahedral sites—exactly the pattern expected for an “inverse” spinel. Crucially, the data showed that in the best‑ordered film, both nickel and iron carry unusually large orbital magnetic moments, a sign that local structural distortions and strong mixing between metal and oxygen orbitals partially “unfreeze” orbital motion that is usually locked in place. This unfreezing strengthens spin‑orbit coupling and boosts the directional preference, or anisotropy, of the magnetization.
What this means for future devices
For spintronic technology, the combination of soft ferrimagnetism, significant magnetic anisotropy, and controllable electronic structure is especially attractive. This work shows that simply tuning the substrate temperature during spray pyrolysis can steer the grain size, strain, and defect landscape in NiFe2O4 films, which in turn controls their magnetic strength and anisotropy through subtle changes at the atomic level. In plain terms, getting the heating conditions “just right” yields smoother, better‑ordered films in which nickel and iron spins, along with their orbital motion, cooperate to produce robust, tunable magnetism at room temperature—exactly what is needed for reliable, low‑power spintronic components.
Citation: Patra, J., Parida, P., Patel, P. et al. Tailoring electronic structure and magnetic anisotropy in spray-pyrolyzed NiFe2O4 thin films for spintronic applications. Sci Rep 16, 13485 (2026). https://doi.org/10.1038/s41598-026-43296-z
Keywords: nickel ferrite, thin films, magnetic anisotropy, spray pyrolysis, spintronics