Clear Sky Science · en
Sustainable treatment of ceramic manufacturing wastewater using combined advanced oxidation and coagulation/precipitation processes with green nano zero-valent iron: multi-metal corrosion monitoring
Why dirty factory water matters to you
Ceramic tiles, dishes, and bathroom fixtures all start their lives in factories that use huge amounts of water. The dirty water that leaves these plants is not just muddy—it can quietly eat away at pipes, pumps, and tanks, driving up costs and risking leaks of polluted water into the environment. This study looks at a smarter way to clean ceramic factory wastewater using a "green" iron-based treatment, and asks a simple but crucial question: after treatment, how kind—or how harsh—is this water to the metals that carry it?
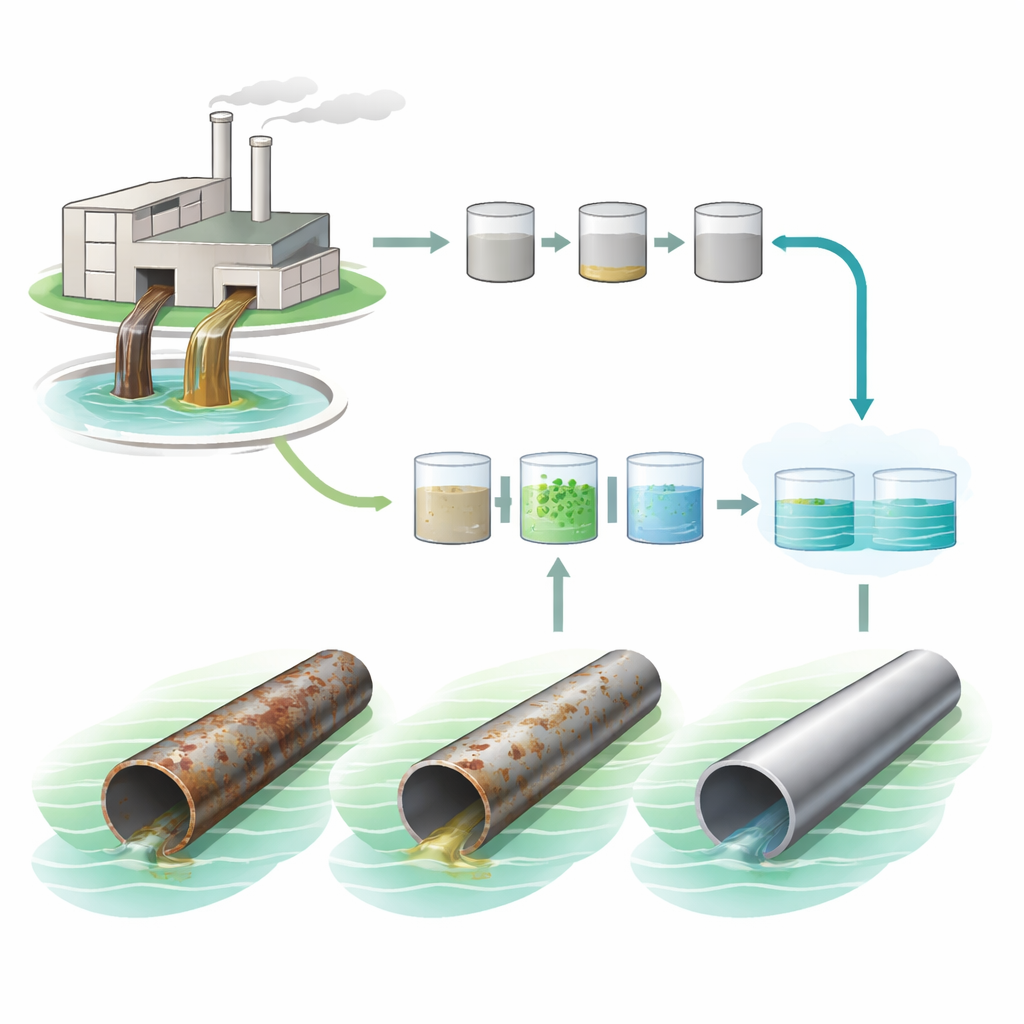
How tile factories turn water into a hidden hazard
The authors focus on a large ceramic factory in Egypt, where making each square meter of tile uses about 20 liters of water. Along the production line, this water picks up fine clays, silicates, colorants, and a mix of aggressive salts like chlorides and sulfates, plus traces of heavy metals and organic residues. Left untreated, this cocktail can harm rivers and soils. But even inside the factory fence it causes trouble: it speeds up rusting and pitting in steel pipes, stainless-steel tanks, and copper lines, forcing frequent repairs and replacements. Conventional treatment—mainly settling, filtration, and simple chemical steps—can make the water look cleaner without truly solving its corrosive bite.
A greener recipe for safer reuse
The researchers compare three versions of the same wastewater: completely untreated, water treated by the factory’s usual alum-based process, and water treated by a more advanced train of steps. This upgraded train combines Fenton oxidation (a powerful reaction between iron and hydrogen peroxide that breaks down stubborn organics), a second coagulation step using ferric chloride to pull out solids, and finally a dose of “green” nano zero-valent iron. These iron nanoparticles are made using black tea extract instead of harsh chemicals, so plant-based compounds help shape and protect tiny iron cores. The result is a highly reactive but more environmentally friendly material that can interact strongly with oxygen and dissolved pollutants.
What happens to steel, stainless steel, and copper
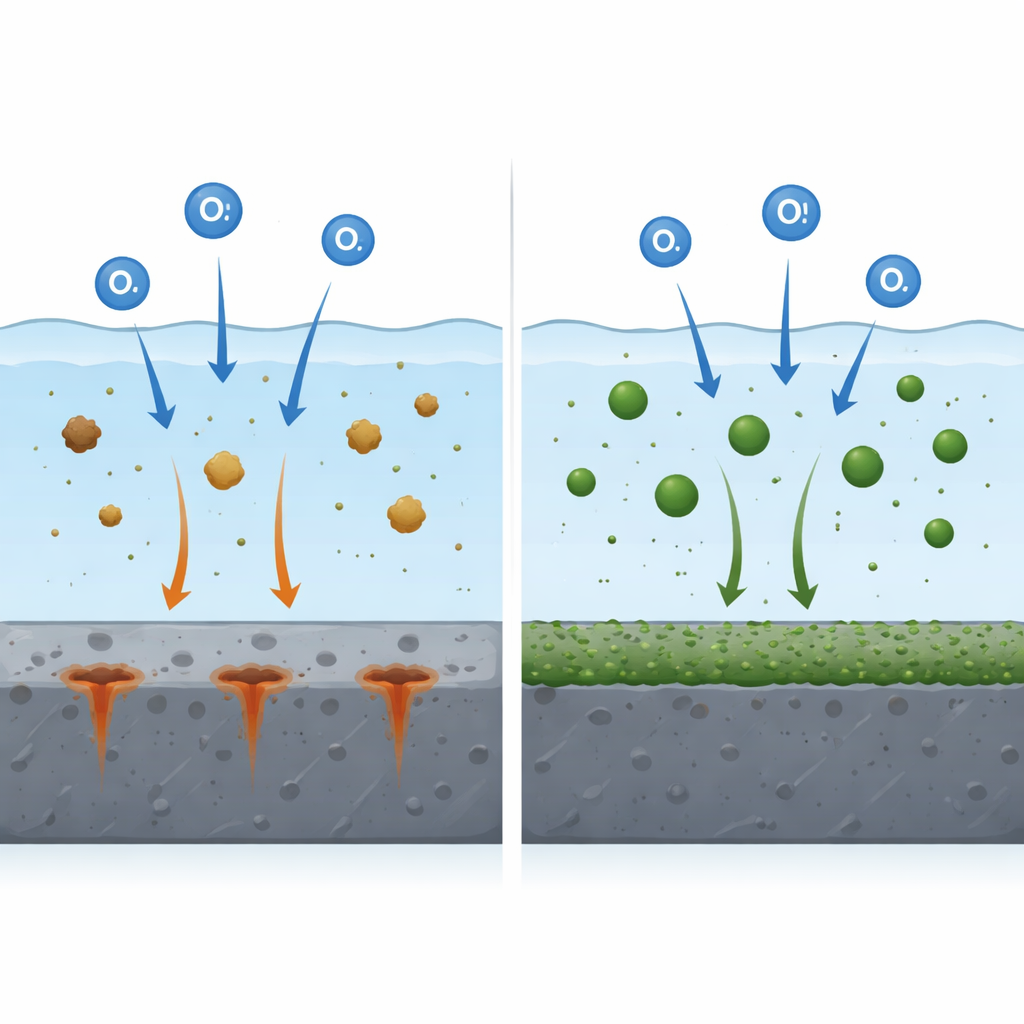
To find out how each water type affects real hardware, the team immersed samples of mild steel, stainless steel, and copper in the three waters and probed their behavior with sensitive electrochemical tools. For steel, the story is striking: basic factory treatment cuts corrosion by roughly 30 percent, but the advanced process with nano iron slashes it by about 86 percent. The measurements show that the treated water builds a tougher barrier at the steel surface and reduces the electrical pathways that drive rust. Stainless steel, which already relies on a thin passive film for protection, benefits only modestly from the advanced water and actually fares slightly worse in the factory-treated water, where the lower pH and leftover contaminants weaken its natural shield.
When cleaner water is not safer for every metal
Copper tells a more nuanced story. In untreated water, natural organic residues and higher phosphorus levels seem to form a thin protective film that modestly slows down copper dissolution. Both factory-treated and advanced-treated waters, however, disturb this balance. More sulfate and chloride ions and less protective phosphorus mean the films on copper become thinner and less stable, and the electrochemical tests reveal slightly faster attack. In other words, a treatment that is excellent for steel can quietly make conditions harsher for copper, an important warning for mixed-metal systems in real industrial plants.
From lab models to practical choices
To help plant operators make decisions, the authors also build simple mathematical models that link water properties such as acidity (pH), salt content, and phosphorus to the corrosion resistance of each metal. Although based on a small dataset, these models show clear trends: higher pH and the presence of nano iron greatly favor steel, while stainless steel and copper respond differently to shifts in pH and dissolved solids. Statistical tests confirm that the improvements for steel with the advanced treatment are not just random noise but robust, repeatable gains.
What this means for cleaner industry and water reuse
For a general reader, the takeaway is straightforward: by adding a carefully designed, tea-derived nano iron step to existing treatment, ceramic factories can turn a troublesome waste stream into water that is far less damaging to steel equipment and more suitable for reuse in industry or even agriculture. That means fewer leaks, longer-lived infrastructure, and less demand on scarce freshwater supplies. At the same time, the study highlights that “one size fits all” does not apply—copper components may need extra protection or different treatment recipes. Overall, the work shows how smart chemistry can make heavy industry both more economical and more environmentally responsible.
Citation: Khamis, E., Abd-El-Khalek, D.E., Hagar, M. et al. Sustainable treatment of ceramic manufacturing wastewater using combined advanced oxidation and coagulation/precipitation processes with green nano zero-valent iron: multi-metal corrosion monitoring. Sci Rep 16, 10491 (2026). https://doi.org/10.1038/s41598-026-42824-1
Keywords: ceramic wastewater, nano zero valent iron, metal corrosion, advanced oxidation, water reuse