Clear Sky Science · en
Molecular analysis of somatic mutations at the HPRT locus in lymphocytes of human population exposed to chronic high background natural radiation
Why living with natural radiation matters
Along parts of India’s southwest coast, people have lived for generations on beaches naturally enriched with radioactive minerals. That means their bodies quietly receive more radiation each year than most of us ever do. This study asks a question with big implications for public health and nuclear safety: does that lifelong, slightly elevated exposure leave extra scars on their DNA, or have our cells learned to cope without obvious harm?
A coastal community under the microscope
The researchers focused on men living in Kerala’s “high-level natural radiation areas,” where radioactive elements such as thorium and uranium occur in monazite-rich beach sands. Annual external doses in these zones can be several to many times higher than in nearby areas with typical background radiation. The team recruited 37 healthy adult men: some from normal radiation regions and others from areas with low and higher ends of the natural radiation range. All were long-term residents with similar lifestyles, allowing the scientists to home in on radiation as the main environmental difference.
A genetic early-warning signal in blood cells
To look for subtle genetic damage, the study used a well-known “sentinel” gene called HPRT, located on the X chromosome in blood T cells. Changes in this gene can be detected using a special growth test: normal cells die when exposed to a toxic drug, but mutant cells lacking working HPRT survive and form colonies. By counting these colonies and then examining the structure of the gene, the researchers can estimate how often mutations arise and what kinds of changes occur—small tweaks, missing pieces, or large chunk deletions that hint at serious DNA mishaps.
Checking for hidden breaks and missing pieces
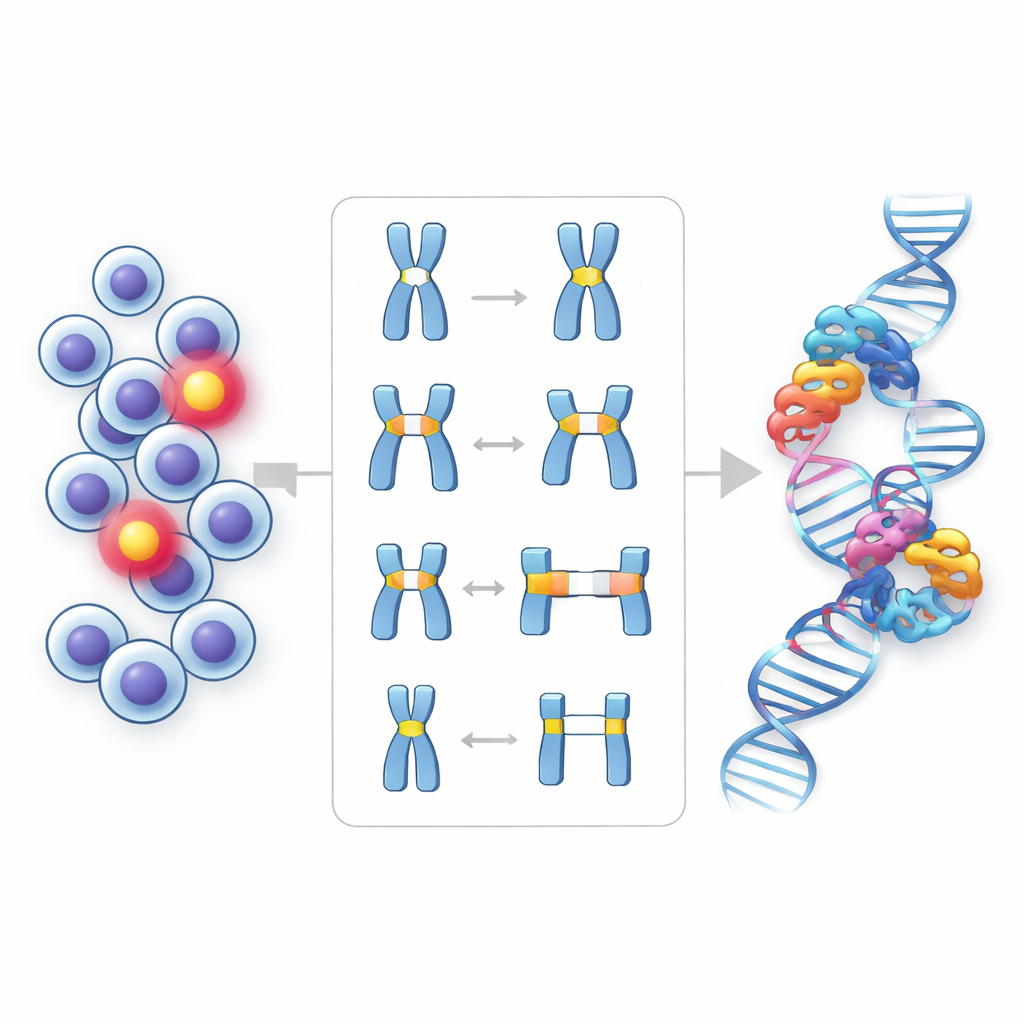
The team isolated lymphocytes from each volunteer’s blood and grew them under conditions that select for HPRT mutants. They then analyzed 224 mutant colonies using a combination of polymerase chain reaction (PCR) and fine-scale chromosome mapping. This allowed them to see whether whole segments of the gene were missing, whether only end sections had been cut off, or whether smaller internal stretches had been lost. They also used nearby genetic landmarks spanning more than three million DNA letters to understand how far some deletions extended beyond the HPRT gene itself, and they checked whether repeated copies of the same mutation came from a single rogue cell that had multiplied.
What the DNA actually revealed
Despite clear differences in environmental radiation levels, the frequency of HPRT mutations in blood cells was strikingly similar across all groups, including those receiving the highest natural doses. The overall pattern of mutations—whether the gene was intact, partially deleted, or entirely missing—also did not differ in any meaningful way. Large deletions reaching up to about 1.2 million DNA letters occurred in both low- and high-radiation groups, but neither size nor type of these deletions increased with higher background exposure. Most mutants (about three out of four) did not involve deletions at all, suggesting more subtle, small-scale changes that still need to be cataloged by sequencing.
Subtle shifts in the cell’s repair machinery
While the amount and pattern of damage looked similar, the researchers did notice differences in how cells responded. When they compared gene activity in mutant and non-mutant T-cell colonies, several genes involved in recognizing DNA damage and coordinating repair—such as those linked to fixing breaks or correcting mismatches—were more active in mutant cells. This held true in both normal and high-radiation regions, though the specific repair genes showing higher activity varied somewhat between groups. The pattern suggests that once the HPRT gene is altered, the cell’s broader repair network may adjust its activity, perhaps as a compensatory or adaptive response.
What this means for people living with higher natural radiation
For residents of Kerala’s high-radiation beaches—and for scientists shaping radiation safety guidelines—the take-home message is cautiously reassuring. In this study, chronic exposure to somewhat higher natural background radiation did not increase the rate of detectable HPRT mutations in blood T cells, nor did it cause more or larger deletions in this gene region compared with people from normal areas. At the same time, the altered activity of certain repair genes hints that cells may fine-tune their internal defense systems in response to ongoing low-level stress. Together, these results support the idea that, at least for this key genetic marker in blood cells, living in a naturally more radioactive environment does not necessarily translate into greater long-term genetic damage, while underscoring the need for broader, genome-wide studies to fully map the limits of our cellular resilience.
Citation: Gopinathan, A., Jain, V., Sharma, D. et al. Molecular analysis of somatic mutations at the HPRT locus in lymphocytes of human population exposed to chronic high background natural radiation. Sci Rep 16, 13709 (2026). https://doi.org/10.1038/s41598-026-43100-y
Keywords: natural background radiation, somatic mutations, DNA repair, Kerala coast, genome stability