Clear Sky Science · en
Treatment management algorithm for natural frozen embryo transfer cycles using a real-time ovulation prediction machine learning model
Why timing matters in fertility treatment
For people undergoing in vitro fertilization, the moment an embryo is returned to the uterus can make the difference between a failed attempt and a healthy pregnancy. In natural frozen embryo transfer cycles, doctors try to match embryo transfer to a woman’s own ovulation, but finding that exact day usually means repeated blood tests, ultrasounds, and expert judgment. This study introduces a new artificial intelligence (AI) tool that predicts ovulation in real time and suggests when to test and when to transfer, aiming to make treatment both more accurate and less burdensome.
A new digital helper for natural cycles
The researchers set out to build a “treatment management algorithm” for natural-cycle frozen embryo transfer (NC-FET). In these cycles, embryos that were previously frozen are transferred back into the uterus without using hormones to artificially control the menstrual cycle. NC-FET is increasingly popular because it is linked to lower rates of pregnancy loss and fewer complications such as high blood pressure in pregnancy. However, it requires careful tracking of the body’s own signals to correctly identify ovulation, which has traditionally meant frequent clinic visits, blood draws, and ultrasound scans.
Teaching an AI to read the body’s signals
To create their algorithm, the team used a “teacher-student” approach. First, an earlier model—the “teacher”—had been trained on hundreds of cycles where ovulation timing was decided by three fertility specialists using hormone levels and ultrasound images. That teacher model was highly accurate and was then used to assign ovulation days in nearly 4,000 additional natural frozen transfer cycles. The new “student” model learned from this much larger, labeled dataset. It looks at one or two monitoring days per cycle, combining information such as luteinizing hormone (LH), estrogen, progesterone, the ratio between estrogen and progesterone, follicle size, and thickness of the uterine lining. Based on these inputs, it predicts how many days remain until, or have passed since, ovulation.
From prediction to full-cycle management
The innovation in this paper is not just predicting ovulation, but using that prediction to manage the whole treatment. The Natural FET Treatment Management Algorithm (NTMA) starts by recommending when a patient should first be tested, typically around day 8 of the cycle. After each test, it uses the model’s output to decide whether another test is needed, when that test should occur, or whether ovulation timing is already clear enough to schedule the embryo transfer. If the model concludes that ovulation is already more than two days in the past and cannot be dated precisely, the cycle can be safely canceled before wasting an embryo. This step-by-step logic is evaluated using a statistical simulation that combines real-world ovulation patterns, the model’s accuracy at different time points, and the decision rules of the algorithm.
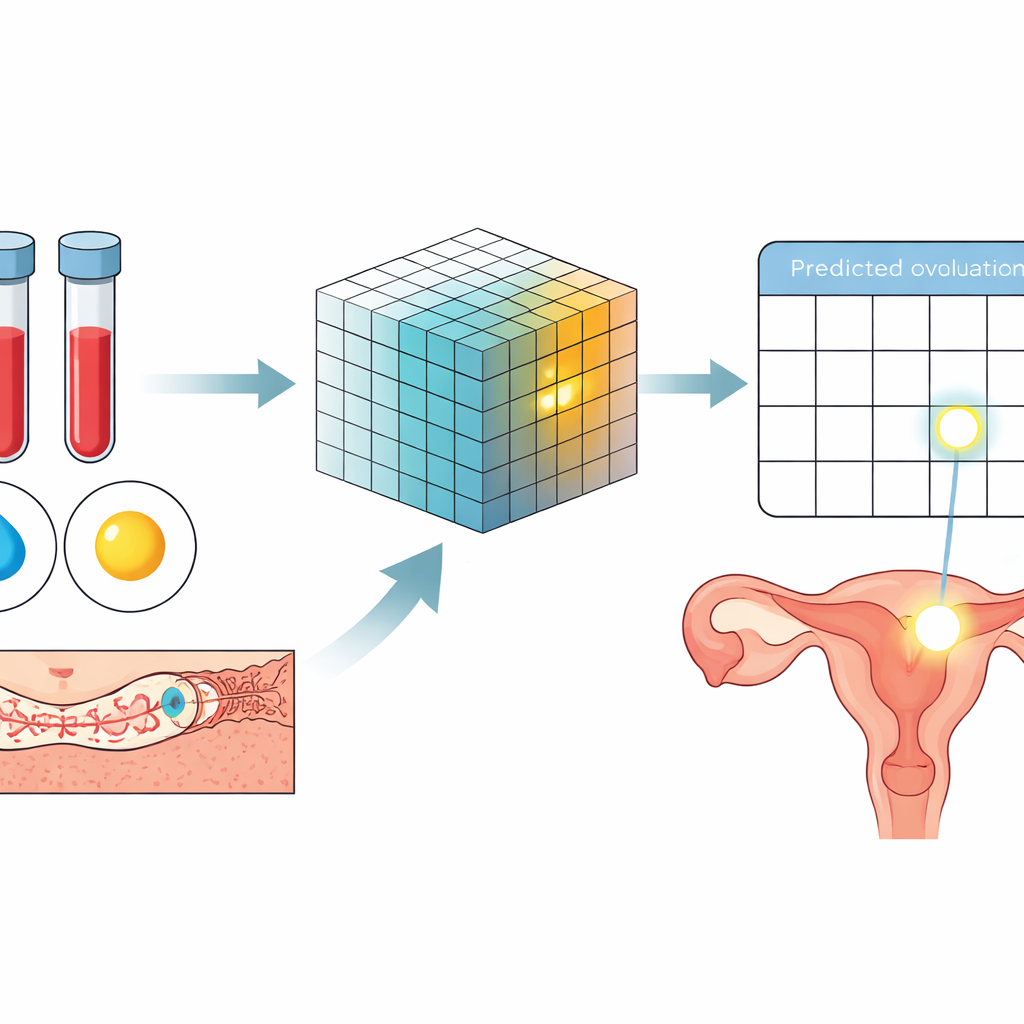
How well the system works
When tested, the real-time model proved especially accurate both far from ovulation (early in the cycle, when it can confidently say that ovulation is not near) and very close to it (the day before and the day of ovulation), where hormone changes are strongest. In a group of cycles with detailed ultrasound proof of ovulation, the model correctly identified the day before and the day of ovulation in about 95% of cases. When the full NTMA was evaluated through simulation, it successfully pinpointed the correct ovulation day in just over 92% of cycles, failed to make a prediction in about 1% of cycles, and was wrong in about 7%. On average, women would need only about 3.1 monitoring visits per cycle, fewer than in the historical data, suggesting a lower monitoring burden without sacrificing precision.
What this could mean for patients
To everyday patients and clinicians, this work suggests that an AI assistant can safely take on much of the day-to-day decision making in natural frozen embryo transfer cycles. By digesting complex patterns in hormone tests and ultrasound findings, the algorithm can recommend when to come in for testing and when to schedule transfer, with accuracy comparable to expert-driven approaches and with fewer clinic visits. The study is retrospective and does not yet prove higher live birth rates, so prospective trials are still needed. Still, this AI-driven management system points toward a future in which fertility care is more personalized, more efficient, and potentially more successful—helping embryos be transferred at just the right moment in the body’s own rhythm.
Citation: Moran, E., Hourvitz, A., Luz, A. et al. Treatment management algorithm for natural frozen embryo transfer cycles using a real-time ovulation prediction machine learning model. Sci Rep 16, 13727 (2026). https://doi.org/10.1038/s41598-026-42921-1
Keywords: fertility, artificial intelligence, ovulation, frozen embryo transfer, reproductive health