Clear Sky Science · en
Epigenetic alterations in type 1 diabetes and their association with poor glycemic control: the SED1-EPI substudy
Why tiny blood messengers matter
For people living with type 1 diabetes, day-to-day life already involves constant blood sugar checks, insulin dosing, and medical visits. Yet doctors still lack simple tools to predict who is at higher risk of long-term complications or whose blood sugar control is quietly slipping. This study explores whether tiny molecules that circulate in the blood, called microRNAs, could act as early warning signals—offering a new window into how well diabetes is being managed and how stressed the insulin-producing cells of the pancreas really are.
A closer look at type 1 diabetes
Type 1 diabetes is an autoimmune disease in which the body’s own defenses destroy the insulin-producing beta cells in the pancreas. Without insulin, sugar builds up in the bloodstream, so people with type 1 diabetes must rely on insulin injections or pumps for life. Even with careful treatment, many struggle to keep their blood sugar in the recommended range, and long-term high levels are linked to eye, kidney, nerve, and heart problems. Doctors routinely use a lab test called HbA1c to capture the average blood sugar over several months, but this number does not fully explain why some people develop complications while others do not.
What microRNAs can reveal
MicroRNAs are short strands of genetic material that do not make proteins themselves but instead act as fine-tuning switches on many genes at once. They circulate in all body fluids and can be measured from a simple blood sample, making them attractive candidates as “liquid biopsy” markers. Earlier work from the same research group had suggested that certain microRNAs are altered in people with long-standing type 1 diabetes. In this new study, the authors set out to confirm those results and see how strongly these markers relate to blood sugar control and body measurements in a larger and more varied group of adults from across Spain.
The Spanish multicenter study
The team analyzed blood plasma from 125 volunteers, including 76 adults with type 1 diabetes and 49 people without diabetes, recruited at nine hospitals. They focused on eight specific microRNAs chosen from a previous discovery study because they showed the largest differences and were suspected to play roles in diabetes-related damage. Using a sensitive laboratory technique, they quantified the levels of each microRNA and compared them between groups, while also examining links with HbA1c, body mass index, and other clinical features. Advanced statistical methods were used to adjust for age and body size so that any differences would more likely reflect the disease itself.
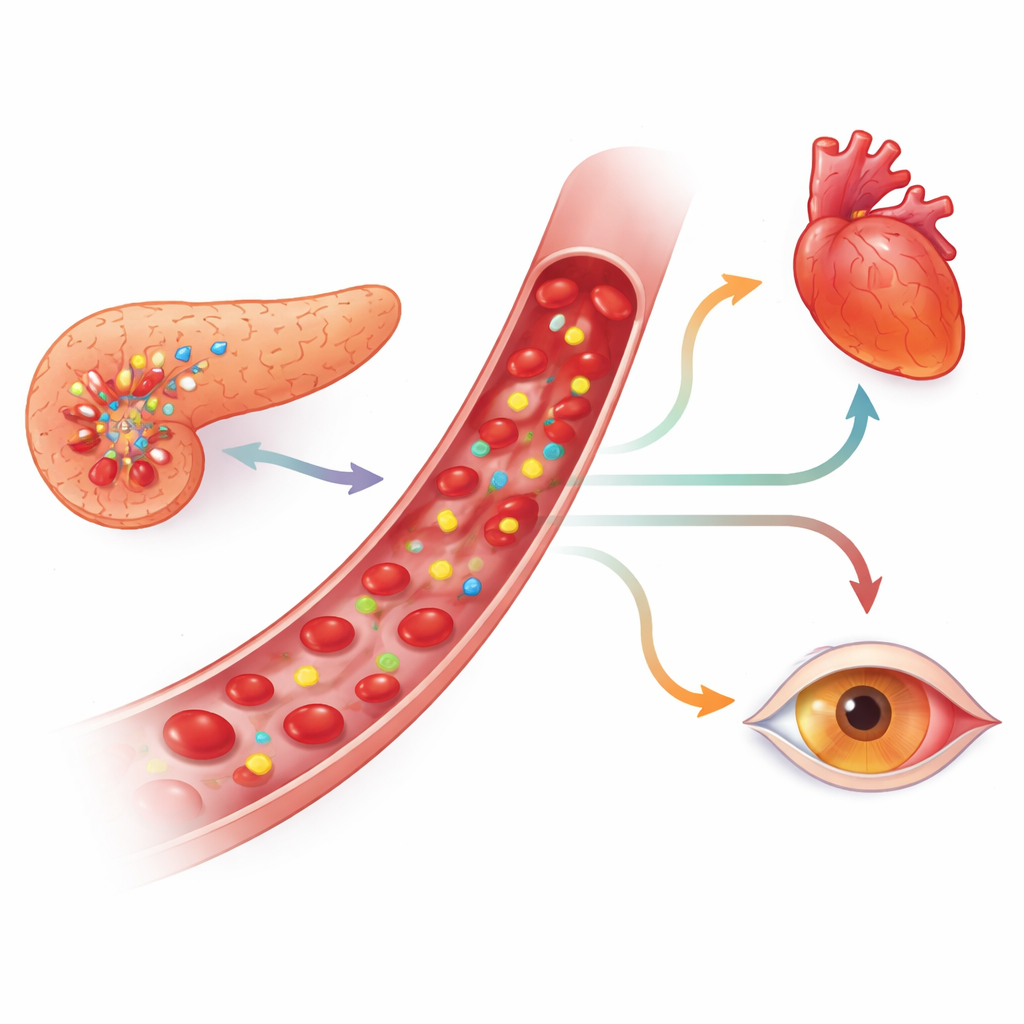
Signals of blood sugar control and cell stress
Among the eight candidates, one microRNA—called hsa-miR-200a-3p—stood out. It was consistently higher in people with type 1 diabetes than in those without, even after accounting for age and weight. Within the diabetes group, higher levels of this microRNA, along with two others (hsa-miR-1-3p and hsa-miR-340-5p), tracked with higher HbA1c values, meaning worse average blood sugar. People whose HbA1c was 7.5% or above showed clearly elevated levels of these three markers compared with those with better control. Another microRNA, hsa-miR-224-5p, was linked to body mass index, hinting at ties between weight and epigenetic changes in diabetes.
Clues to future complications
To understand what these patterns might mean biologically, the researchers used computer tools to map which genes and pathways are likely influenced by the three glucose-related microRNAs. The analysis highlighted networks involved in insulin signaling and several heart and blood vessel pathways, including those connected to heart muscle contraction and certain forms of heart thickening. These links fit with earlier reports that microRNAs in the same families are active in pancreatic beta cells and in tissues vulnerable to diabetes complications, such as the heart and retina. While the study did not find clear differences based on the presence of diagnosed complications or high blood pressure, it suggests that these microRNAs could be early indicators of stress and vascular risk before damage fully appears.
What this means going forward
In everyday terms, the findings suggest that specific tiny molecules in the blood may mirror how hard the body is working to cope with type 1 diabetes. One of them, hsa-miR-200a-3p, seems closely tied to the stress of insulin-producing cells, while hsa-miR-1-3p and hsa-miR-340-5p appear to track how well blood sugar is controlled and possibly the strain on the heart and blood vessels. The study is not large enough, and the clinical data are not detailed enough, to change care today, and the authors note important limitations in how these markers were measured. Still, their work strengthens the idea that a simple blood test could someday help doctors identify patients at higher risk earlier, tailor treatments more precisely, and monitor the hidden impact of long-term high blood sugar in type 1 diabetes.
Citation: García, A.V., Lambert, C., Villa-Fernández, E. et al. Epigenetic alterations in type 1 diabetes and their association with poor glycemic control: the SED1-EPI substudy. Sci Rep 16, 13430 (2026). https://doi.org/10.1038/s41598-026-42995-x
Keywords: type 1 diabetes, microRNAs, epigenetics, glycemic control, cardiovascular risk