Clear Sky Science · en
Bandgap engineering and enhanced charge separation in Zn-modified BiW11 polyoxometalate for azo dye photodegradation
Why cleaning up colored water matters
From bright clothing to vivid food packaging, synthetic dyes are part of daily life—but when they end up in rivers and lakes, they can be toxic and hard to remove. Many conventional treatments simply move the problem around, trapping dyes on filters or powders that then become waste themselves. This study explores a different approach: using light and a specially designed inorganic material to break dye molecules apart, turning them into more harmless substances instead of just capturing them.
A light-powered way to destroy stubborn dyes
The researchers focused on two common “azo dyes,” Congo Red and Phenol Red, which are used widely in textiles and laboratories and are known for being persistent in water. They investigated a family of metal-oxygen clusters called polyoxometalates, which behave somewhat like tiny, inorganic sponges for light and electrons. A particular cluster built from bismuth and tungsten, known as BiW11, was chosen as the base material because it is stable and already known to catalyze chemical reactions under light.
To make this base material more effective, the team modified it by introducing zinc ions, creating a new form called Zn–BiW11. The idea was that carefully adding zinc would subtly change how the material absorbs light and handles electrical charge without breaking its underlying structure. When exposed to ultraviolet (UV) light, these clusters can promote reactions that attack dye molecules, potentially breaking them down into smaller, less harmful pieces.

Tuning a tiny structure to capture more light
The scientists first verified how zinc changed the material’s structure and behavior. Using techniques that probe vibrations of chemical bonds and the arrangement of atoms, they showed that the characteristic framework of BiW11 remained intact after zinc was added. However, small shifts in the signals revealed that zinc was successfully integrated into the cluster, subtly distorting its atomic environment. Microscopy images showed that the zinc-modified particles formed rough, nanometer-scale grains with a high density of surface sites where reactions can occur.
Most importantly for light-driven chemistry, measurements of how the materials absorb ultraviolet light showed that Zn–BiW11 has a slightly smaller “band gap” than the original BiW11. In simple terms, this means the modified cluster needs a bit less energy to kick electrons into an excited state, allowing it to harvest UV light more efficiently. With more excited electrons and matching positive “holes” available, the material is better equipped to drive the chemical steps that break apart dye molecules.
Putting the new material to the test
The team then tested how well both versions of the catalyst could clean water containing Congo Red or Phenol Red when illuminated with UV light. In carefully controlled experiments, the dye solutions were mixed with either BiW11 or Zn–BiW11 and exposed to a handheld UV lamp. Over several hours, the researchers tracked how the characteristic color of each dye faded as the molecules were destroyed. While both materials accelerated dye breakdown compared with UV light alone, the zinc-modified version clearly performed better.
For Congo Red, Zn–BiW11 removed about two-thirds of the dye, compared with roughly half for the unmodified material. For Phenol Red, the improvement was even more striking: the zinc-doped catalyst eliminated more than three-quarters of the dye. The data followed a pattern known as pseudo–first-order behavior, which is typical of many catalytic reactions and allowed the team to extract rate constants that quantify how quickly the dyes were degraded. In every case, the zinc-modified material acted faster than its parent.
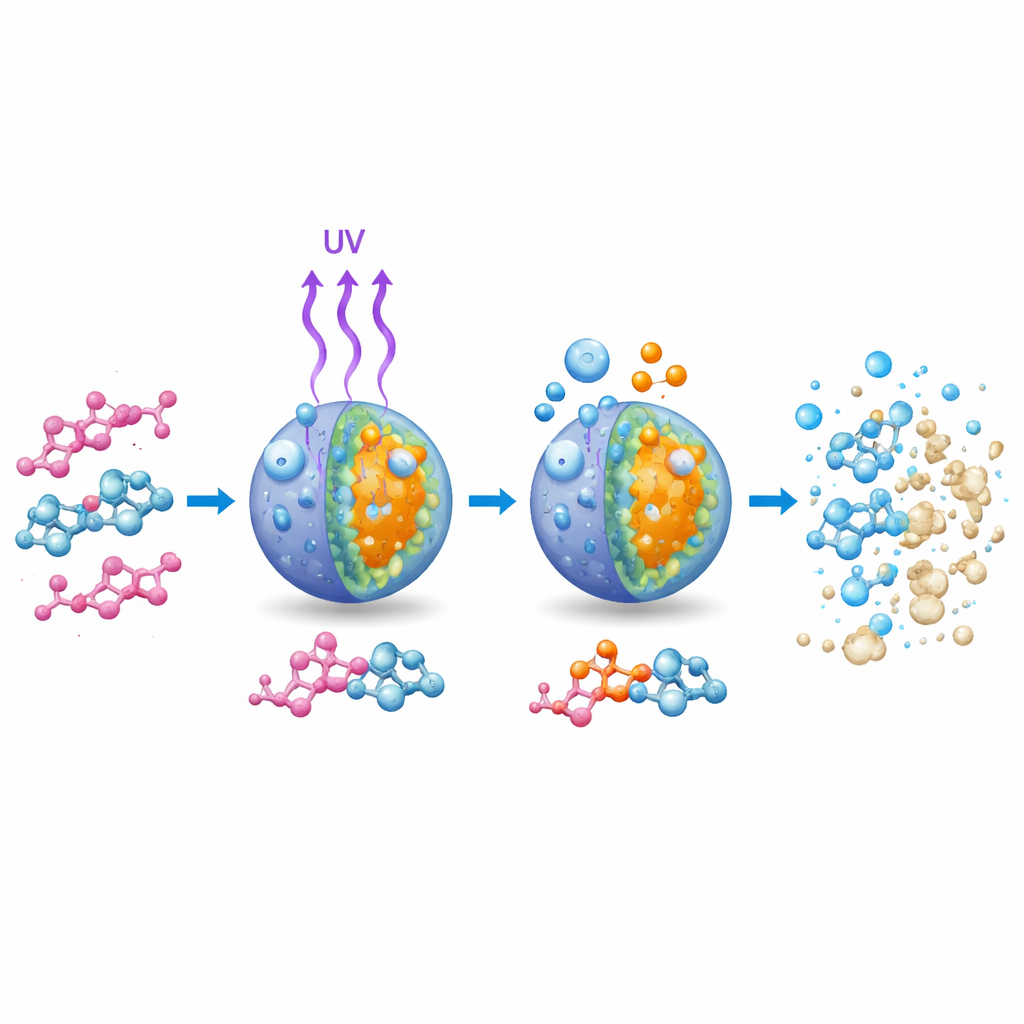
How light, oxygen, and the catalyst work together
The authors propose a step-by-step picture of what happens during this process. When UV light strikes the catalyst, electrons in the material are promoted to a higher-energy state, leaving behind positively charged holes. Rather than immediately recombining and wasting their energy as heat, these charges migrate to the surface, where they interact with water and dissolved oxygen. The excited electrons help convert oxygen into highly reactive superoxide species, while the holes help generate powerful hydroxyl radicals. These short-lived particles attack the dye molecules, especially their long, color-giving chains, progressively chopping them into smaller, less colored fragments and, in ideal cases, into carbon dioxide and water.
What this means for cleaner water
In everyday terms, this work shows that by subtly redesigning a light-sensitive inorganic cluster with zinc, it is possible to make a more effective “sun-powered cleaner” for stubborn dye pollutants. The zinc-modified material absorbs UV light more efficiently and keeps the generated electrical charges separated long enough to create aggressive chemical species that can dismantle dye molecules. While the study was carried out under laboratory UV lamps and with model dyes, it points toward engineered polyoxometalate catalysts as promising tools for treating dye-contaminated wastewater in a way that does not simply shift pollution from one place to another, but actively destroys it at the molecular level.
Citation: Bani-Atta, S.A., Alatawi, N.M., El-Zaidia, E.F.M. et al. Bandgap engineering and enhanced charge separation in Zn-modified BiW11 polyoxometalate for azo dye photodegradation. Sci Rep 16, 13679 (2026). https://doi.org/10.1038/s41598-026-42532-w
Keywords: photocatalytic dye degradation, wastewater treatment, polyoxometalates, zinc doping, azo dyes