Clear Sky Science · en
Proteomic analysis of tissue-derived extracellular vesicles shows region-specific molecular changes in a rat model of takotsubo syndrome
When Stress Leaves a Temporary Heartmark
Most people think of a heart attack as permanent damage, but there is a puzzling condition where the heart suddenly weakens and then largely recovers within days or weeks. This is called takotsubo syndrome, often triggered by intense emotional or physical stress. The new study summarized here looks inside tiny biological “message bubbles” released by heart cells in a rat model of this syndrome, revealing how different parts of the heart respond in their own distinct way to a sudden storm of stress hormones.
A Temporary Heart Failure with Lasting Questions
Takotsubo syndrome mimics a classic heart attack: patients arrive with chest pain, alarming electrocardiograms, and reduced pumping function. Yet their coronary arteries are not blocked, and the affected heart regions usually recover. A hallmark pattern is that the tip (apex) of the left ventricle becomes nearly still, while the base continues to contract. Despite its growing recognition, the biological “conversation” between stressed heart cells—how they signal distress, damage, and repair—remains poorly understood. The researchers used a well-established rat model that closely reproduces human takotsubo features after a carefully controlled dose of a stress‑like drug, capturing the heart 24 hours into the early recovery phase.
Tiny Bubbles Carrying Heart Messages
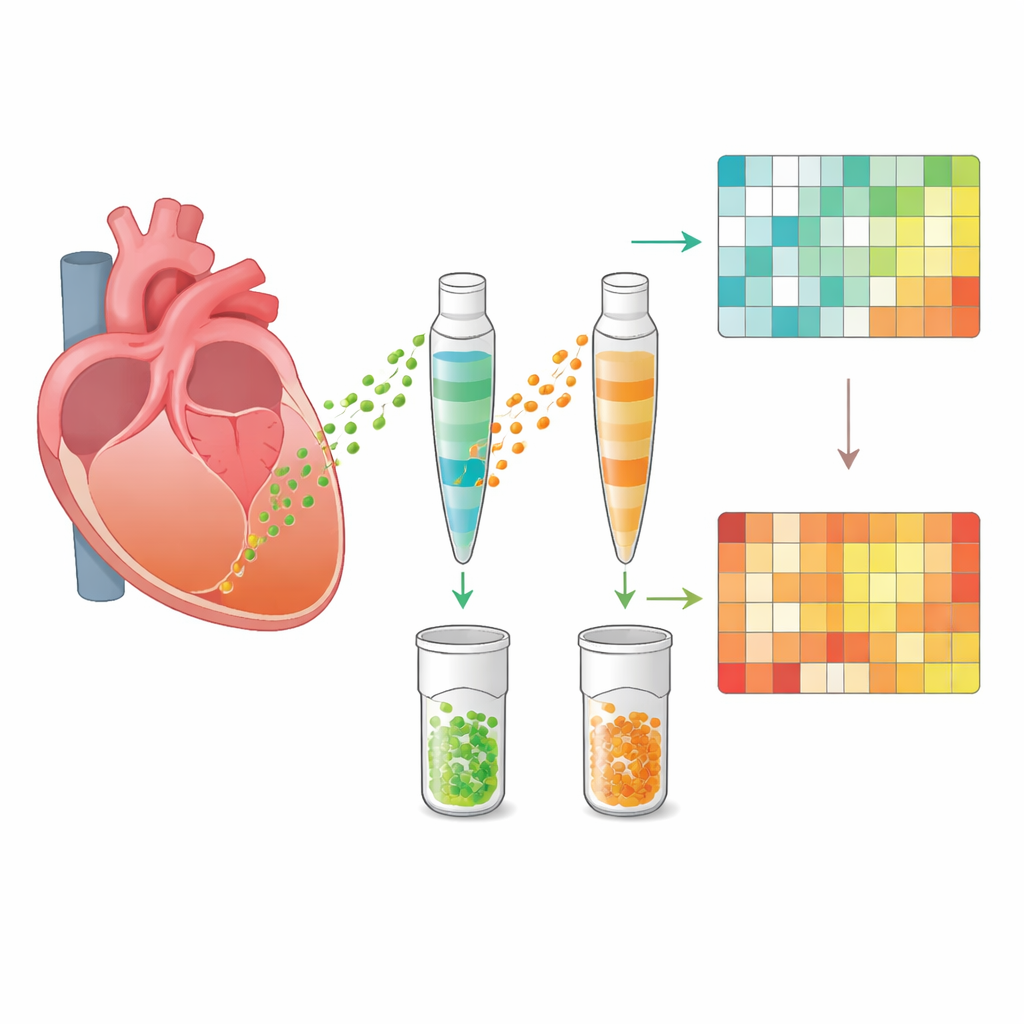
Every cell in the body releases nanoscale packages called extracellular vesicles. These membrane‑wrapped bubbles carry proteins, fats, and genetic material that can alter the behavior of neighboring or distant cells. In heart disease, such vesicles can either worsen damage by fueling inflammation or promote healing by supporting blood vessel growth and tissue repair. In this work, scientists carefully isolated vesicles directly from the apex and base of rat hearts, rather than from blood. Using electron microscopy, particle tracking, and protein markers, they confirmed they had clean preparations of typical vesicles, ranging from about 50 to 400 nanometers in size. Importantly, only the stressed apical region in takotsubo hearts produced fewer vesicles, while the unaffected base and normal hearts released similar numbers, hinting at a region‑specific response to stress.
Two Halves of the Heart, Two Molecular Stories
The team then applied high‑resolution proteomics, a technique that catalogues and quantifies thousands of proteins at once. Across all samples, they identified just over 2,000 proteins inside the vesicles. In healthy hearts, vesicles from apex and base looked remarkably similar, suggesting a uniform “baseline” communication profile. After stress, however, vesicles from the apical segment shifted dramatically, while those from the base barely changed. Hundreds of proteins were altered in the apex vesicles compared with both healthy apex and stressed base. Many of the most strongly increased proteins were linked to the immune system and inflammation, whereas many of the decreased proteins supported mitochondrial function—the cell’s energy factories. Advanced network and pathway analyses consistently pointed to an organized pattern: dialed‑down energy metabolism alongside heightened immune and tissue‑remodeling activity in the damaged segment.
Inflammation, Fats, and Repair Signals
Looking more closely at the interconnected protein networks, the researchers saw that proteins involved in lipid (fat) handling were tightly linked to those driving inflammation. This fits with earlier observations that fat droplets and swelling appear in the heart soon after stress, followed by immune‑cell infiltration and later scar‑like changes. Vesicles from the affected apex were enriched for proteins associated with blood clotting, blood vessel growth, and collagen formation—features of both repair and scarring. At the same time, they carried proteins belonging to key survival signaling routes, especially the MAPK pathways, which help cells withstand injury and may support the heart’s ability to recover its pumping strength. Altogether, the vesicle cargo painted a picture of a heart region undergoing an energy crunch, active inflammation, structural remodeling, and attempts at self‑preservation.
What This Means for Patients and Future Care
This study is the first to map in detail the protein cargo of vesicles released from different regions of the heart in takotsubo syndrome. To a non‑specialist, the central message is that the stunned tip of the heart is not quietly waiting to recover; it is buzzing with molecular activity, sending out tiny packages that signal energy stress, inflammation, tissue repair, and survival. These vesicles may one day serve as sensitive blood‑based markers to diagnose takotsubo, distinguish it from a heart attack, or identify patients at risk of slower recovery. They may also highlight new treatment targets—pathways that could be tuned to reduce harmful inflammation or support healthy repair. While the work was done in male rats at a single time point and remains exploratory, it opens a new window on how the stressed heart talks to itself during this mysterious, usually reversible, form of heart failure.
Citation: Zulfaj, E., Nejat, A., Kalani, M. et al. Proteomic analysis of tissue-derived extracellular vesicles shows region-specific molecular changes in a rat model of takotsubo syndrome. Sci Rep 16, 8731 (2026). https://doi.org/10.1038/s41598-026-42812-5
Keywords: takotsubo syndrome, extracellular vesicles, cardiac stress, proteomics, heart inflammation