Clear Sky Science · en
The influence of multi-solvent systems on the electrospinning process
Why tiny fibers and simple liquids matter
From breathable face masks to smart bandages and food packaging, many modern materials rely on mats of extremely thin plastic fibers. One of the most versatile ways to make these nanofibers is a technique called electrospinning, which pulls strands from a liquid using high voltage. This paper asks a deceptively simple question with big practical consequences: how does the choice and mixing of everyday laboratory solvents—the liquids used to dissolve the plastic—control whether neat, useful fibers are formed or the liquid suddenly turns into a useless gel?
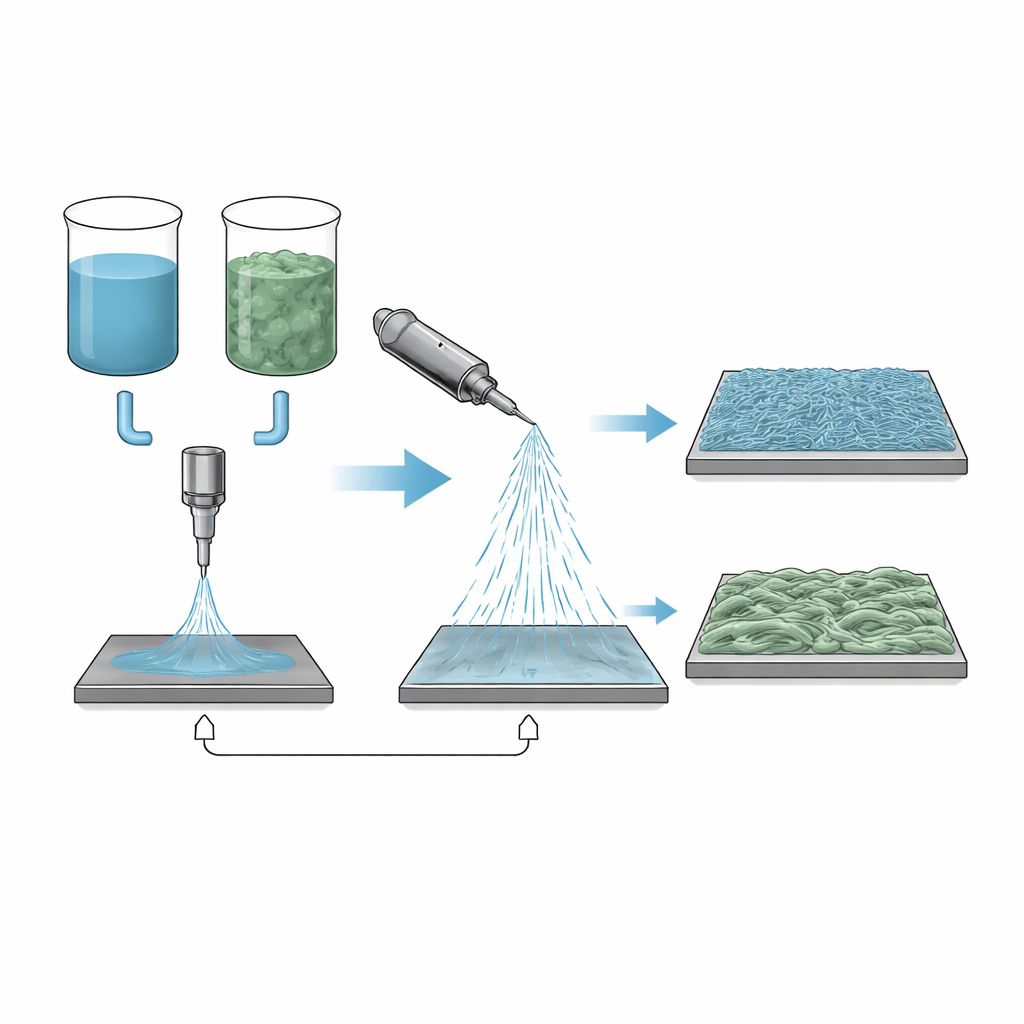
Spinning webs from charged liquid
Electrospinning starts with a polymer dissolved in a solvent and fed through a small nozzle. When a high voltage is applied, a thin stream shoots toward a collector plate and solidifies into a web of fibers thinner than a human hair. The beauty of the method is that the fiber diameter, smoothness and porosity can be tuned for different jobs, from drug-carrying dressings to air and water filters. But the process is extremely sensitive to the liquid’s properties: its thickness, surface tension, and ability to conduct electricity all depend on which solvent, or combination of solvents, is used. In this study the authors focus on a biodegradable plastic, polybutylene succinate (PBS), and explore what happens when it is dissolved in chloroform together with a second, higher-boiling liquid.
When a clear liquid suddenly turns into jelly
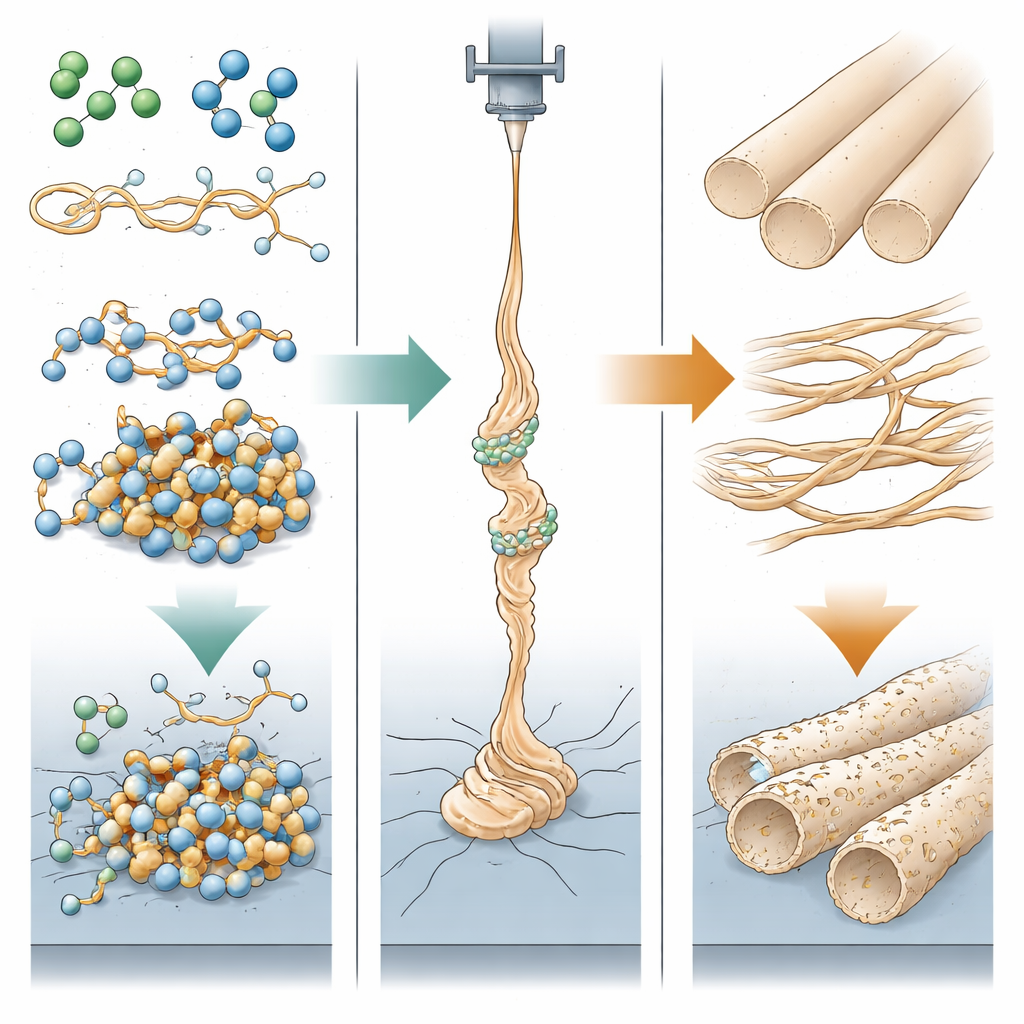
The team systematically mixed chloroform with three common organic solvents—dimethylformamide (DMF), dimethyl sulfoxide (DMSO) and d-limonene—and dissolved two commercial PBS grades in them. They observed that many of these two-solvent mixtures, instead of staying clear, slowly turned cloudy and finally into a jelly-like or grease-like mass, especially when DMF or DMSO were present. By carefully heating and cooling the solutions and tracking when they switched between liquid and gel, the researchers mapped transition temperatures and estimated the energy barrier for this change. Their analysis of solvent molecular shapes and charge distributions suggests that highly polar DMF and DMSO strongly associate both with each other and with specific sites on the PBS chains. These contacts effectively lock parts of the chain together, reducing mobility and nudging the whole mixture toward local precipitation and gelation.
Small differences in plastic, big differences in behavior
Interestingly, the two PBS products, although almost identical in molecular weight, responded quite differently. One grade (BioPBS FD 92) gelled only when the polar co-solvent content was relatively high, while the other (PBE 003) began to turn cloudy with much smaller additions. The authors link this contrast to subtle differences in density, chain architecture and factory-added processing aids, which change how easily solvent molecules can tuck themselves between polymer segments. Viscosity measurements showed that BioPBS solutions were thicker, which helped stabilize the charged jet during electrospinning, leading to more uniform fibers. In contrast, the lower-viscosity PBE 003 solutions were more prone to instabilities, producing fibers with a wider spread in diameter and more spindle-like defects, especially as gelation progressed over time.
From smooth strands to porous and unstable fibers
Using electron microscopes, the researchers compared the fibers produced from each solvent combination. With DMF as the second solvent, they obtained smooth, continuous fibers. When DMSO was used instead, the fibers developed a porous surface. The authors attribute this to a steam-induced phase separation: highly water-loving DMSO absorbs moisture from the surrounding air, causing the dissolved PBS to precipitate at the fiber surface before the solvents fully evaporate, leaving behind tiny voids. For mixtures that gelled quickly, the useful processing window shrank to about half an hour. Beyond that, the jet broke, fiber diameters became erratic and thick spindles appeared, showing how time-dependent gelation directly undermines product quality and scalability.
What this means for greener fiber materials
In everyday terms, this work shows that dissolving a plastic in “just any” blend of compatible liquids is not enough if one wants reliable, finely tuned nanofibers. The exact pairing and mixing ratio of solvents can quietly steer the liquid from a friendly, spinnable state into a stiff jelly that clogs the process, or into a solution that forms either smooth or porous fibers. For biodegradable PBS, polar partners such as DMF and DMSO offer useful control over fiber size and texture but can also trigger gelation unless their content and temperature are carefully managed. By identifying these hidden interactions and their impact on processing time, this study provides a practical roadmap for designing safer, greener nanofiber products for medicine, packaging and filtration without unpleasant surprises in the spinning line.
Citation: Borowczak, M., Sobczyk, K. & Leluk, K. The influence of multi-solvent systems on the electrospinning process. Sci Rep 16, 8666 (2026). https://doi.org/10.1038/s41598-026-42574-0
Keywords: electrospinning, nanofibers, biodegradable polymers, solvent mixtures, gelation