Clear Sky Science · en
Feasibility study on enhancing the biodegradability of fresh and old landfill leachate using combined chemical precipitation and Fenton processes
Why the Water Under Landfills Matters
Every landfill leaks. As rainwater trickles through piles of garbage, it picks up a cocktail of dissolved chemicals, metals, and nitrogen compounds, forming a dark liquid called leachate. If this leachate is not treated properly, it can seep into rivers and groundwater, threatening drinking water and ecosystems. The study behind this article asks a practical question: can we turn this stubborn, highly polluted liquid into something that nature’s own microbes can finish cleaning up more easily?
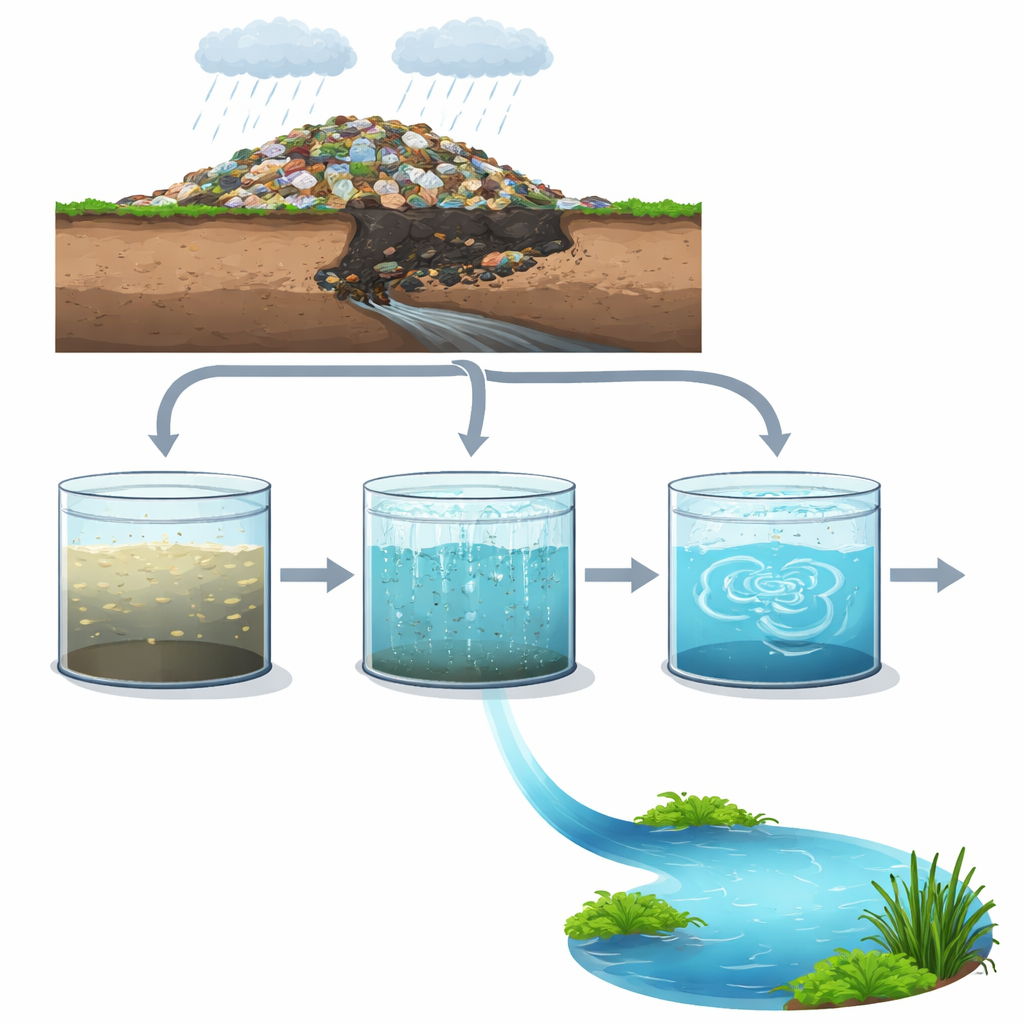
The Hidden Liquid Beneath Our Trash
Leachate is more than just dirty water. It can contain heavy metals like lead and arsenic, high levels of ammonia, and long‑lasting organic substances that do not break down easily. Its makeup changes as a landfill ages. In “fresh” sections, less than about five years old, the leachate is rich in easily digestible organic matter, so biological treatment can work fairly well. In “old” sections, more than ten years old, the easy food is gone and what remains is a mix of tough, complex molecules such as humic and fulvic acids, along with rising levels of ammonia. These older leachates are harder and more expensive to treat, and they can overwhelm conventional biological systems.
A Three‑Step Cleanup Train
The researchers focused on the Aradkooh landfill complex near Tehran, which has both young and old waste zones, allowing a direct comparison of fresh and old leachate under the same climate and geology. They tested a treatment train with three main steps. First, they added lime, a simple, inexpensive powder, to raise the pH and cause dissolved metals to form solid particles that can settle out. Second, they stripped ammonia by making the water strongly alkaline and bubbling air through it, driving ammonia from the liquid into the gas phase where it can be captured. Third, they used a chemical method called the Fenton process, where hydrogen peroxide and iron react to generate highly reactive hydroxyl radicals that attack and break apart stubborn organic molecules.
Making Tough Pollution Easier for Microbes
The team judged success by watching how the balance between “food” and total organic load changed, using a common index that compares how much oxygen microbes would need to eat the material to how much is present overall. Higher values mean the water is more biodegradable. Lime treatment alone removed a great deal of heavy metals and cut ammonia by about 93 percent after the stripping step, and it slightly improved this biodegradability index in both fresh and old leachate. The real transformation came after the Fenton stage. By carefully tuning how much hydrogen peroxide and iron they used, and how long they let the reaction run, the researchers were able to more than double this index. In fresh leachate it rose from about 0.29 to 0.67, and in old leachate from about 0.23 to 0.73, pushing both into a range where biological treatment becomes far more effective.
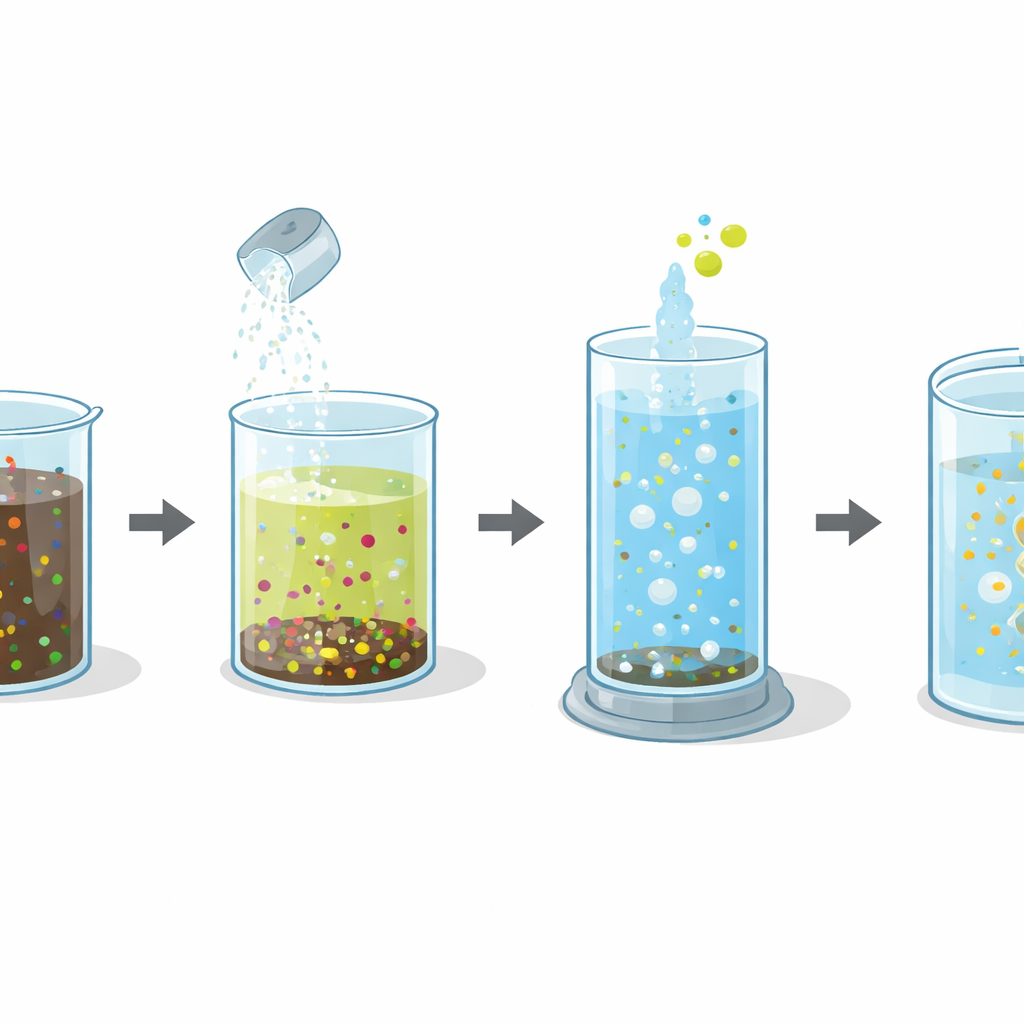
Fine‑Tuning a Powerful Chemical Tool
Because the Fenton process can either help or hinder treatment depending on how it is run, the researchers used statistical design tools to map out how three factors worked together: the ratio of hydrogen peroxide to iron, the total dose of peroxide, and the reaction time. They found that too little peroxide or iron produced not enough reactive radicals, while too much led to wasteful side reactions that actually consumed these radicals. The sweet spot differed slightly between fresh and old leachate, but in both cases a moderate peroxide‑to‑iron ratio, near‑acidic conditions, and reaction times around one hour to a little over an hour produced the best balance of pollutant removal and improved biodegradability.
From Lab Bench to Real‑World Landfills
In the end, the combined lime and Fenton approach removed more than 80 percent of the overall organic load from both fresh and old leachate, sharply reduced toxic metals and ammonia, and turned a very stubborn wastewater into one that biological systems can handle much more readily. For a layperson, the key message is that a relatively simple sequence of chemical steps can make the dangerous liquid seeping from landfills far safer and easier to clean. Before such methods are widely deployed, the authors note that engineers will need to test them in pilot‑scale systems, manage the extra sludge and chemical use, and optimize costs. But this work shows a promising path toward cleaner water and safer landfills, even decades after the trash is buried.
Citation: Rasolevandi, T., Naddafi, K., Hassanvand, M.S. et al. Feasibility study on enhancing the biodegradability of fresh and old landfill leachate using combined chemical precipitation and Fenton processes. Sci Rep 16, 14154 (2026). https://doi.org/10.1038/s41598-026-42622-9
Keywords: landfill leachate, wastewater treatment, advanced oxidation, environmental pollution, water quality