Clear Sky Science · en
Immunoproteomic insights into inflammatory diseases of the critically endangered black rhinoceros (Diceros bicornis)
Why Sick Rhinos Matter to Us
The black rhinoceros is one of the planet’s most endangered large mammals, with only a few thousand left in the wild and fewer than 200 living in zoos and breeding centers. These ex situ animals are meant to be a safety net against extinction, yet many develop puzzling health problems—chronic inflammation, dental disease, gut trouble, and liver damage—that are rarely seen in wild rhinos. This study uses cutting‑edge protein analysis and machine learning to peer inside the immune cells of these animals, in an effort to uncover what is driving their illness and how we might better protect them.
Hidden Health Problems in Captive Rhinos
Black rhinos kept under human care are not simply wild animals in safer surroundings. They often suffer from a cluster of disorders: sore and infected gums, unstable gut function, and a tendency to accumulate iron in the liver. Routine blood tests and basic inflammation markers have shown that many of these rhinos live in a chronically inflamed state, but those tests cannot explain what is going wrong at a cellular level. To tackle this mystery, the researchers collected blood from 27 captive black rhinos—representing a large fraction of the North American population—and isolated a type of white blood cell that constantly patrols the body for trouble. They then measured, in a sweeping way, which proteins were present in these cells.
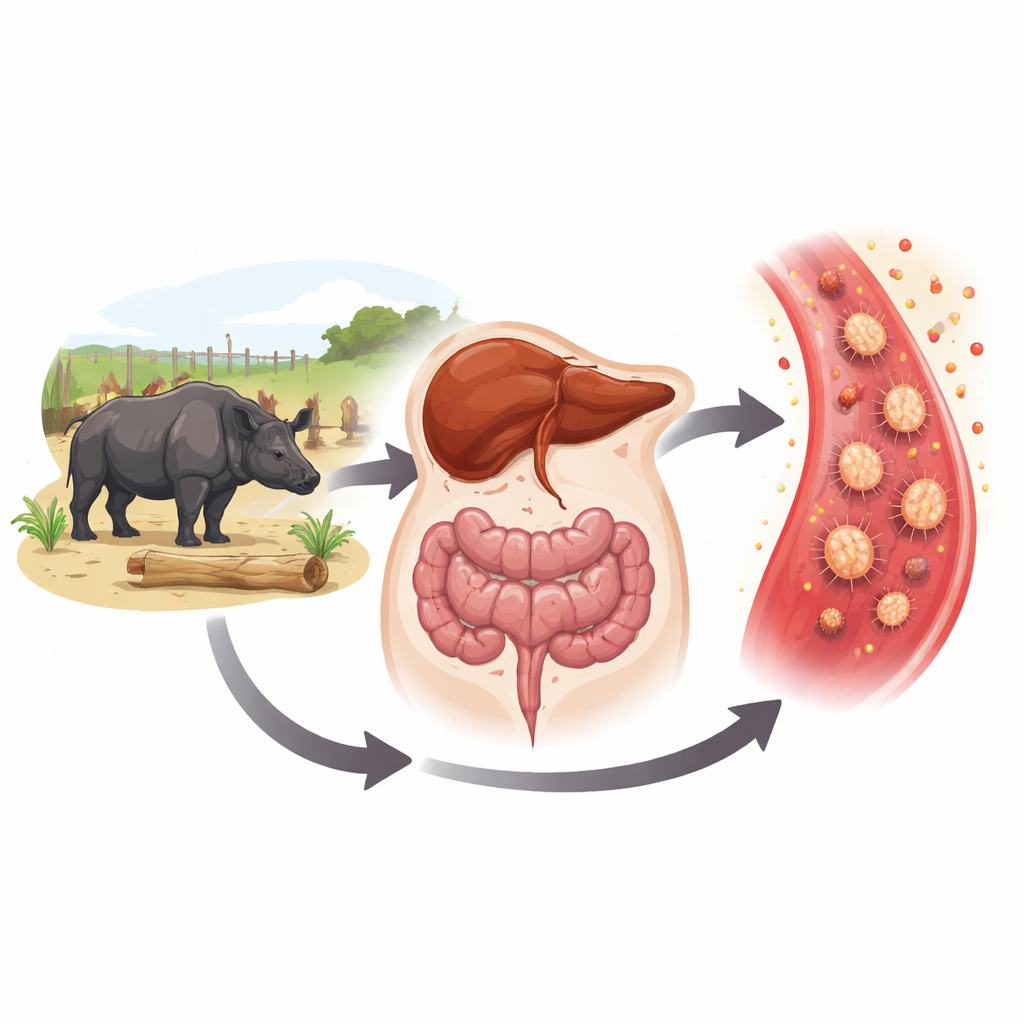
Mapping the Immune Landscape
The team created what they call an “immunoproteome”: a catalog of 1,311 different proteins found in the rhinos’ immune cells. At first, they tried to see whether this protein landscape lined up with obvious categories, such as sex, subspecies, or whether a rhino was thought to be clinically healthy or inflamed based on veterinary records. Surprisingly, none of these standard labels matched clear differences in protein patterns. Even a commonly used inflammation marker, serum amyloid A, failed to cleanly separate “healthy” from “sick” animals, and some supposedly healthy rhinos had values hinting at hidden disease. This suggested that many animals might be experiencing silent or fluctuating illness that does not always show up in clinical exams.
Letting the Data Speak for Itself
Because traditional groupings did not explain the results, the scientists turned to an unsupervised machine learning method called consensus clustering. Instead of telling the computer which samples belonged together, they let the structure of the protein data define its own groups. This approach revealed two distinct classes of samples that were not tied to sex, subspecies, age, or prior health labels. Forty‑three proteins were mainly responsible for separating these classes, and most of them are linked to inflammation in humans and laboratory animals, including conditions such as gum disease, disturbed gut communities, and whole‑body inflammatory syndromes. Notably, repeat samples from the same rhino sometimes switched from one class to the other over time, indicating that individuals can move in and out of a more inflamed state.
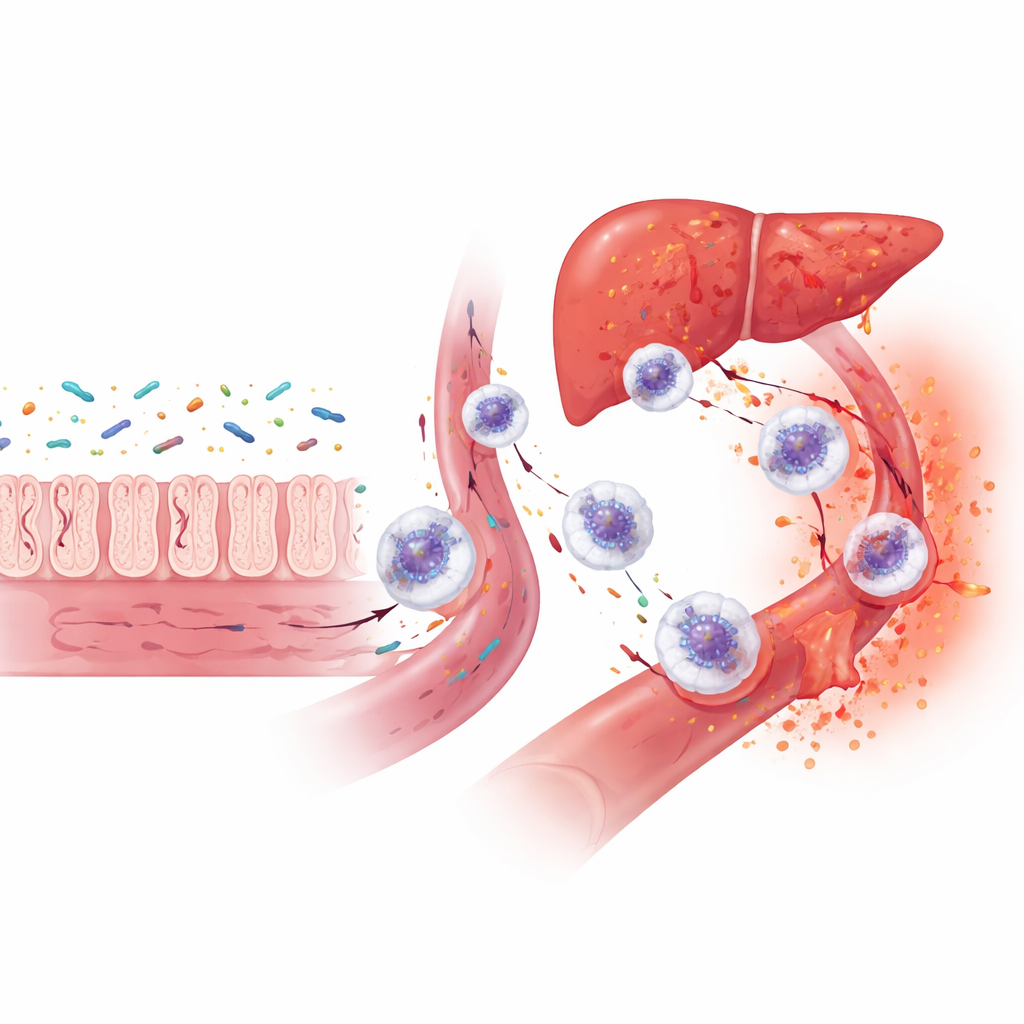
A Troubled Highway from Mouth to Liver
When the researchers examined how these 43 proteins function together, a dominant theme emerged: intense activity of neutrophils, a front‑line white blood cell that releases enzyme‑filled granules to attack microbes. This process, called degranulation, is useful in short bursts but can damage the body’s own tissues if it becomes excessive or chronic. Many of the proteins linked to the inflamed class are the same ones elevated in people with severe gum disease, inflammatory bowel conditions, or liver injury. Putting this evidence together, the authors propose that in captive black rhinos, microbes and their products leak from the mouth and gut into the bloodstream, travel along the “oral‑gut‑liver” axis, and trigger repeated waves of neutrophil activity. Over time, this could erode dental tissues, upset the gut, and inflame the liver, contributing to the complex disease picture seen in these animals.
What This Means for Saving Rhinos
This work provides the first detailed immune‑cell protein map for any endangered mammal and offers promising molecular signposts—those 43 proteins—that could one day help veterinarians detect early, hidden disease in black rhinos. The findings also shift attention toward managing the health of the whole mouth–gut–liver system, for example by adjusting diet, improving dental care, and monitoring inflammation more precisely. With further testing and validation, these candidate biomarkers and the oral‑gut‑liver framework could guide better treatment and husbandry strategies, improving the odds that ex situ rhino populations remain robust enough to support future reintroductions to the wild.
Citation: Corder, M.L., Abulez, T., Cleland, T. et al. Immunoproteomic insights into inflammatory diseases of the critically endangered black rhinoceros (Diceros bicornis). Sci Rep 16, 13535 (2026). https://doi.org/10.1038/s41598-026-43055-0
Keywords: black rhinoceros, inflammation, oral-gut-liver axis, proteomics, wildlife conservation