Clear Sky Science · en
Direct recycling of end-of-life lithium-ion batteries cathode active materials by hydrothermal route
Why Old Car Batteries Still Matter
Electric cars rely on lithium-ion batteries packed with valuable metals like nickel, manganese, cobalt and lithium. As millions of these batteries reach the end of their lives, the world faces a double challenge: how to avoid mountains of toxic waste while also cutting our dependence on new mining. This study explores a way to "heal" one of the most important parts of these batteries—the cathode material—so it can be used again in new batteries with much less energy, pollution and cost than today’s recycling methods.
From Waste Problem to Resource Loop
The authors frame end-of-life battery packs as a strategic resource rather than trash. Current recycling in Europe is far below what will be needed as electric vehicles scale up, and traditional methods rely on high-temperature smelting or strong acids. These processes recover only some metals, often lose lithium, consume a lot of energy and create extra waste streams. In contrast, the approach studied here—called direct recycling—aims to keep the cathode’s structure mostly intact and simply restore what has been lost during use. This is especially relevant for a widely used cathode known as NMC622, found in commercial electric vehicles such as the Hyundai KONA.
Gently Repairing a Tired Battery Material
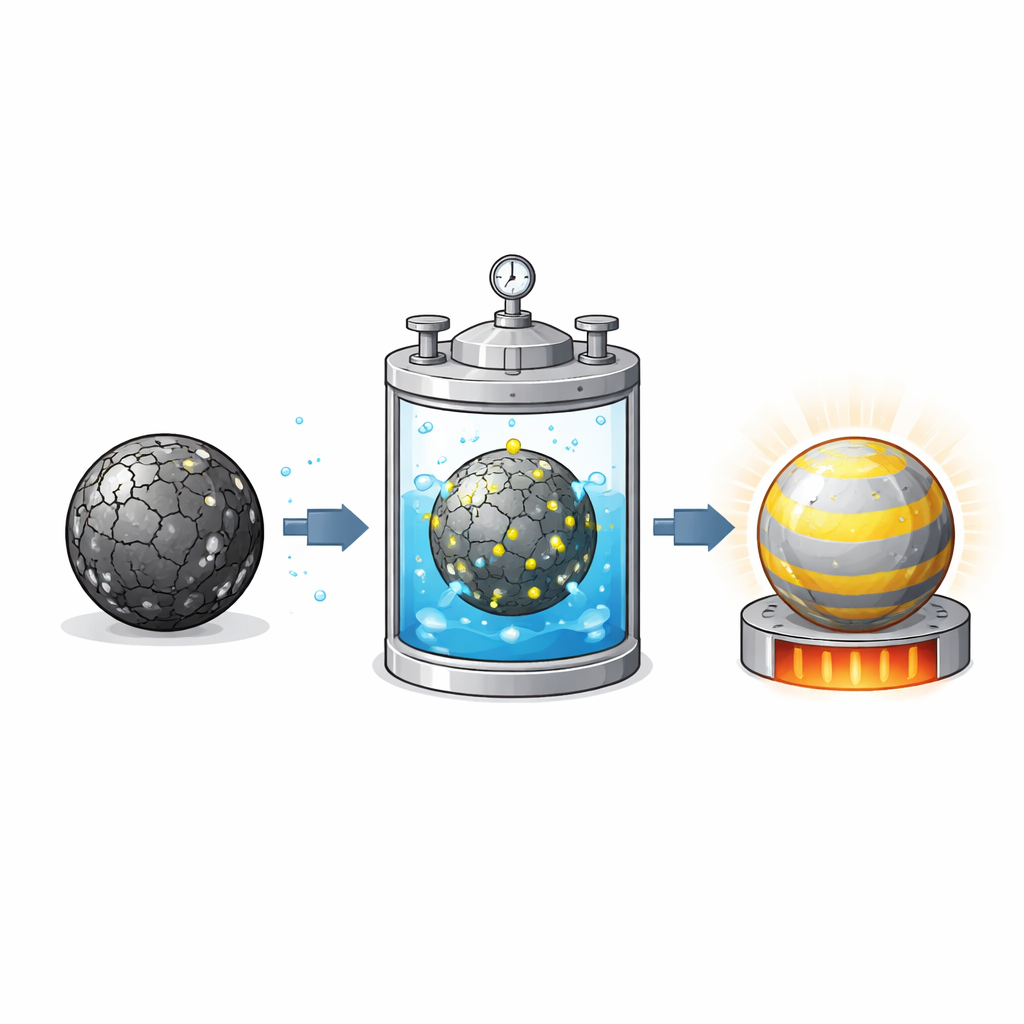
Instead of grinding everything down to basic elements, the team starts from real, degraded car battery cells and carefully separates a clean powder containing only the cathode material. They then use a water-based process called hydrothermal re-lithiation: the powder is mixed with a lithium-rich solution, sealed in a pressure vessel at moderate temperature, and later given a short high-temperature heat treatment. During this sequence, lithium ions flow back into the depleted particles and the crystal structure is tidied up, restoring the material’s ability to store and release energy. By designing a systematic set of experiments, the researchers vary lithium concentration, temperature and reaction time to see which combination best repairs the material.
Finding the Sweet Spot in the Repair Recipe
Careful measurements show that the starting cathode powder is short of lithium and has a damaged surface and extra, unwanted phases that no longer work well in a battery. After treatment, the best samples regain a clean, layered crystal structure very close to that of fresh commercial NMC622. Statistical analysis reveals that lithium concentration and temperature play the biggest roles in successful repair, while the effect of time depends on how much lithium is present. A key finding is that milder conditions—160 °C, a relatively concentrated lithium solution, and a short one-hour treatment—produce a well-ordered material with fewer defects, better lithium mobility and lower electrical resistance than samples treated for longer.
Putting Rebuilt Cathodes to the Test
To see if the repaired powders really behave like new, the authors build coin-sized test cells and compare them directly with a commercial NMC622 cathode. The best regenerated sample delivers discharge capacities close to the fresh material, maintains about 80% of its capacity after 50 charge–discharge cycles, and handles higher charging rates surprisingly well—at the fastest rate tested, it even outperforms the commercial reference. Other repaired samples that experienced harsher treatment show more mixing of atoms inside the crystal and slower lithium movement, which translate into higher internal resistance and faster performance loss. This side-by-side comparison links the processing conditions to microscopic structure and, in turn, to real-world battery behavior.
Cleaner Batteries for a Circular Future
Beyond restoring performance, the hydrothermal repair route offers strong environmental and economic advantages. Because it works at lower temperatures and avoids aggressive acids, it uses only a fraction of the energy of mainstream recycling methods and produces far fewer greenhouse-gas emissions and hazardous wastes. Almost the entire cathode is reused directly, rather than being broken down and rebuilt from scratch. The study concludes that optimized direct recycling of Ni-rich cathodes like NMC622 can plug seamlessly into future battery factories, reducing the need for new mines and helping make electric vehicles truly part of a circular, low-impact energy system.
Citation: Castro, J., Gómez, M., Acebes, P.J. et al. Direct recycling of end-of-life lithium-ion batteries cathode active materials by hydrothermal route. Sci Rep 16, 11594 (2026). https://doi.org/10.1038/s41598-026-41973-7
Keywords: lithium-ion battery recycling, cathode regeneration, hydrothermal relithiation, electric vehicle batteries, circular economy