Clear Sky Science · en
Green synthesis of amorphous Ce-MOFs as efficient adsorbents towards Ofloxacin antibiotics
Why Cleaning Drug-Tainted Water Matters
Traces of antibiotics now routinely show up in rivers, lakes, and even drinking water, largely because medicines taken by people and animals are not fully broken down in the body. One widely used drug, the antibiotic ofloxacin, can linger in the environment, potentially harming aquatic life and fueling antibiotic resistance. This study explores a new, environmentally friendly material that can sponge ofloxacin out of water efficiently, while also being simpler and greener to make than many existing options.
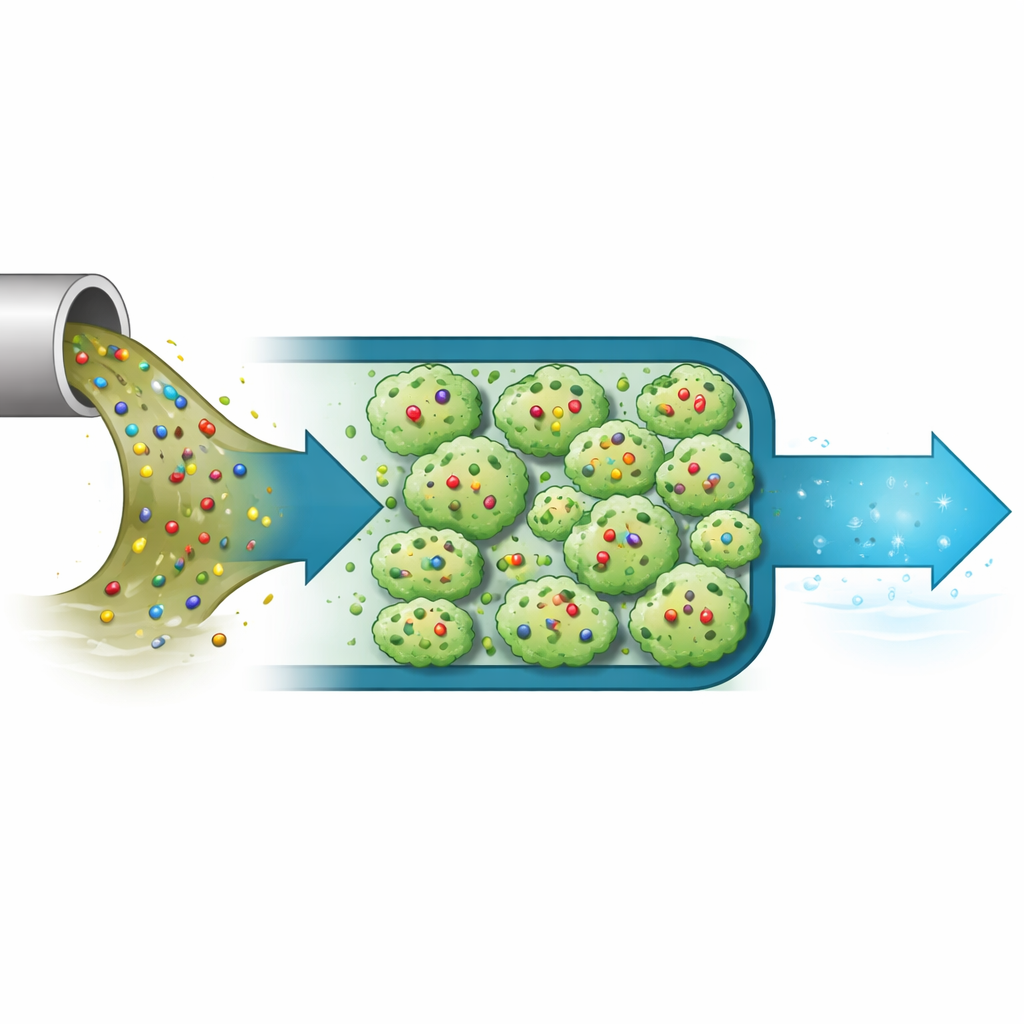
Building Tiny Sponges from Metal and Organic Pieces
The materials at the heart of this work are called metal–organic frameworks, or MOFs. They are built like scaffolds from metal atoms linked by organic molecules, creating an immense amount of internal surface area and tiny pores where contaminants can be trapped. Traditionally, MOFs are carefully ordered crystals made using organic solvents and heat. Here, the researchers focused instead on “amorphous” MOFs based on the element cerium—structures that lack long-range order but still keep the essential metal–organic connections. Amorphous versions can be easier to produce on a large scale, more mechanically robust, and rich in defect sites that can serve as extra docking spots for pollutants.
A Green Recipe Using Only Water
To make these amorphous cerium MOFs, the team developed a room-temperature method that uses water as the only solvent, both for building and activating the material. They mixed a cerium salt with an organic linker in water containing a salt that helps the framework form. By adjusting how they handled undissolved linker and which liquids they used during washing, they created several variations with different pore sizes and surface areas. One key trick was filtering out linker particles that did not dissolve, preventing them from clogging the pores. Tests using X-ray diffraction and infrared spectroscopy confirmed that the products were indeed amorphous but still preserved the basic chemical building blocks found in their crystalline cousins.
How Well the New Sponges Catch Ofloxacin
The researchers then tested each amorphous MOF as an adsorbent—a material that can capture molecules from water—using ofloxacin as a model pollutant. Nitrogen gas adsorption measurements showed that the particles possessed mesopores, pores large enough for ofloxacin molecules to enter and move around. Among the different samples, one labeled Ce-MOF-A-2 struck the best balance between pore size and surface area. Under favorable conditions close to room temperature and a pH around that of neutral water, it soaked up ofloxacin to an experimental capacity of about 139 milligrams per gram of material. Analysis of how fast and how strongly ofloxacin attached suggested a uniform layer of molecules forming on the surface and within the pores, with the process controlled mainly by chemical interactions rather than simple physical trapping. Notably, the maximum capacity calculated from the data slightly exceeded that of a comparable crystalline MOF and was more than twice that of a common benchmark: activated carbon.
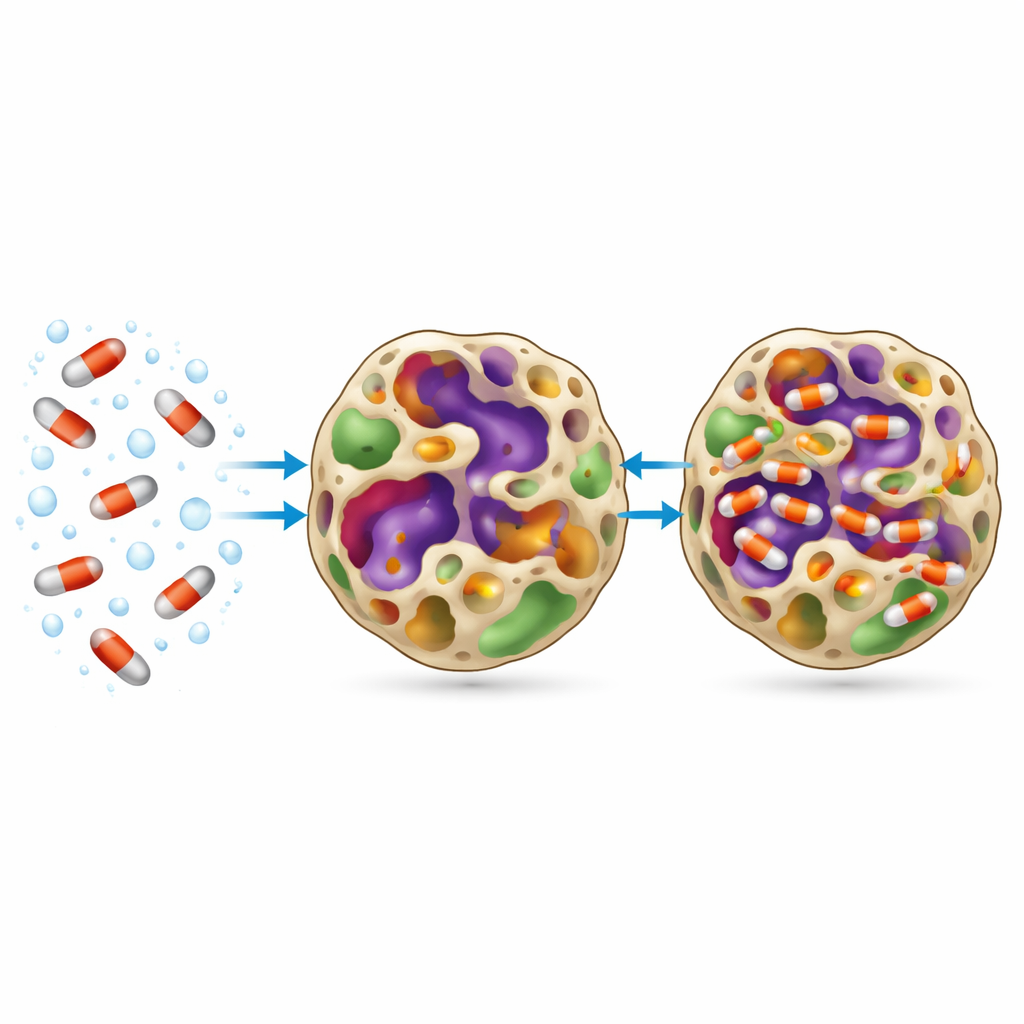
What Controls the Capture and Release
To mimic real wastewater conditions, the team examined how pH, dissolved salts, and temperature influenced performance. The material worked best near neutral pH, where ofloxacin exists in a partly charged form and the MOF surface carries a mild negative charge. Under these circumstances, several forces cooperate: gentle attraction between charges, hydrogen bonding, stacking between ring-like parts of the drug and the organic framework, and simple filling of the pores. Adding common salt boosted removal further by making ofloxacin less comfortable staying in water, driving it toward the MOF. Higher temperatures also increased uptake, indicating that the adsorption process benefits from added thermal energy. The material could be reused several times after washing, with some loss of capacity due to drug molecules that remained wedged in the pores, but its overall structure and thermal stability stayed largely intact.
What This Means for Safer Water
In everyday terms, the study shows that it is possible to make highly effective “molecular sponges” for antibiotic pollution using a simple, water-only process at room temperature. The best-performing amorphous cerium MOF captured more ofloxacin than many crystalline MOFs and far more than activated carbon, while remaining stable and partially reusable. Because the method relies on benign ingredients and avoids harsh conditions, it points toward cost-effective production of advanced filters that could be packed into treatment columns or cartridges. If scaled up, such green-made amorphous MOFs could become powerful tools to help keep pharmaceutical residues out of the water we and other living creatures depend on.
Citation: Molavi, H., Saeedi, S. & Ghorbani, A. Green synthesis of amorphous Ce-MOFs as efficient adsorbents towards Ofloxacin antibiotics. Sci Rep 16, 11322 (2026). https://doi.org/10.1038/s41598-026-42188-6
Keywords: water purification, antibiotic removal, metal-organic frameworks, green synthesis, ofloxacin