Clear Sky Science · en
Synthesis, characterization, and biological activities of two new pyrazole Schiff base derivatives
Why this matters for everyday health
Doctors are increasingly facing two linked problems: infections that no longer respond to usual antibiotics and cancers that are hard to treat. Many cancer patients also develop serious hospital-acquired infections while their immune systems are weakened. This study explores a new type of laboratory-made molecule that aims to tackle both challenges at once—fighting dangerous microbes and attacking cancer cells—while also checking how safe it might be for human use.
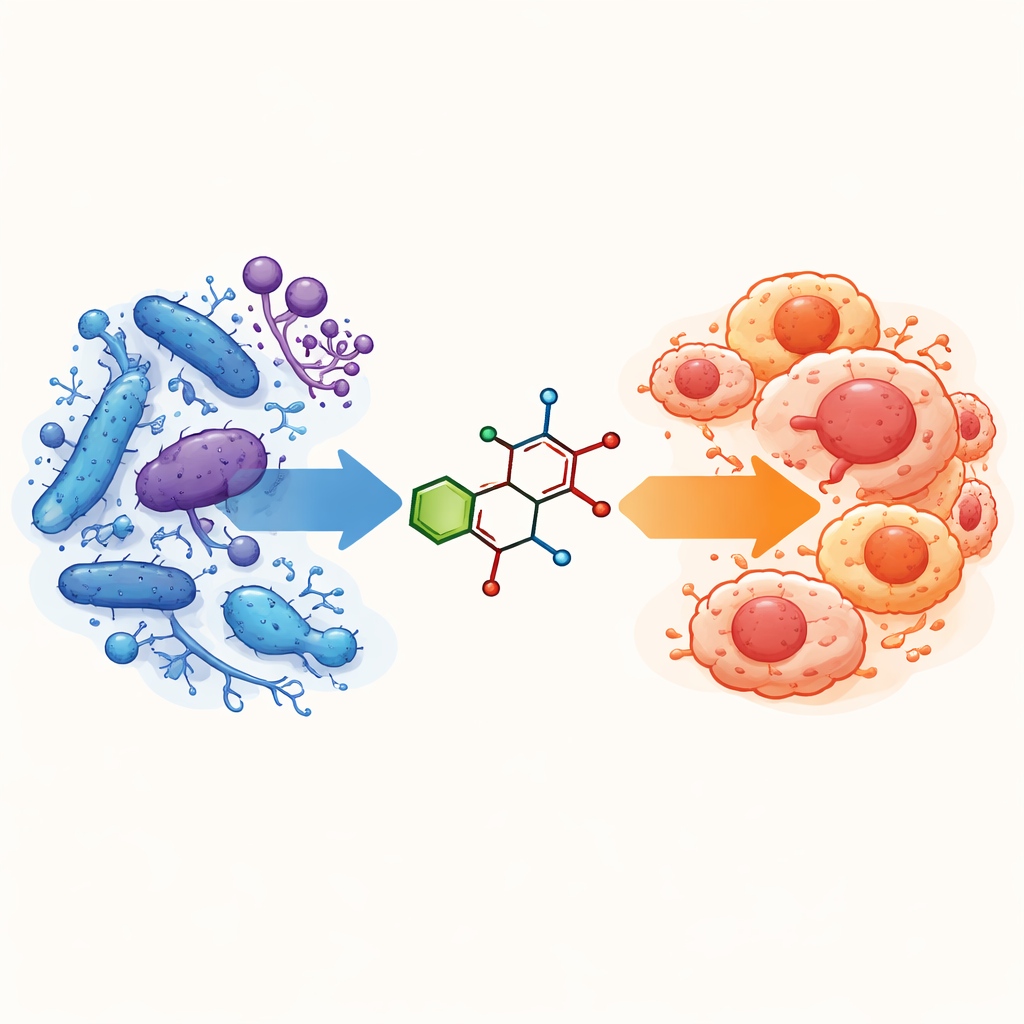
Building a new kind of small molecule
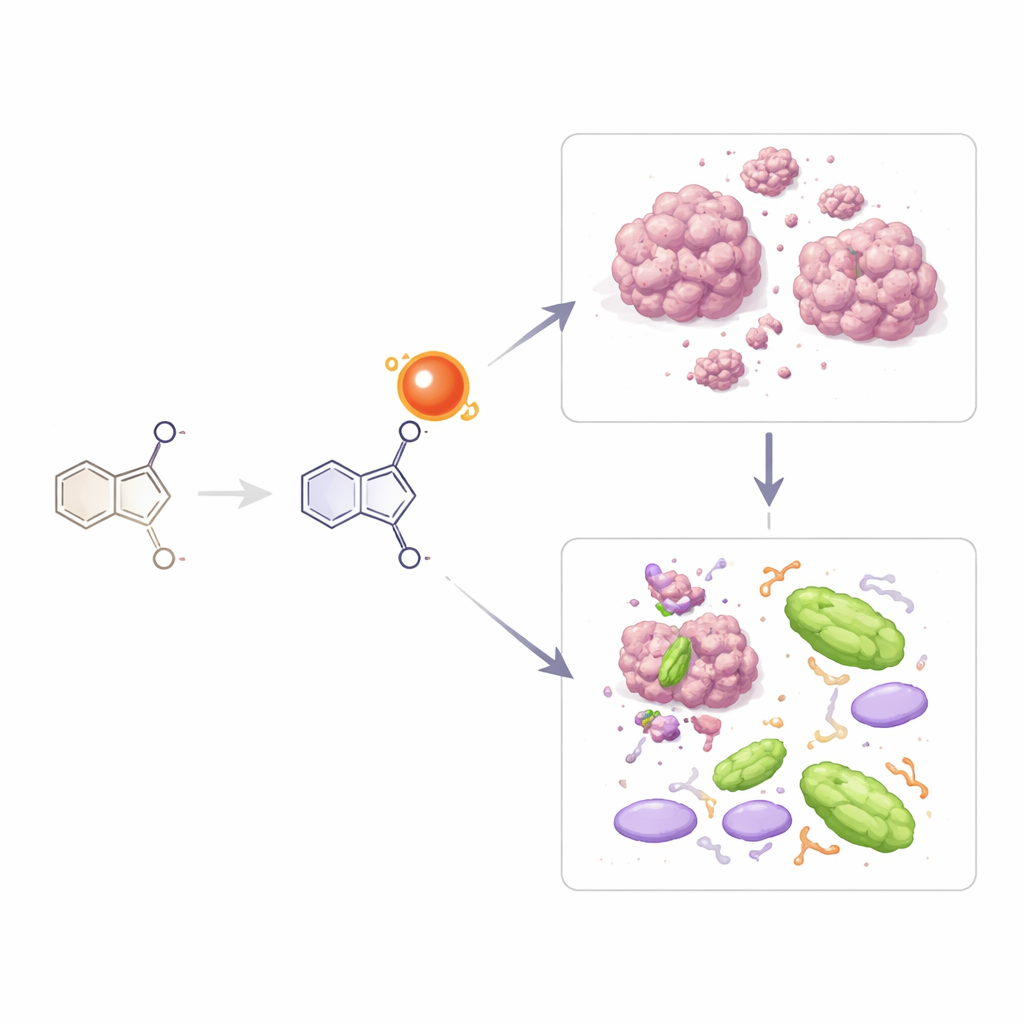
The researchers designed two closely related small molecules, called 3a and 3b, by joining together three common building blocks used in medicinal chemistry. One part comes from a ring structure known to appear in many modern drugs, another from a sulfur-containing ring, and the third from a group called a Schiff base that often binds metals and interacts strongly with proteins. The only difference between 3a and 3b is that 3b carries a bromine atom in a specific position on one ring. The team created these molecules in a simple one-step reaction, purified them, and then confirmed their structures using several standard tools: infrared light absorption, nuclear magnetic resonance, ultraviolet–visible light spectra, mass spectrometry, and elemental analysis. These tests showed that the planned structures had indeed formed and were stable in common laboratory solvents.
Testing against germs that threaten patients
To see whether the new molecules could fight infection, the team tested them against a panel of harmful microbes, including common hospital bacteria and several species of the yeast Candida, which often causes serious infections in people with weakened immune systems. A first, simpler test on solid agar plates showed little effect. However, a more sensitive liquid test, which measures the lowest dose needed to stop growth, told a different story. Molecule 3b, the bromine‑containing version, strongly inhibited Staphylococcus aureus and showed promising activity against disease‑causing strains of Escherichia coli, Pseudomonas aeruginosa, and Klebsiella pneumoniae. It was also clearly effective against three Candida species at relatively low concentrations, whereas 3a worked only modestly against these yeasts and was inactive against most bacteria. These results suggest that the added bromine atom in 3b makes the molecule better able to get into or disrupt microbial cells.
Going after cancer cells while sparing healthy tissue
The scientists next examined whether 3a and 3b could kill cancer cells grown in dishes. They tested four human cancer cell lines—two breast cancers, a prostate cancer, and a lung cancer—along with normal skin cells to gauge safety. Molecule 3a showed weak effects and never reached the level at which half the cells died, even at its highest workable dose. In contrast, 3b displayed much stronger activity, particularly against breast cancer cells of the MCF7 line, and also against prostate and another breast cancer line. By comparing the dose needed to harm cancer cells with the dose that affected normal fibroblasts, the team concluded that 3b has at least some selectivity, especially for one breast cancer type. They also noted that 3b is far more soluble than 3a, which likely helps it enter cells and exert its action.
Checking genetic safety and blood compatibility
Promising activity is not enough; any future medicine must also be safe. The researchers therefore ran two additional tests. First, they used a standard bacterial assay, known as the Ames test, to see whether 3a or 3b could damage DNA and cause mutations. Across several doses, and with or without an added system that mimics liver metabolism, neither molecule increased mutation rates compared with controls, indicating that both are non‑mutagenic under the tested conditions. Second, they mixed the compounds with human red blood cells to see whether the cells would burst and release their contents, a sign of potential damage to blood in the body. Here, both molecules caused high levels of cell rupture at all tested concentrations, with 3b slightly more damaging at the lowest dose. This strong tendency to lyse red blood cells flags an important safety concern that must be solved before any clinical use.
What the findings mean going forward
Overall, this study shows that a small change in structure—adding a bromine atom—can turn a weakly active molecule (3a) into a much more powerful compound (3b) that can kill both microbes and cancer cells in the lab without directly damaging DNA. At the same time, the strong destructive effect on red blood cells warns that 3b, in its current form, could be harmful if given freely in the bloodstream. The authors suggest that future work could modify 3b further or package it in delivery systems, such as nanoparticles or metal complexes, that protect blood cells while directing the drug toward tumors and infections. In that sense, 3b serves as a promising starting scaffold—a proof of concept that one carefully tuned molecule might one day help treat both cancer and drug‑resistant infections together.
Citation: Matar, S., Abu-Yamin, AA., Taher, D. et al. Synthesis, characterization, and biological activities of two new pyrazole Schiff base derivatives. Sci Rep 16, 14195 (2026). https://doi.org/10.1038/s41598-026-43254-9
Keywords: pyrazole Schiff base, dual antimicrobial anticancer, halogenated small molecules, drug resistant infections, candidate cancer therapeutics