Clear Sky Science · en
Cycloastragenol attenuates osteoarthritis by restoring chondrocyte senescence via the NRF2/NF-κB signaling axis
Why this matters for aching joints
Osteoarthritis is often described as simple “wear and tear” of the joints, but growing research shows that the story is much more complex—and more hopeful. This study explores a natural compound called cycloastragenol, derived from a traditional Chinese herb, and asks whether it can slow the cellular aging processes that drive joint damage. By protecting the tiny cells that maintain cartilage, the work points toward future treatments that might do more than just relieve pain and instead help preserve the joint itself. 
The hidden life of cartilage cells
Healthy knee cartilage is a smooth, springy cushion built and maintained by specialized cells called chondrocytes. In osteoarthritis, these cells are bombarded by chronic low-grade inflammation and chemical stress from reactive oxygen species—highly reactive molecules that damage DNA, proteins, and cell structures. Over time, many chondrocytes enter a state known as cellular aging or senescence: they stop dividing, lose their ability to repair cartilage, and start secreting a mix of inflammatory and tissue‑breaking substances that further erode the joint. This creates a vicious cycle in which damaged cells poison their neighborhood, accelerating cartilage loss and pain.
A plant molecule with anti‑aging promise
Cycloastragenol has attracted attention because of its reported anti‑aging and antioxidant properties in other tissues. The researchers asked whether it could protect rat cartilage cells exposed to strong oxidative stress in the lab and whether it could ease osteoarthritis in a rat knee model. They first confirmed that cycloastragenol did not harm the cells at the tested doses. When the cells were challenged with a chemical that mimics oxidative stress, they showed clear signs of aging: classic senescence markers rose, more cells stained positive for a standard aging test, and their ability to proliferate dropped. Adding cycloastragenol before the stress largely reversed these changes, reducing aging markers and partially restoring cell growth.
Protecting the cartilage scaffold
Cartilage strength depends on a carefully balanced scaffold of proteins such as type II collagen and aggrecan. In osteoarthritis, senescent chondrocytes dial down production of these building blocks and ramp up enzymes that chew them apart. In the stressed cell model, the authors observed exactly this pattern: protective matrix components fell, while destructive enzymes increased. Cycloastragenol shifted the balance back toward protection—boosting the good structural proteins and suppressing the breakdown enzymes. Chemical staining showed that the sugary chains that help cartilage hold water, which were depleted by oxidative stress, were also better preserved when cycloastragenol was present.
A molecular tug‑of‑war inside the cell
Digging deeper, the team focused on two major control systems that sense cell stress. One, centered on the protein NRF2, acts as an internal defense switch that turns on antioxidant and detoxifying genes. The other, built around NF‑κB, drives inflammatory responses and many of the damaging secretions from aged cells. Computer modeling suggested that cycloastragenol can bind to KEAP1, a protein that normally keeps NRF2 in check. In experiments, cycloastragenol activated NRF2, increased its movement into the nucleus, and boosted several of its downstream protective proteins. At the same time, it calmed NF‑κB signaling, reducing key steps that normally allow this pathway to turn on inflammatory and cartilage‑degrading factors. When the researchers deliberately reduced NRF2 levels with genetic tools, cycloastragenol lost much of its power: oxidative stress rose again, mitochondrial health worsened, NF‑κB reactivated, and both cell aging and matrix damage rebounded. 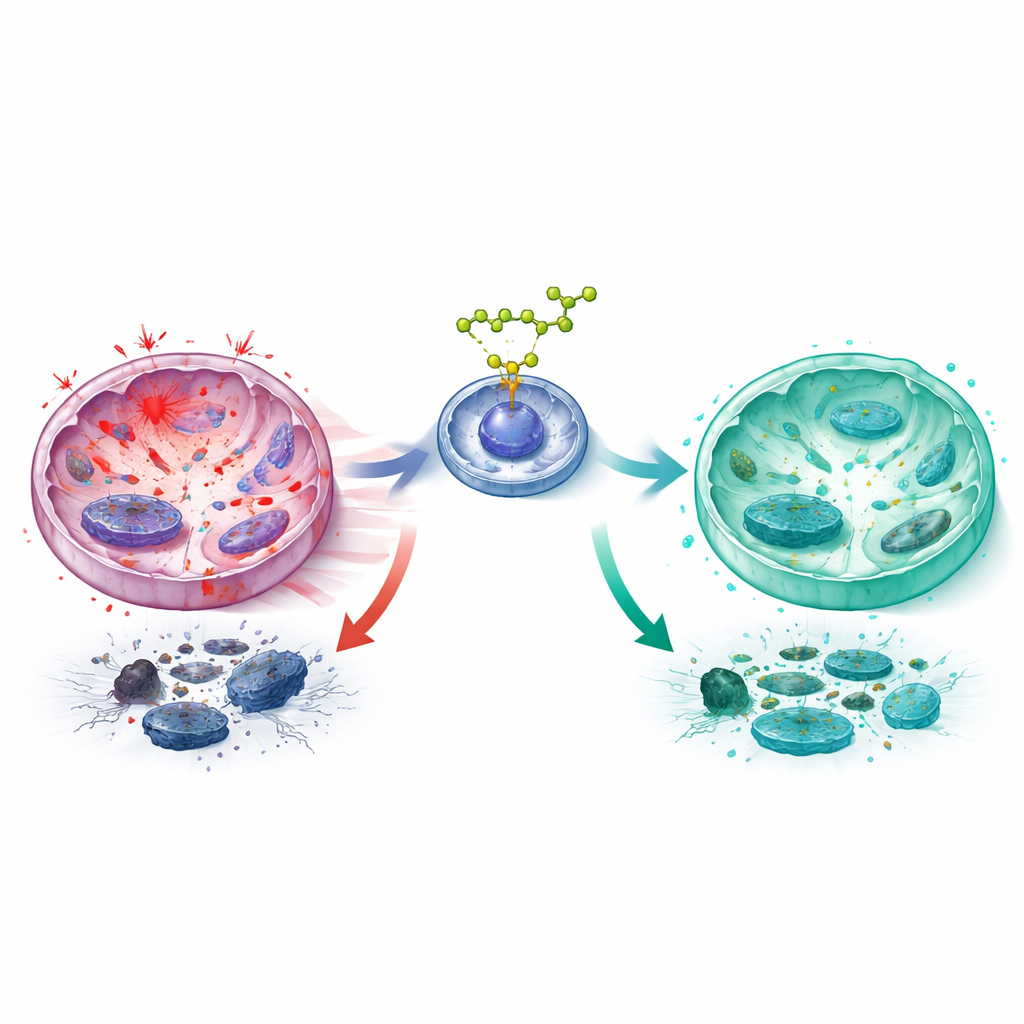
From petri dish to living joint
To see whether these cellular effects matter in a whole joint, the team used rats with chemically induced osteoarthritis of the knee. Animals that received weekly injections of cycloastragenol into the joint showed thicker, better‑organized cartilage, with fewer gaps and less loss of the cartilage‑specific pigments seen in standard stains. Microscopic analysis revealed more of the key structural protein collagen II, less of the destructive enzyme ADAMTS5, fewer senescent cells, and a higher presence of NRF2 in cartilage cell nuclei. Standard scoring systems for cartilage damage confirmed that treated joints were significantly healthier than untreated osteoarthritic joints.
What this could mean for future therapies
Taken together, the findings suggest that cycloastragenol helps protect joint cartilage by reawakening the cell’s own antioxidant defenses through NRF2 and dialing down inflammatory NF‑κB signals that drive cell aging and tissue breakdown. For a layperson, this means that instead of simply masking pain, a future drug inspired by cycloastragenol might help keep the “repair crew” cells in cartilage younger and more functional, slowing the structural decline of the joint. While this work is still in animals and lab‑grown cells, and questions remain about dosing, long‑term safety, and how well the results will translate to humans, it adds to a growing body of evidence that targeting cellular aging pathways could open a new chapter in osteoarthritis treatment.
Citation: Zhang, S., Zou, Y., Long, J. et al. Cycloastragenol attenuates osteoarthritis by restoring chondrocyte senescence via the NRF2/NF-κB signaling axis. Sci Rep 16, 13203 (2026). https://doi.org/10.1038/s41598-026-43064-z
Keywords: osteoarthritis, cartilage, cellular aging, antioxidants, natural compounds