Clear Sky Science · en
Microstructure-guided design of biopolymer-supported tri-phasic TiO2 for sustainable lead and cadmium detoxification
Cleaner Water for a Crowded Planet
Access to safe drinking water is becoming harder as cities grow and industries expand, releasing toxic metals like lead and cadmium into rivers and groundwater. These metals do not break down and can build up in our bodies, damaging the brain, kidneys, and other organs. This study explores a new, eco-friendly material that combines a natural biopolymer with specially engineered titanium dioxide nanoparticles to pull these metals out of water quickly and efficiently, using only mild conditions and even simple sunlight.
A Natural Helper Meets Smart Nanotechnology
The heart of this work is a partnership between chitosan, a substance derived from shells of crustaceans and other organisms, and titanium dioxide, a common white mineral found in paints and sunscreens. Chitosan is attractive for water treatment because it is abundant, biodegradable, and naturally rich in chemical groups that can latch onto metal ions. Titanium dioxide, meanwhile, is robust, inexpensive, and well known for its ability to absorb light and drive chemical reactions. By immobilizing tiny titanium dioxide particles inside a chitosan matrix, the researchers set out to build a small but powerful sponge that can selectively grab heavy metals from contaminated water. 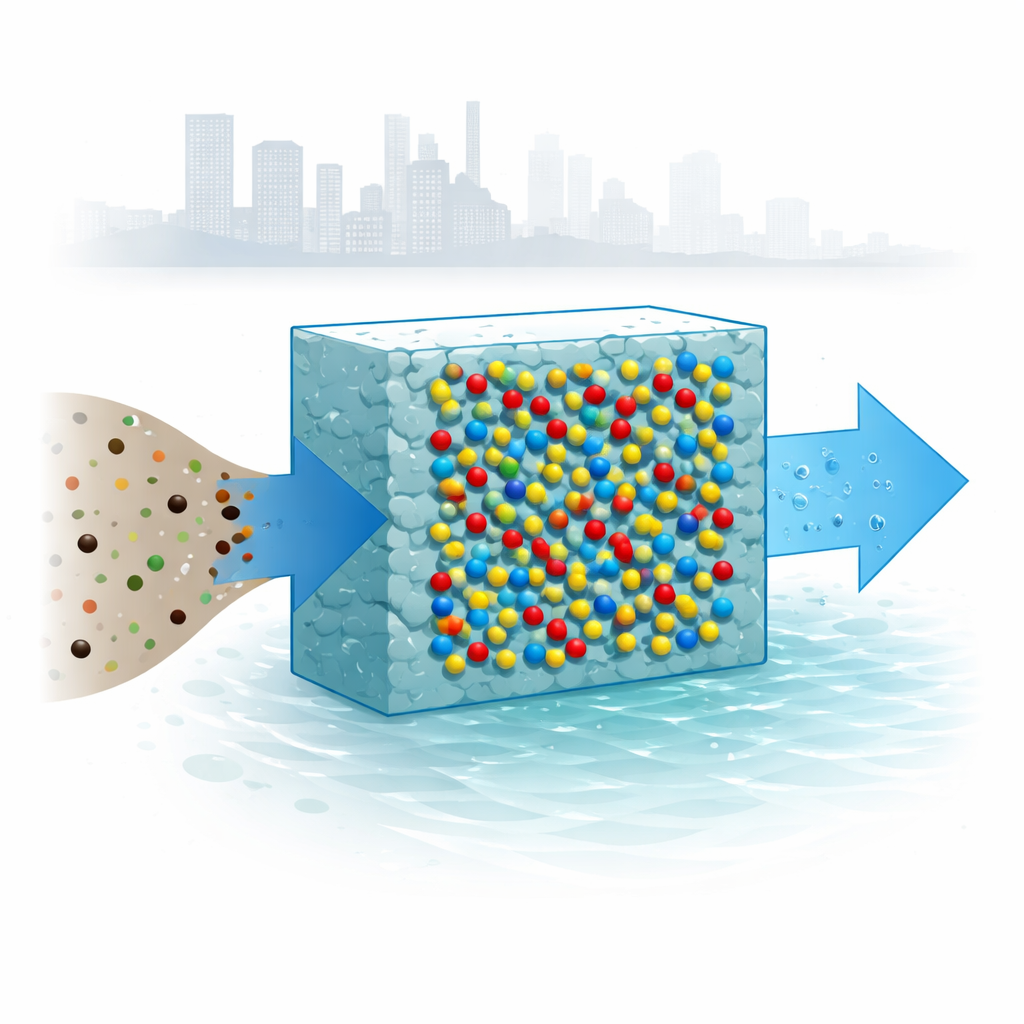
Three Faces of One Mineral Working Together
Titanium dioxide can crystallize in several forms, much like carbon can appear as graphite or diamond. Instead of using only one of these forms, the team deliberately created a blend of three—anatase, rutile, and brookite—within each nanoparticle. Advanced tools, including X-ray diffraction and electron microscopy, confirmed that all three phases coexisted in well-defined proportions and were tightly anchored within the chitosan network. This “tri-phasic” design creates many internal boundaries inside each particle, which helps separate electrical charges and raises the number of active sites on the surface. As a result, the composite can interact more strongly with lead and cadmium ions and can make better use of sunlight, since its light-absorbing properties are shifted to work under natural illumination rather than only under strong ultraviolet lamps.
How the Material Grabs and Holds Toxic Metals
To test performance, the researchers placed the chitosan–titanium dioxide composite into solutions containing known amounts of lead or cadmium. They then varied key conditions such as acidity (pH), contact time, and starting metal concentration. At mildly neutral pH—close to that of many natural waters—the composite removed almost all of the metals: about 99.9% of lead and 97.9% of cadmium. The material reached its full capacity in roughly 90 minutes for lead and 120 minutes for cadmium, much faster than many conventional sorbents. At the microscopic level, negatively charged and electron-rich groups on chitosan, together with reactive spots on the titanium dioxide surfaces, attract the positively charged metal ions. First, the ions rapidly attach to the outer surface; then they slowly move into the interior pores, forming multiple layers held by a mix of physical and chemical forces. Mathematical models of the data support this two-step, multi-layer binding process and show that the surface of the composite is highly heterogeneous—full of sites that can bind metals with different strengths.
From Lab Tests to Real-World Promise
When the team compared their composite with other metal-removing materials reported in the literature, they found that while some alternatives can hold slightly more metal per gram, they often require longer treatment times, harsher chemical conditions, or are made from less sustainable ingredients. In contrast, the chitosan–titanium dioxide system operates efficiently at room temperature, at near-neutral pH, and under sunlight, while being built from low-cost, widely available components. This combination of high removal efficiency, speed, and environmental friendliness makes it especially promising for decentralized or low-resource water treatment, where complex equipment and continuous power may not be available. 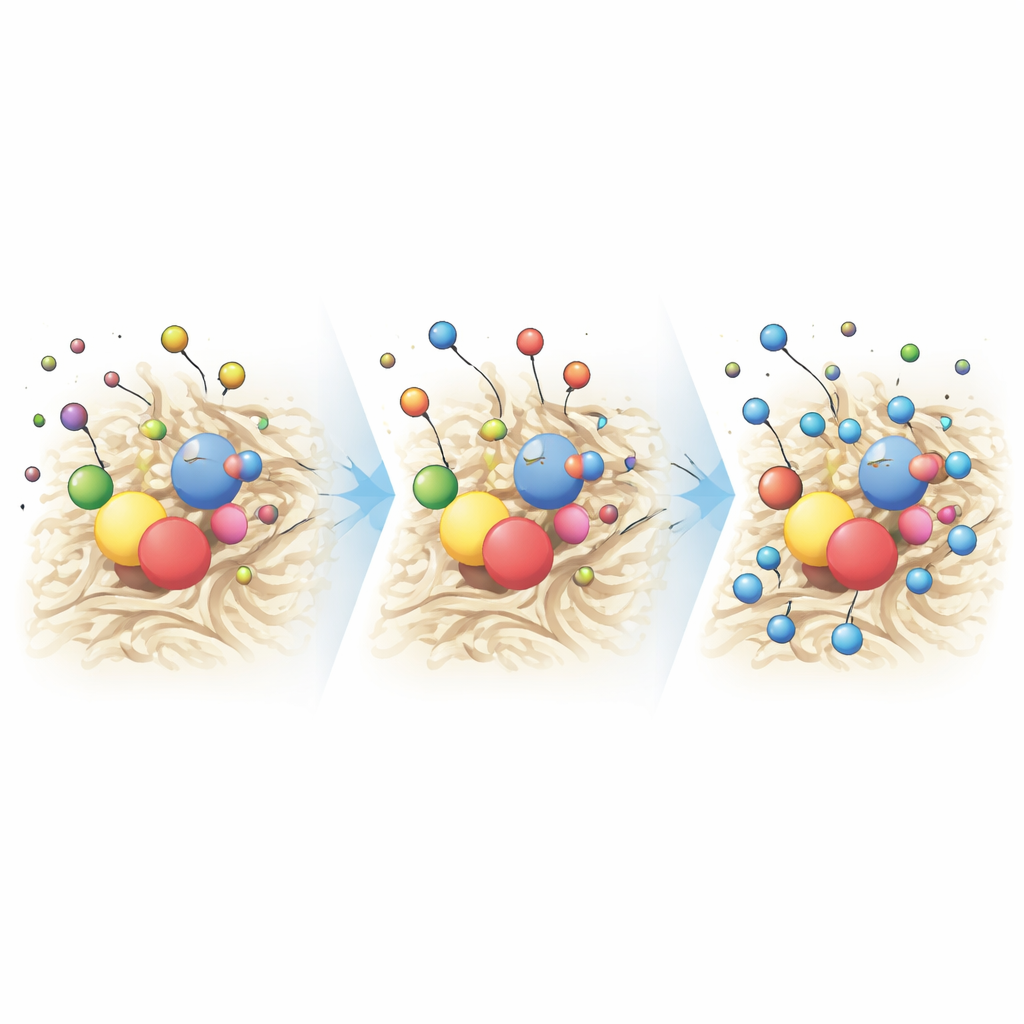
Toward Safer Water with Gentle Materials
In everyday terms, this study shows that a carefully engineered blend of a natural “bio-sponge” and a tri-phase mineral can strip dangerous metals like lead and cadmium from water to almost undetectable levels, using only modest amounts of material and simple operating conditions. While further work is needed to test long-term reuse, performance with real wastewater, and large-scale economics, the findings point toward a future in which clean water can be produced using sunlight, gentle chemistry, and materials that are kind to both people and the planet.
Citation: Erian, G.R., Abdelmonem, N., Abdelghany, A. et al. Microstructure-guided design of biopolymer-supported tri-phasic TiO2 for sustainable lead and cadmium detoxification. Sci Rep 16, 10530 (2026). https://doi.org/10.1038/s41598-026-43155-x
Keywords: heavy metal removal, water purification, chitosan composite, titanium dioxide, nanomaterials