Clear Sky Science · en
Green synthesis of Mohr’s salt–modified keratin composite for selective removal of arsenate from polluted water
Turning Poultry Waste into Clean Water
In many parts of the world, especially across South Asia and Latin America, wells that look safe can in fact be laced with invisible arsenic. Long-term exposure to this toxic element can cause serious health problems, yet removing it from water in a cheap, reliable, and environmentally friendly way has been a major challenge. This study explores an inventive solution: transforming discarded chicken feathers into a high-performing filter material that can pull arsenic out of polluted groundwater without adding new contaminants in the process.
Why Arsenic in Groundwater Matters
Arsenic naturally leaks from certain rocks and is also released by human activities such as mining and past pesticide use. Once in oxygen-rich groundwater, it often appears as arsenate, a negatively charged form that moves easily with water and is hard to trap. In some regions of India, including districts in Punjab, arsenic levels have been measured hundreds of times higher than drinking-water limits. Conventional treatment methods can be costly, energy-intensive, or generate their own waste streams, prompting a search for low-cost materials that can be produced locally and used safely in rural settings.
Feathers as a Hidden Resource
Chicken feathers are produced in huge quantities as slaughterhouse waste and are rich in a tough structural protein called keratin. Keratin contains many nitrogen and sulfur groups that can grab onto metal ions, making it a promising base for water-cleaning materials. However, untreated keratin on its own does not bind arsenate strongly or selectively enough to be practical. The researchers therefore set out to redesign feather-derived keratin so that it could host iron species known to latch firmly onto arsenic, while still keeping the process simple, gentle, and in line with green-chemistry principles.
Building a Smart Iron–Keratin Sponge
The team extracted keratin from cleaned and processed chicken feathers, then combined it with a common laboratory compound called Mohr’s salt, which supplies iron in a particularly stable form. By carefully controlling the acidity and stirring the mixture for several hours, they encouraged iron to spread evenly throughout the keratin and form tiny iron oxide and oxyhydroxide domains inside and on the surface of the protein. Microscopy and spectroscopy showed that this treatment transformed the originally dense, smooth keratin into a rough, porous network studded with well-dispersed iron-rich spots, while preserving the feather protein’s sulfur- and nitrogen-containing groups that help anchor the metal.
How the New Material Traps Arsenic
When the modified material, called MSMK, was shaken with arsenic-contaminated water under realistic conditions, it removed up to 98.5 percent of dissolved arsenate at near-room temperature and a mildly acidic to neutral pH around 6. At this pH, the surface of MSMK carries a positive charge, which attracts the negatively charged arsenate species. Once near the surface, arsenate does more than just stick loosely: it forms strong, inner-layer bonds with iron–oxygen groups within narrow pores about two nanometers wide. Tests of how quickly and how much arsenate is taken up over time and concentration suggest that movement through these tiny channels is the main step that limits the overall rate, while a patchwork of binding sites with slightly different strengths allows efficient capture even at low arsenic levels.
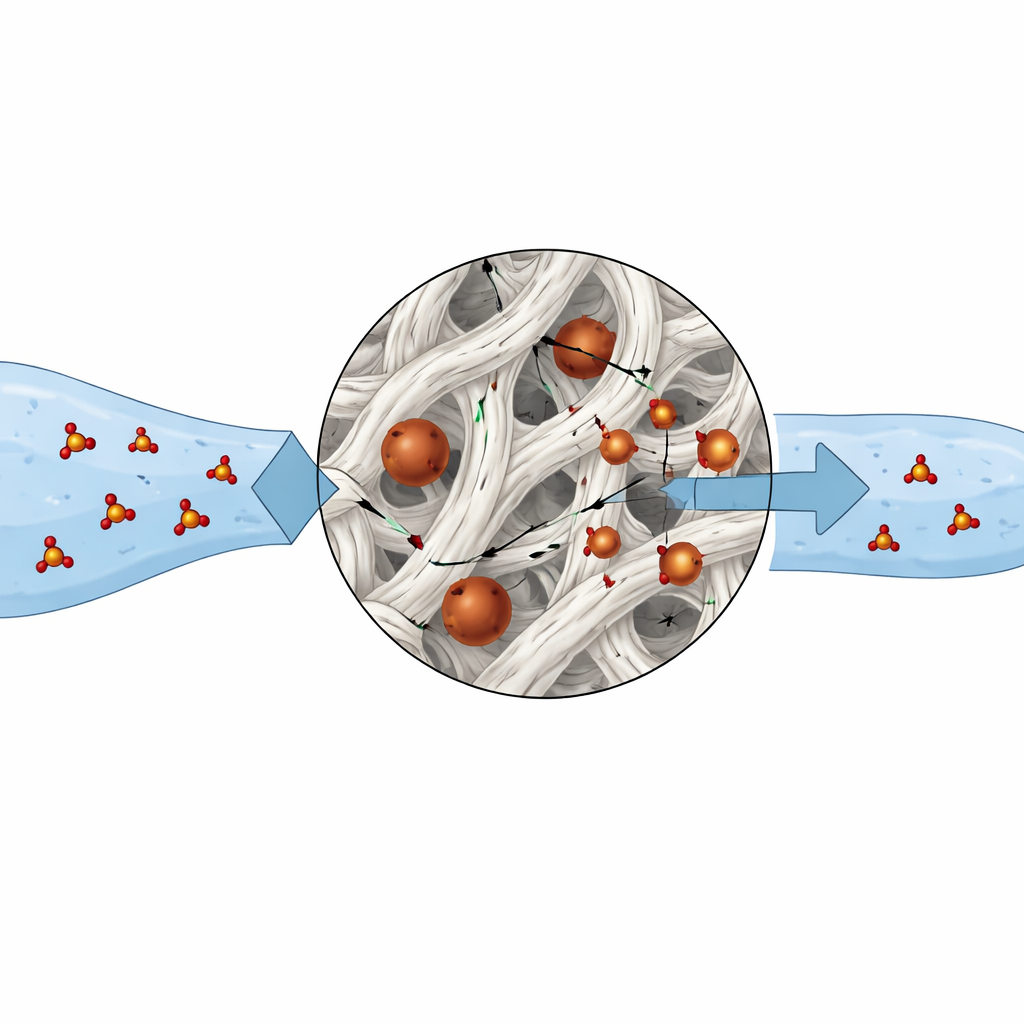
Performance, Safety, and Sustainability
The researchers also examined how durable and selective MSMK is. It maintained high arsenic removal over a range of temperatures close to natural groundwater conditions and still performed well in water containing many other common ions that often interfere with treatment. Importantly, iron did not leach from the material at detectable levels across a broad pH range, meaning the filter does not introduce new metal contamination. The composite could be regenerated several times using a mild alkaline wash, retaining more than 60 percent of its original efficiency after five cycles—better than many comparable bio-based filters. A simple green-chemistry assessment indicated that the synthesis uses benign ingredients, modest energy, and produces relatively little solid waste, while turning a problematic agricultural byproduct into a useful resource.
What This Means for Safer Drinking Water
In straightforward terms, this work shows that chicken feathers, when carefully treated with a stable iron salt, can become an effective and environmentally friendly “sponge” for arsenic in groundwater. The Mohr’s salt–modified keratin composite pulls arsenate out of water with high efficiency, resists shedding its own iron, and can be reused multiple times. Because the ingredients are inexpensive and the preparation avoids harsh conditions, the material has realistic potential for use in community-level filters or treatment units in regions hardest hit by arsenic contamination. With further testing in continuous-flow systems and real wells, this feather-based technology could help deliver safer drinking water while reducing agricultural waste.
Citation: Manju, Sharma, S. Green synthesis of Mohr’s salt–modified keratin composite for selective removal of arsenate from polluted water. Sci Rep 16, 14552 (2026). https://doi.org/10.1038/s41598-026-43190-8
Keywords: arsenic removal, groundwater purification, keratin adsorbent, iron-based filter, water treatment