Clear Sky Science · en
A physics-informed machine learning framework for predicting and mitigating doxorubicin nanocarrier toxicity in normal cells
Why safer cancer drugs matter
Chemotherapy drugs like doxorubicin are powerful weapons against cancer, but they can also harm healthy tissues, especially the heart. Researchers have tried packaging these drugs inside tiny particles called nanocarriers to steer more medicine to tumors and less to the rest of the body. Yet tuning the size, coating, and drug load of these particles has largely been a slow trial‑and‑error process. This study shows how blending physics with artificial intelligence can turn that guesswork into a more precise, predictive science—helping design drug carriers that attack cancer while sparing normal cells.
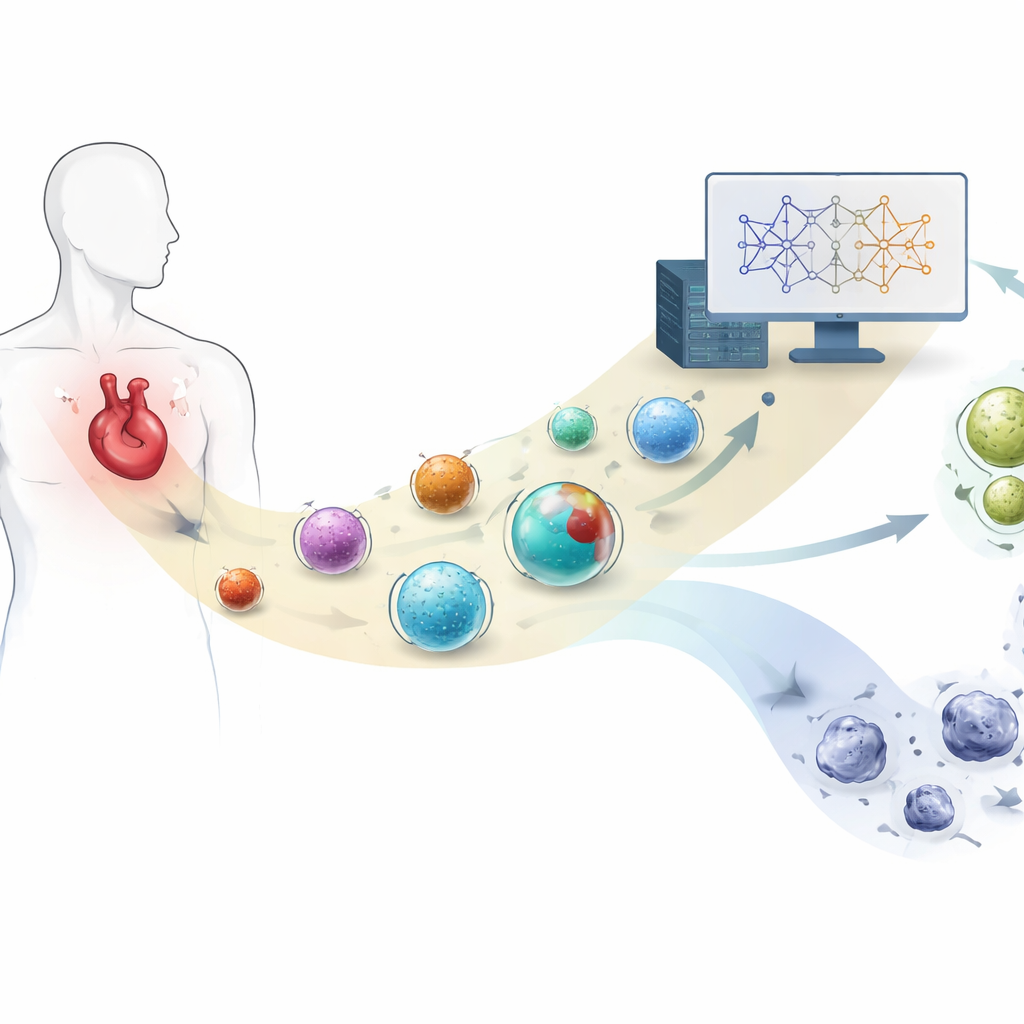
Tiny carriers with big trade‑offs
Doxorubicin has long been a mainstay in treating cancers such as breast tumors and leukemias, but its benefits come at the cost of serious side effects, including irreversible heart damage. Encapsulating the drug in nanosized particles offers a way to change where and how it moves in the body. By adjusting features like particle size, surface charge, and how much drug is packed inside, scientists hope to concentrate doxorubicin in tumors and reduce its exposure to healthy organs. However, these design choices interact in complex and non‑obvious ways, making it hard to know which combination will keep normal cells safe without endless lab experiments.
Turning scattered studies into a single map
The authors gathered detailed information from 77 different doxorubicin‑loaded nanocarrier systems reported in the scientific literature. These formulations spanned many materials—polymer particles, liposomes, inorganic nanoparticles, and more—and had been tested on a variety of healthy cell types, from heart and blood‑vessel cells to skin and lung cells. Because the original studies used different toxicity tests and reporting styles, the team carefully converted all results into a common scale of “normal cell viability,” ranging from 0 to 100 percent survival at comparable drug doses. They also standardized key physical properties such as particle size, surface charge (zeta potential), and measures of how efficiently the drug was loaded and trapped inside each carrier.
Adding physical rules to machine learning
With this harmonized dataset, the researchers trained several machine‑learning models to predict how toxic a given nanocarrier design would be to normal cells. They compared common approaches like random forests, gradient‑boosted trees, and standard neural networks with a more advanced method known as a physics‑informed neural network. In this framework, the model does not just fit past data; it is gently nudged to obey basic physical principles that govern how particles release drug and stay stable in liquid. Equations describing diffusion‑driven drug release, size‑dependent motion, and charge‑based stability were woven into the learning process as soft constraints, guiding the model away from predictions that would violate well‑established science.
Finding the sweet spot in particle design
The physics‑informed model proved to be the most accurate and reliable, capturing nearly 90 percent of the variation in normal cell survival and keeping prediction errors to within a few percentage points. Tools that explain model decisions revealed that two properties dominate safety: particle size and surface charge. Carriers that were moderately sized—roughly 120 to 150 nanometers across—and carried a moderately negative charge tended to be gentler on healthy cells. In contrast, very small or strongly charged particles were more likely to cling to unintended tissues or shed their cargo too quickly, raising toxicity. The model also identified a safe range for drug loading: enough doxorubicin to be useful, but not so much that the carrier becomes unstable or releases a sudden burst of drug.
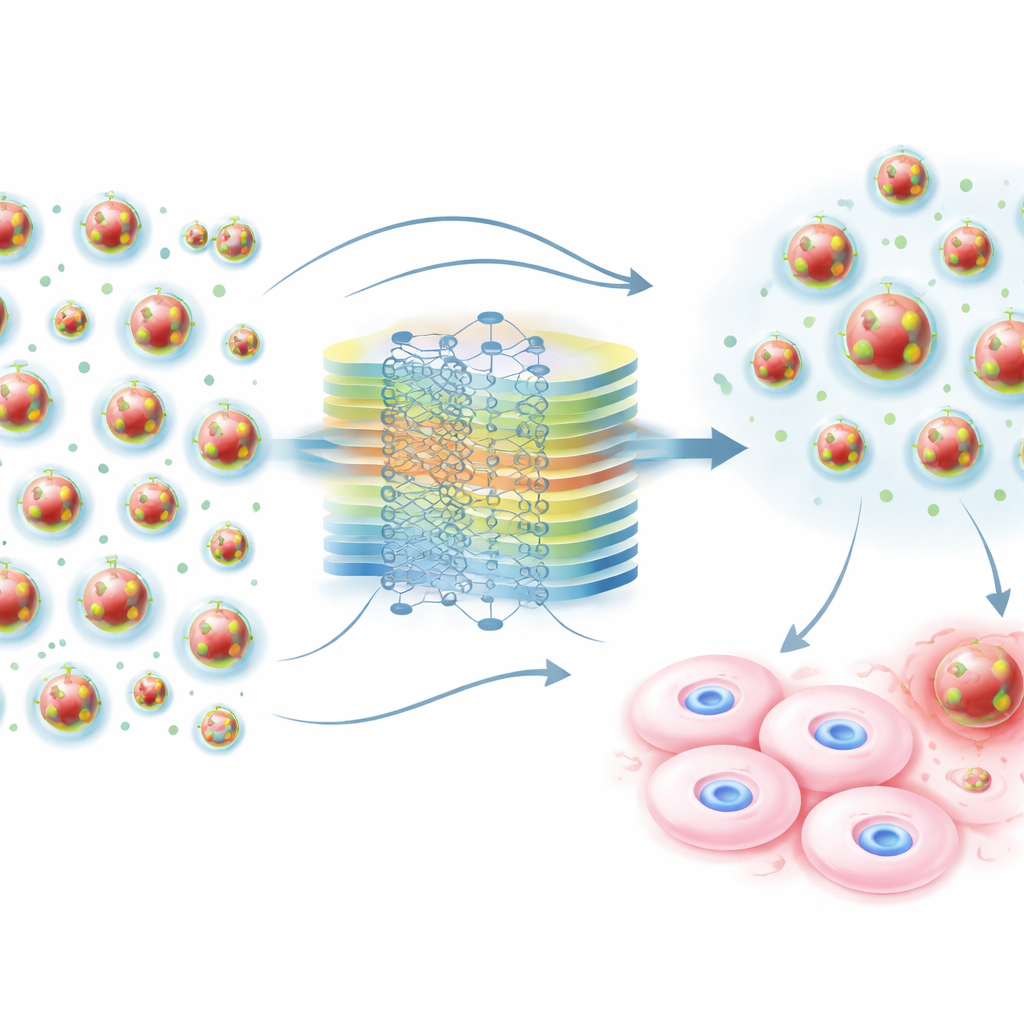
From guesswork to guided design
By blending physics with data‑driven learning, this study delivers practical design guidelines for safer doxorubicin nanocarriers and a reusable blueprint for other drug‑delivery problems. Instead of testing countless formulations in the lab, scientists can now use the model to narrow down to nanocarriers that are likely to keep more than 90 percent of normal cells alive, then focus experiments on this promising “sweet spot.” While the work is based mainly on cell‑culture data and cannot yet capture the full complexity of whole‑body responses, it marks an important step toward rational, computer‑guided design of cancer treatments that strike tumors hard while going easier on the rest of the body.
Citation: Rahdar, A., Fathi-karkan, S. A physics-informed machine learning framework for predicting and mitigating doxorubicin nanocarrier toxicity in normal cells. Sci Rep 16, 10837 (2026). https://doi.org/10.1038/s41598-026-42209-4
Keywords: doxorubicin, nanocarriers, machine learning, drug toxicity, physics-informed models