Clear Sky Science · en
Adsorption of doripenem and meropenem antibiotics on activated carbon derived from snake fruit seeds: single-compound and binary mechanism via experiments and modelling
Turning Fruit Waste into a Water Cleaner
Antibiotic pollution in rivers and lakes may sound remote, but it affects anyone who drinks tap water or worries about drug-resistant infections. This study explores an inventive way to clean such pollutants from water by turning discarded seeds from a tropical snack, snake fruit, into a powerful filter material. The work shows not only how well this material can trap two important hospital antibiotics, doripenem and meropenem, but also explains in detail how the process works and how it could support a more circular, low‑waste economy.
Why Medicines End Up in Our Water
Modern healthcare relies heavily on antibiotics, including last‑resort drugs used when other treatments fail. After use, however, traces of these medicines often pass through patients and enter sewage systems. Conventional treatment plants are not designed to fully remove such complex molecules, so tiny amounts can slip into rivers, groundwater, and even drinking water. Even at low levels, these residues can encourage bacteria to evolve resistance, undermining the life‑saving power of antibiotics. Finding efficient, affordable ways to strip these compounds from water has therefore become an urgent environmental and public‑health priority.
Giving Snake Fruit Seeds a Second Life
Snake fruit, a common crop in Southeast Asia, produces large numbers of inedible seeds that are typically thrown away. The researchers saw these seeds as a free and sustainable starting material for making activated carbon, a highly porous form of carbon widely used in filters. They dried, crushed, and heated the seeds in carefully controlled steps, first in nitrogen and carbon dioxide gases and then with potassium hydroxide. This double activation created a sponge‑like carbon with a huge internal surface area and a network of tiny passages called mesopores. Tests using electron microscopy and gas adsorption confirmed a rough, cavity‑rich surface and a high surface area of about 1,260 square meters per gram—features that make it ideal for catching relatively large antibiotic molecules. 
How the New Filter Grabs Antibiotics
To see how well this carbon can clean water, the team exposed it to solutions containing doripenem and meropenem, either one at a time or both together. In single‑compound tests, each gram of carbon could hold up to 193 milligrams of doripenem and 171 milligrams of meropenem, values that compare favorably with or outperform many other low‑cost sorbents reported in the literature. When both drugs were present, the total uptake dropped, especially for meropenem, revealing that the two antibiotics compete for the same internal sites. More detailed experiments, including X‑ray analysis, nitrogen gas sorption, and infrared spectroscopy, showed that the drugs lodge inside the pores and attach mainly through weak physical forces and hydrogen bonds with oxygen‑rich groups on the carbon surface, rather than forming permanent chemical bonds.
Peering Inside the Pores
To move beyond simple capacity numbers, the researchers applied advanced statistical models that treat adsorption as a process occurring on many identical receptor sites within the carbon. These models allowed them to estimate how many molecules of each antibiotic can crowd onto a single site, how densely these sites are distributed, and how the process changes with temperature. The results indicated that both doripenem and meropenem share the same kinds of sites and that when one occupies a site, the other is effectively excluded—an antagonist relationship that explains the stronger loss of meropenem uptake in mixed solutions. Increasing the water temperature from 30 to 50 degrees Celsius consistently improved adsorption, and calculated energies confirmed that the process is endothermic but still governed by reversible physical attraction. This picture was reinforced by the lack of new chemical peaks in the infrared spectra, supporting a mechanism based on pore filling and non‑permanent interactions. 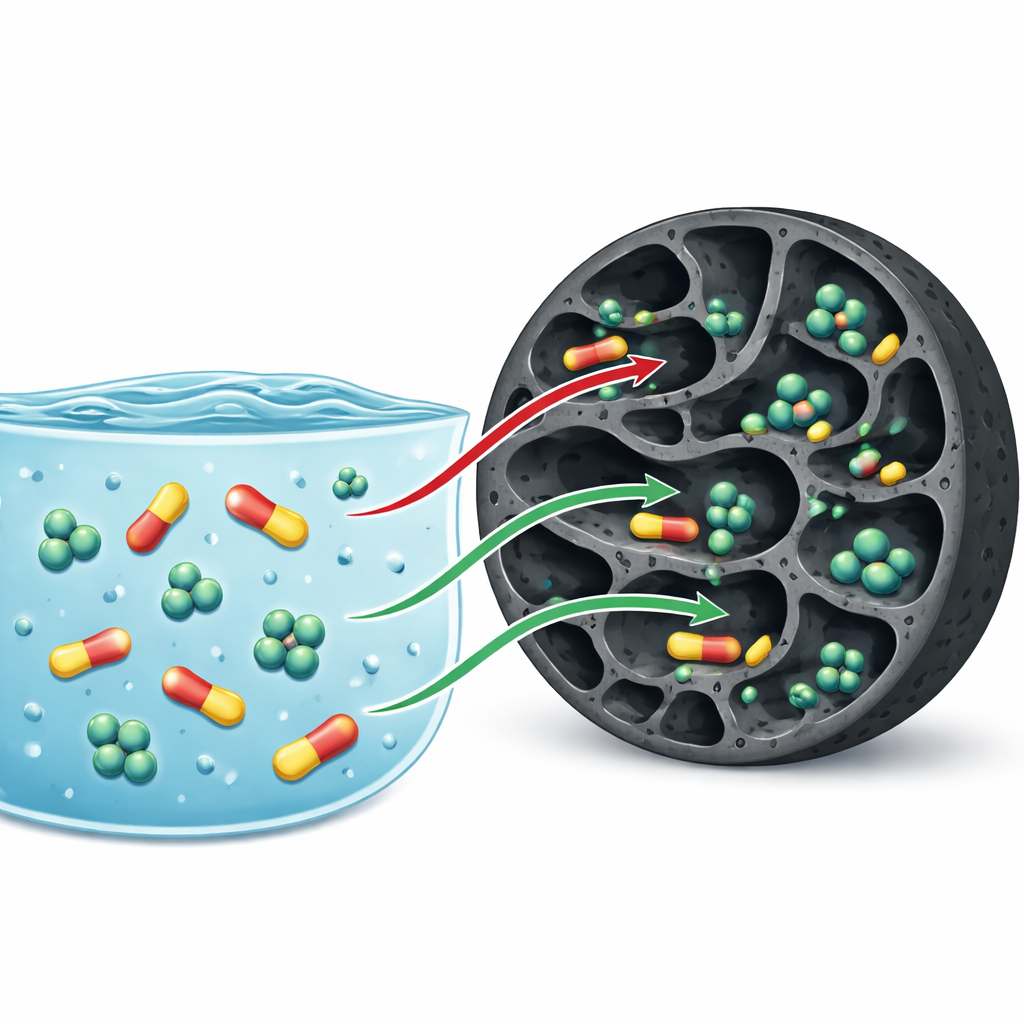
Reusable and Ready for Real Wastewater
Because real filters must be used many times, the team tested how well the carbon could be regenerated. After several adsorption–desorption cycles using mild acid or base rinses, the material still removed more than about 80 percent of the antibiotics, with only a gradual decline due mainly to partial pore blockage. The authors suggest next steps such as testing the material in continuous‑flow columns and in genuine hospital or municipal wastewaters, where many other dissolved substances might compete for the same sites. They also note the broader benefits of converting agricultural waste into a valuable water‑treatment resource that does not compete with food production.
What This Means for Safer Water
For non‑specialists, the key message is that an abundant and otherwise discarded seed can be transformed into a highly effective filter for some of the most important antibiotics in modern medicine. The snake fruit seed‑based carbon captures large amounts of doripenem and meropenem, does so mainly through gentle, reversible forces, and remains stable after repeated use. While further testing under real‑world conditions is needed, this approach illustrates how smart materials design and waste valorization can combine to tackle antibiotic pollution and, in turn, help limit the spread of drug‑resistant microbes in our water systems.
Citation: Alzahrani, E.A., Sellaoui, L., Soetaredjo, F.E. et al. Adsorption of doripenem and meropenem antibiotics on activated carbon derived from snake fruit seeds: single-compound and binary mechanism via experiments and modelling. Sci Rep 16, 13053 (2026). https://doi.org/10.1038/s41598-026-41972-8
Keywords: water purification, activated carbon, antibiotic pollution, pharmaceutical wastewater, biomass waste