Clear Sky Science · en
Preparation of Co–Ce–Ru/γ-Al2O3 catalyst for degradation rhodamine B in dye wastewater
Why dirty dyes in water matter
Colorful clothes, plastics, and printed materials all rely on synthetic dyes, but the leftover dye water is extremely hard to clean and can linger in rivers and lakes for a long time. This study explores a new way to strip a bright red dye called rhodamine B from water almost completely, using a specially designed solid catalyst that works together with an oxidizing agent. The work matters because it points to a practical method for treating stubborn industrial wastewater without creating new pollution problems.
A new helper for cleaning tough dye water
The researchers focused on a group of treatment methods known as advanced oxidation processes, which rely on very reactive forms of oxygen and sulfur to tear apart complex chemicals. A widely used oxidizing agent, peroxymonosulfate, can be activated by certain metals to form powerful radicals that attack dye molecules. Cobalt is especially good at this job, but dissolved cobalt in water is a toxic heavy metal. To get cobalt to work hard while keeping it firmly in place, the team attached it to a porous support material called gamma alumina and then improved this base recipe by adding tiny amounts of the rare-earth metal cerium and the noble metal ruthenium.
Building a smarter solid catalyst
The scientists prepared several versions of the catalyst by soaking the alumina support in metal salt solutions and then heating it at controlled temperatures. One sample contained only cobalt, another combined cobalt and cerium, and the most advanced version contained cobalt, cerium, and ruthenium together. Detailed tests showed that the upgraded catalyst had a large internal surface area full of medium-sized pores, giving many spots where dye and oxidant could meet. The metals were spread evenly across the surface, and the presence of cerium and ruthenium helped keep cobalt in very small clusters, improved thermal stability, and created oxygen vacancies that favor fast reactions. Overall, the structure allowed more active sites to be available while using less cobalt.
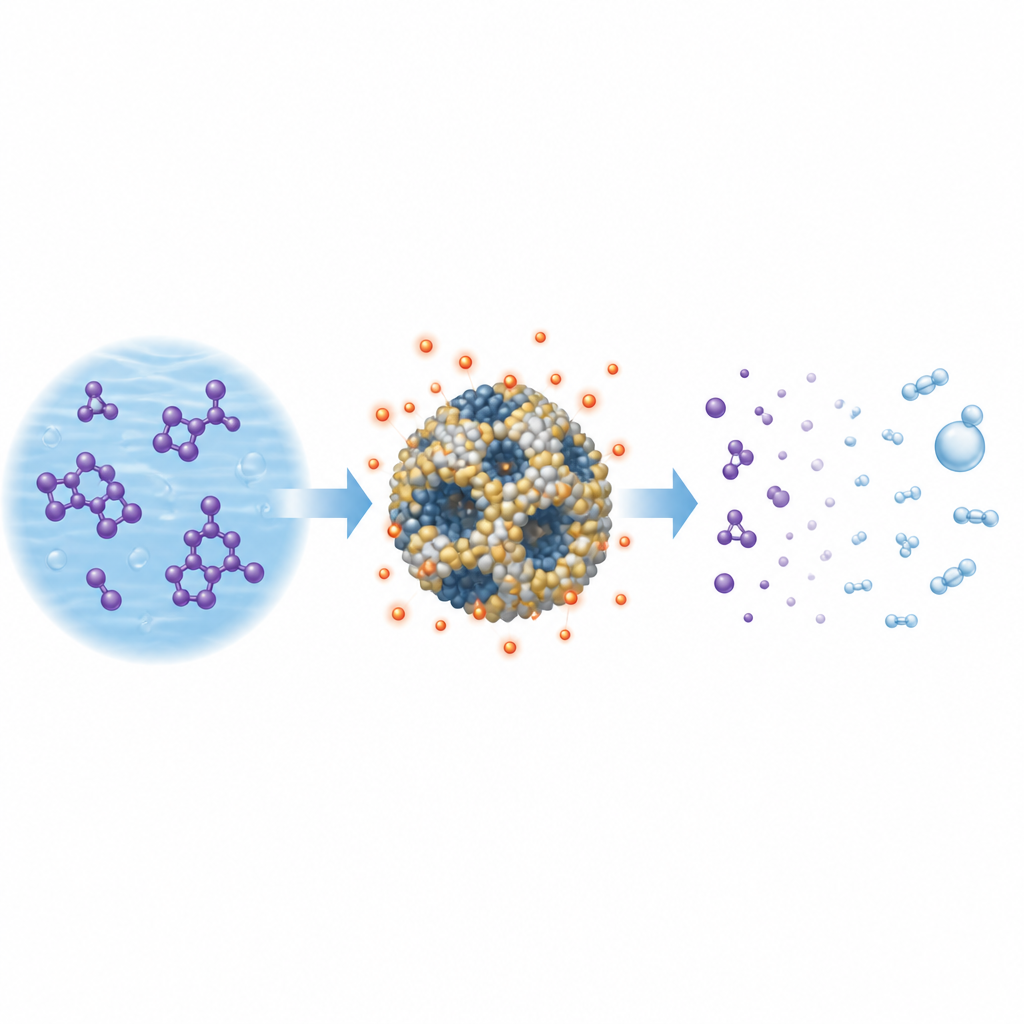
Putting the catalyst to the test
To see how well the materials worked in practice, the team treated water containing rhodamine B at realistic concentrations. In each test they mixed the dye solution with a measured amount of catalyst, allowed the dye to attach to the surface, and then added peroxymonosulfate. By tracking the loss of color over time with a light-absorption method, they could follow how fast the dye broke down. The cobalt–cerium–ruthenium catalyst almost completely removed the color, reaching close to 100 percent decolorization within about 20 to 30 minutes at room temperature with modest doses of catalyst and oxidant. The reaction followed simple first-order behavior, meaning that the rate scaled with how much dye remained, and the calculated half-life of the dye was only a few minutes.
How the cleaning action happens
Special magnetic measurements revealed which short-lived species were at work during treatment. The catalyst and peroxymonosulfate together generated both sulfate radicals and hydroxyl radicals, which are highly reactive forms of sulfur- and oxygen-based species that can rip apart complex dye structures. There was also evidence for a non-radical pathway involving a more selective form of oxygen. Cobalt on the catalyst surface cycled between different charge states, repeatedly activating fresh oxidant molecules. Cerium contributed by creating oxygen vacancies and shifting the balance of oxygen forms on the surface, while ruthenium subtly improved dispersion and stability. By adjusting how much catalyst, oxidant, and heat were supplied, the researchers identified operating conditions that gave fast treatment without wasting chemicals or causing radical self-quenching.
Durability and environmental safety
For any real-world use, the catalyst must survive many cleaning cycles and should not release large amounts of metals into the treated water. The team reused the cobalt–cerium–ruthenium catalyst four times under the same conditions. Although there was a small drop in performance, it still removed more than 90 percent of the dye after the fourth run, and microscopic images showed only minor surface wear. Measurements of dissolved metals confirmed that cobalt and the added helper metals mostly stayed locked in the solid, with cobalt levels well below 1.0 mg per liter, a common discharge limit. Other substances in the water, such as common salts and some metal ions, had only moderate influence on the process, and the method worked across a broad pH range.
What this means for cleaner water
In plain terms, this study shows that carefully engineering a solid catalyst lets industry harness the cleaning power of strong oxidants to remove stubborn dye molecules while using less toxic metal and avoiding major secondary pollution. The cobalt–cerium–ruthenium material on alumina efficiently activates peroxymonosulfate, survives repeated use, and keeps metal leakage low, making it a promising tool for treating colorful but persistent dye wastewater so that it no longer stains the environment.
Citation: Zhang, Y., Zhang, E., Deng, J. et al. Preparation of Co–Ce–Ru/γ-Al2O3 catalyst for degradation rhodamine B in dye wastewater. Sci Rep 16, 15093 (2026). https://doi.org/10.1038/s41598-026-42905-1
Keywords: dye wastewater, rhodamine B, advanced oxidation, cobalt catalyst, peroxymonosulfate