Clear Sky Science · en
Photocatalytic degradation of polyethylene films using green-synthesized ZnO and Fe3O4 nanoparticles from Acacia nilotica
Turning Plants and Sunlight Against Stubborn Plastics
Plastic shopping bags and packaging are built to last, which is exactly the problem once they escape into rivers, farms, and dumps. This study explores a nature-inspired strategy to help those plastics break down faster. By using leaf extracts from a common tree, Acacia nilotica, the researchers created tiny particles that, when exposed to sunlight in water, help brittle plastic films crack, oxidize, and gradually fall apart. The work points toward a future in which plant-based nanotechnology could make plastic waste less permanent.

Why Everyday Plastics Are So Hard to Get Rid Of
Low-density polyethylene (LDPE) and high-density polyethylene (HDPE) are among the most widely used plastics, found in bags, wraps, and many everyday goods. Their long, tightly bonded chains of carbon and hydrogen resist attack by water, microbes, and sunlight, so they can linger in soil and water for decades. Traditional cleanup methods struggle with these materials, and even advanced treatments often rely on harsh chemicals or high energy input. The authors frame this as a growing global challenge and argue that we need ways to speed up plastic breakdown that are both effective and environmentally gentle.
Using a Medicinal Tree to Make Helpful Nanoparticles
Instead of manufacturing nanoparticles with toxic solvents and high heat, the team turned to plants as miniature chemistry labs. They compared several tropical species and found that Acacia nilotica leaves were especially rich in natural compounds called phenolics and tannins. These molecules act like tiny reducing and stabilizing agents, helping dissolved metal salts transform into solid particles while keeping them from clumping. Using the acacia extract, the researchers produced zinc oxide (ZnO) and iron oxide (Fe₃O₄) nanoparticles. Tests showed that the particles were very small—on the order of tens of nanometers—uniformly dispersed, and crystalline, with lots of surface area and pores. Optical measurements confirmed that they behave like light-sensitive semiconductors, capable of absorbing ultraviolet rays and generating highly reactive forms of oxygen.
Putting Sun-Activated Particles to Work on Plastic Films
To see whether these plant-made nanoparticles could actually damage plastics, the scientists immersed small LDPE and HDPE film squares in water containing either ZnO or Fe₃O₄ and left them outdoors in Nigerian sunlight for 30 days. Similar control samples sat in plain water or in the dark. Over time, the films exposed to nanoparticle suspensions under sunlight lost weight, changed chemistry, and developed visible surface damage, while the controls remained essentially unchanged. LDPE, which has a more open and less ordered structure, lost about a quarter of its mass, whereas the tougher, more crystalline HDPE lost under 10 percent. Measurements of pH and microscopic images supported the idea that sunlight hitting the nanoparticles produces reactive oxygen species that attack the plastic surface, introducing oxygen-rich groups, forming cracks and cavities, and breaking long chains into shorter fragments.
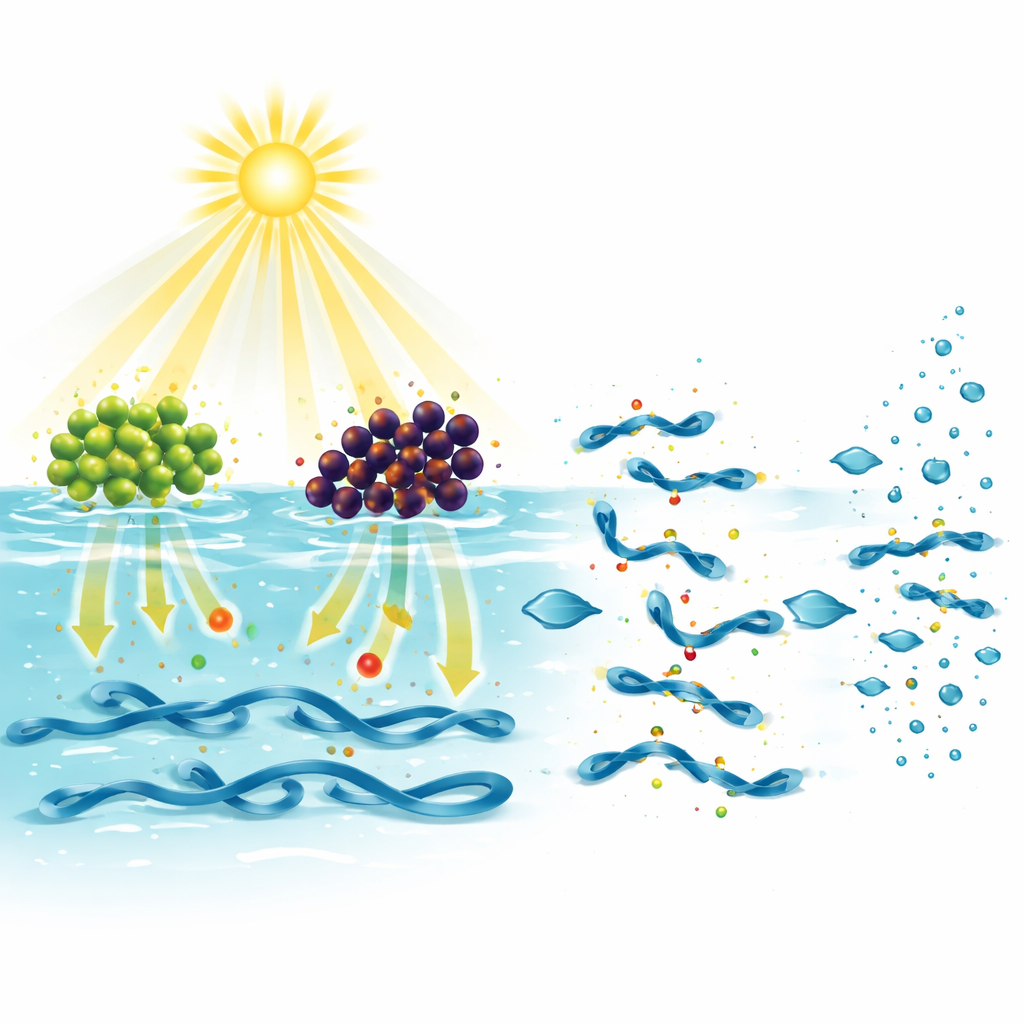
Following the Trail from Cracks to Carbon Dioxide
Beyond surface scarring and weight loss, the team wanted to know how far the breakdown actually went. Chemical fingerprinting of the plastics after treatment revealed new signals for carbonyl, hydroxyl, and related oxygen-containing groups—classic markers that the original hydrocarbon chains have been oxidized. Electron microscopy showed roughened, pitted surfaces with embedded zinc or iron, and stronger oxygen signals than in untreated samples. In carefully sealed systems, the researchers also trapped and measured carbon dioxide gas that formed during sunlight exposure. They found that only a small fraction of the plastic’s carbon, a few percent at most, was fully mineralized to CO₂ over the month-long test. This suggests that the main effect so far is oxidation and fragmentation rather than complete conversion to simple gases.
What This Approach Could Mean for Plastic Waste
In plain terms, the study shows that tiny particles made with help from a common tree can make stubborn plastic films more vulnerable to sunlight and water. The acacia-based zinc and iron oxides are highly active under natural sunlight, roughening and oxidizing polyethylene surfaces and causing measurable mass loss—especially in softer LDPE. At the same time, the process is still far from turning most of the plastic into harmless carbon dioxide within a month. Even so, by replacing harsh manufacturing methods with plant extracts and tapping free solar energy, this work outlines a promising, greener path for weakening persistent plastics and making them easier for the environment or follow-up treatments to finish off.
Citation: Shaibu, A., Tijani, J.O., Abdulkareem, A.S. et al. Photocatalytic degradation of polyethylene films using green-synthesized ZnO and Fe3O4 nanoparticles from Acacia nilotica. Sci Rep 16, 14212 (2026). https://doi.org/10.1038/s41598-026-43013-w
Keywords: plastic degradation, green nanotechnology, zinc oxide nanoparticles, iron oxide nanoparticles, photocatalysis