Clear Sky Science · en
Alkali metal-induced modifications in the electronic and NLO properties of triselenasumanene complexes through DFT study
Light, Electrons, and Smarter Optics
Modern technologies like high-speed internet, precise laser surgery, and advanced displays all rely on materials that can bend and reshape light in sophisticated ways. This study explores how a specially designed, bowl-shaped carbon-and-selenium molecule can be made far more responsive to light simply by attaching tiny amounts of familiar metals such as lithium, sodium, and potassium. By using computer simulations instead of lab trial-and-error, the authors show how a subtle rearrangement of electrons can turn an already promising molecule into a powerful building block for future optical and photonic devices.
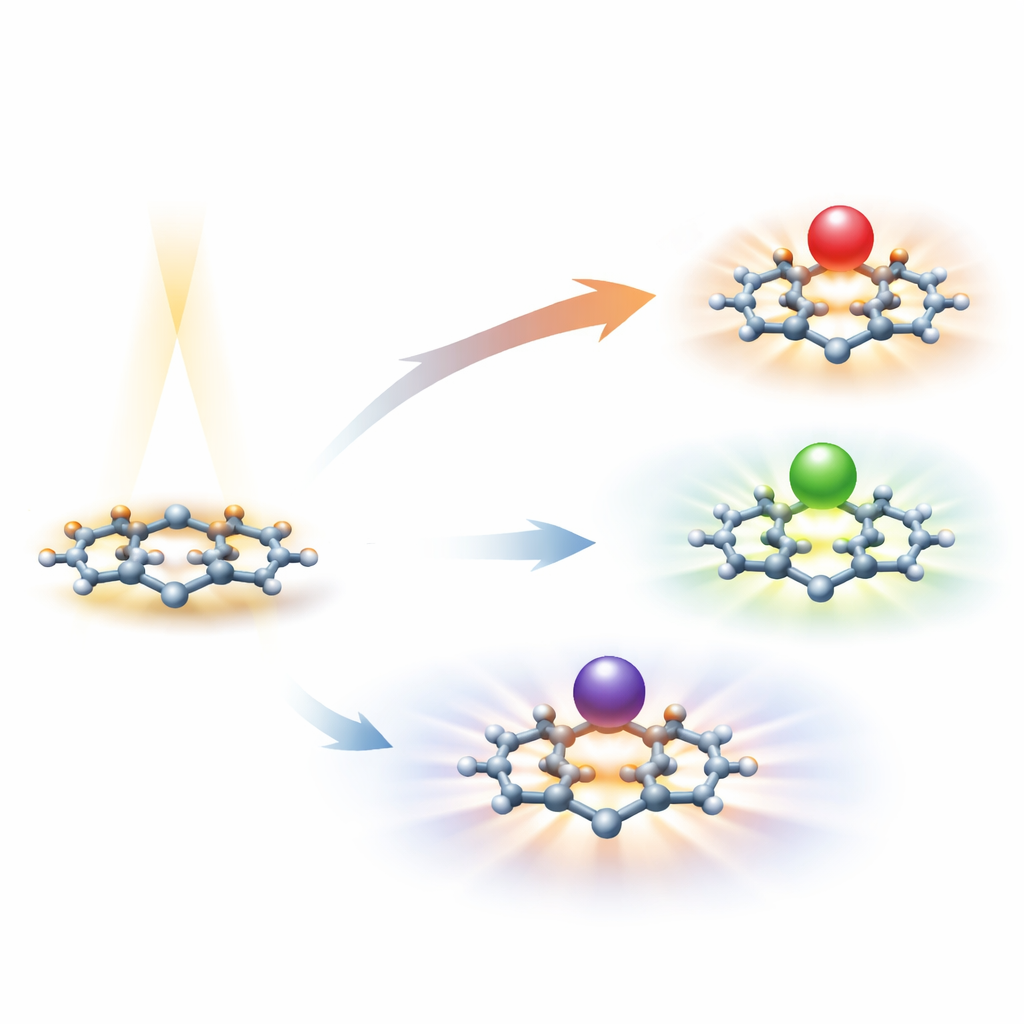
A Designer Molecule Built for Moving Charge
At the heart of the work is a molecule called triselenasumanene, or TSSUM. It is derived from a “buckybowl” framework: a curved, disk-like arrangement of carbon atoms, here embellished with three selenium atoms. This structure gives TSSUM a stable backbone and an extended cloud of electrons that can move across the molecule. Selenium, being heavier and more easily polarizable than many common elements, helps electrons respond strongly to electric fields and light. TSSUM is already known as a good candidate for transporting charges in solar cells, but on its own it shows only a modest ability to twist and reshape light—an ability scientists call the nonlinear optical response.
Using Everyday Metals as Electron Boosters
The researchers asked what would happen if they brought alkali metals—lithium, sodium, and potassium—close to the inner, concave side of the TSSUM bowl. These metals are well known for giving up or rearranging their outer electrons easily. Detailed quantum-mechanical calculations show that each metal settles in a stable spot hovering over the center of the bowl, with lithium sitting closest and potassium farthest away. In a realistic liquid-like environment, the metal–molecule combinations are even more stable than in empty space, because the surrounding medium helps balance and spread out the resulting charge rearrangements.
How Electron Shifts Change the Energy Landscape
Attaching the metals dramatically reshapes the electronic landscape of TSSUM. In the original molecule, there is a sizeable energy gap between the highest occupied and lowest available electron states, which limits how easily electrons can be excited by light. When lithium, sodium, or potassium is added, new electronic levels appear in this gap. The gap shrinks to roughly a quarter of its original size, which means electrons can move more freely when light hits the material. The simulations also reveal clear pathways for charge to flow between the selenium-rich regions of the bowl and the metal atom, confirming that electrons are being shifted and partially shared. This reorganization of charge is precisely what strengthens the molecule’s interaction with light.

From Modest Response to Strong Light Shaping
These electronic changes have a striking effect on how the complexes interact with light across the spectrum. Pure TSSUM mainly absorbs ultraviolet light around 320 nanometers. Once a metal is added, new absorption bands appear at much longer wavelengths that reach into the visible region—around 614 to 704 nanometers—showing that lower-energy light can now trigger electronic motion. Most importantly, a key measure of nonlinear optical strength, called hyperpolarizability, jumps by several hundred to more than a thousand times compared with the original molecule. In solution, where the environment further stabilizes separated charges, these values become even larger. Although sodium gives the strongest effect in empty space and lithium in solution, all three metals turn TSSUM into a far more active light-shaping unit.
Why This Matters for Future Light-Based Devices
In plain terms, the study demonstrates that carefully placing a single metal atom above a curved carbon–selenium framework can transform an ordinary optical molecule into an exceptionally sensitive one. By shrinking the energy gap and encouraging charge to slosh back and forth between the metal and the bowl-shaped scaffold, the material becomes much better at responding in a nonlinear way when light passes through it. Such metal–TSSUM combinations could serve as tunable ingredients in next-generation optoelectronic and photonic components, from faster optical switches to more efficient light-based sensors, where controlling the flow and transformation of light at the molecular level is key.
Citation: Rafee, V., Kamalinahad, S. Alkali metal-induced modifications in the electronic and NLO properties of triselenasumanene complexes through DFT study. Sci Rep 16, 10718 (2026). https://doi.org/10.1038/s41598-026-42303-7
Keywords: nonlinear optics, alkali metal complexes, molecular photonics, charge transfer, optoelectronic materials