Clear Sky Science · en
Environmental remediation and wastewater treatment using silver incorporated dual metal oxide polyaniline nano composites kinetic insights into Pb (II) and As (III) adsorptive removal
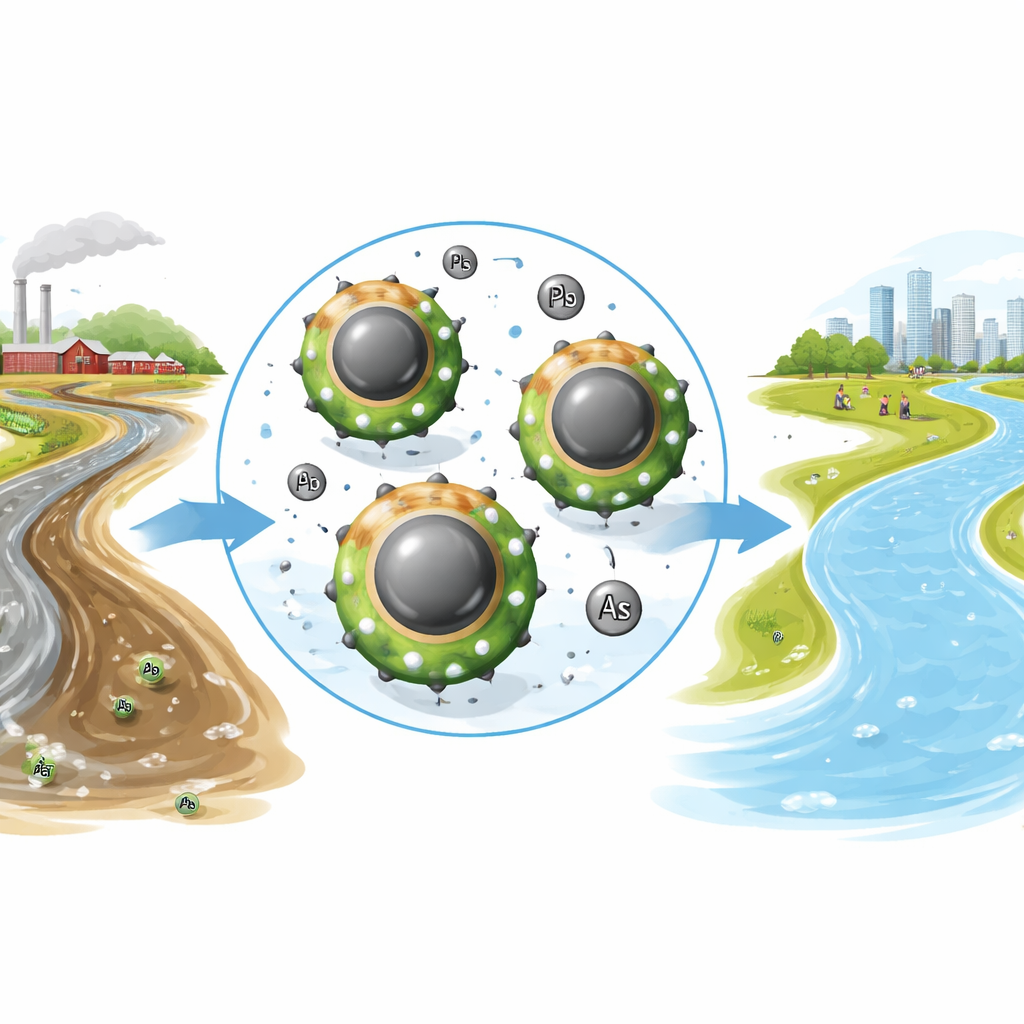
Why cleaning toxic metals from water matters
Lead and arsenic are invisible poisons that can seep into rivers, lakes, and even tap water from factories, mines, farms, and old pipes. Over time they damage the brain, kidneys, liver, lungs, and the heart, and are linked to cancers and developmental problems in children. Because these metals do not break down naturally, scientists are searching for materials that can pull them out of water quickly, cheaply, and safely. This study introduces a new tiny, magnet-friendly material that acts like a reusable sponge for lead and arsenic in polluted water.
A tiny sponge built from several familiar ingredients
The researchers designed a nanoscale composite material by combining several components, each chosen for a particular job. At its heart is magnetite, an iron oxide that makes the particles respond to a magnet, so they can later be pulled back out of the water. Around this core they added copper oxide, which provides many reactive spots where metal pollutants can latch on. They then coated this with polyaniline, a conducting plastic known for its stability and low cost, and used itaconic acid, a harmless, plant-derived powder, to help glue the layers together and provide extra binding groups. Finally, they decorated the surface with silver nanoparticles made using an extract from Piper betel leaves, a green, plant-based method that avoids harsh chemicals and also adds catalytic and antimicrobial properties.
How the new material was made and examined
To build the composite, the team first boiled Piper betel leaves in water to obtain a natural extract that reduces dissolved silver salts to tiny metallic silver particles. They then carried out a stepwise process in water to form the iron and copper oxides, grow the polyaniline network with itaconic acid, and attach the silver. The resulting particles, called Fe₃O₄/CuO-IA/PANI@Ag nanocomposites, are only tens of nanometers across—thousands of times smaller than the width of a human hair. Using powerful tools such as X-ray diffraction, electron microscopes, and infrared spectroscopy, the scientists confirmed that the expected crystal structures were present, the surface was rough and porous, and all the elements—iron, copper, nitrogen from the polymer, oxygen, and silver—were uniformly distributed. This architecture creates a high surface area and many chemical groups that can grab onto metal ions in water.
How well it traps lead and arsenic from water
The team then tested how effectively these particles could remove lead (Pb(II)) and arsenic (As(III)) from water under different conditions that mimic real treatment scenarios. They found that the water’s acidity (pH), the amount of material added, contact time, and temperature all influence performance. Removal increased as the pH rose from strongly acidic toward mildly acidic to near-neutral, peaking around pH 6, where the polymer and acid groups on the surface are best positioned to attract metal ions. Increasing the amount of nanocomposite provided more active sites, boosting removal until a practical optimum was reached. Most of the metal uptake occurred within about 40 minutes, after which the surface approached saturation. Moderate warming of the solution also improved removal before very high temperatures began to reduce efficiency.
What the numbers say about capacity and behavior
By fitting the experimental data to standard models used in water treatment, the researchers showed that the composite behaves like a surface that forms a single, orderly layer of captured metal ions. Under these conditions, the maximum amounts that could be held were about 48 milligrams of lead and 61 milligrams of arsenic per gram of material, competitive with or better than many similar sorbents reported in the literature. Analyses of how quickly metals were taken up indicated that the process is controlled by chemical bonding and complex formation on the surface rather than just simple physical sticking. The presence of multiple functional groups from the polymer and itaconic acid, together with the iron and copper oxides and silver, creates several ways for arsenic and lead to bind, explaining the strong affinity, particularly for arsenic.
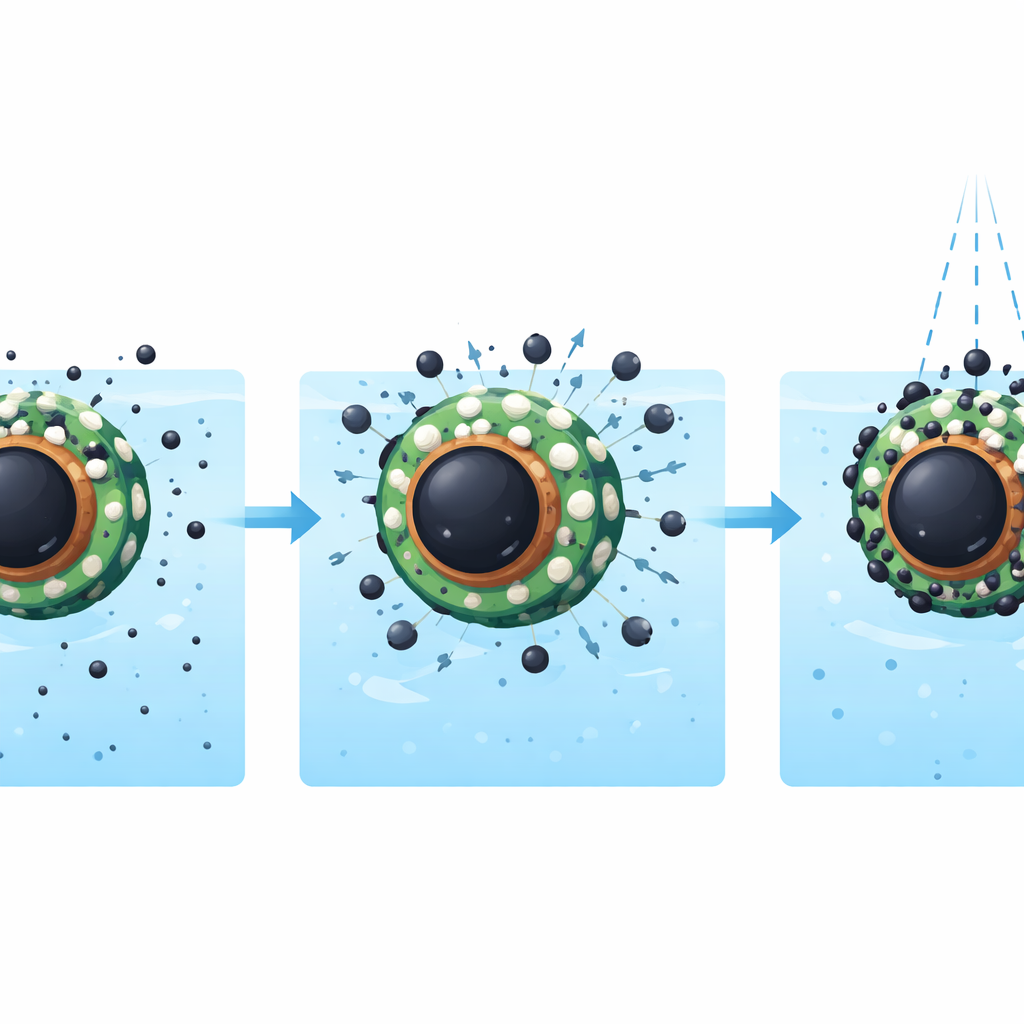
Reusability and promise for real-world cleanup
For any treatment material to be practical, it must be recoverable and reusable. Thanks to the magnetic core, these particles can be pulled out of treated water with a magnet and then washed in a mildly acidic solution to release the trapped metals. In repeated use tests, the composite maintained around 90 percent of its original performance over five cycles, suggesting that it is robust and economical. Overall, the study demonstrates that a carefully engineered, plant-assisted nanocomposite can reliably strip dangerous amounts of lead and arsenic from water and be used again and again. For non-specialists, the takeaway is that smartly designed, tiny, magnet-friendly sponges like these could play a significant role in making drinking water safer and cleaning up contaminated industrial wastewater.
Citation: Parthiban, E., Sudarsan, S., Allasi, H.L. et al. Environmental remediation and wastewater treatment using silver incorporated dual metal oxide polyaniline nano composites kinetic insights into Pb (II) and As (III) adsorptive removal. Sci Rep 16, 14056 (2026). https://doi.org/10.1038/s41598-026-42318-0
Keywords: wastewater treatment, heavy metal removal, nanocomposite adsorbent, lead and arsenic, magnetic nanoparticles