Clear Sky Science · en
Linking lipid profile alterations to antibiotic tolerance and natural product synergy in drug-resistant Mycobacterium tuberculosis clinical isolates
Why the Fatty Coat of TB Bacteria Matters
Tuberculosis (TB) is still one of the world’s deadliest infectious diseases, and it is becoming harder to treat as more strains of the TB bacterium develop resistance to antibiotics. This study looks beyond genes to the fatty outer layers of Mycobacterium tuberculosis, asking how subtle changes in this waxy coat help the microbe withstand drugs—and whether natural plant compounds can help common antibiotics break through this defense.
Stubborn Germs with a Waxy Shield
TB bacteria are wrapped in an unusually thick and greasy envelope made largely of lipids—fat-like molecules that can make up more than half of the cell’s dry weight. Instead of a simple wall, they have two membranes with a complex mesh of sugars and specialized lipids in between. Many of these lipids do more than form a barrier: they help the bacteria slip inside immune cells, tamper with immune signals, and build the lung granulomas that wall off infection. This fatty armor also slows the entry of antibiotics, meaning that changes in the envelope’s composition can tilt the balance between drug sensitivity and drug tolerance.
Testing Old Drugs with Plant Helpers
The researchers assembled a panel of TB strains taken from patients, ranging from fully drug-sensitive to multidrug-resistant (MDR), pre-extensively drug-resistant (pXDR), and extensively drug-resistant (XDR). They measured how much of three standard drugs—rifampicin, isoniazid and ethambutol—was needed to stop each strain from growing. They then tested two natural small molecules, piperine (from black pepper) and thymoquinone (from black cumin), both alone and in combination with these antibiotics. While the plant compounds on their own had only modest activity, combining them with rifampicin consistently made rifampicin far more potent against every strain, and in some cases greatly boosted isoniazid as well. In contrast, ethambutol did not benefit from these combinations.
Reading the Bacteria’s Lipid Fingerprint
To connect these drug responses to the structure of the bacterial envelope, the team used high-resolution liquid chromatography and mass spectrometry to chart the lipid profiles of each strain. Statistical analysis showed that clinical isolates—especially MDR, pXDR and XDR strains—clustered separately from the standard laboratory strain, revealing that real-world TB bacteria remodel their lipids in distinctive ways. Drug-resistant strains tended to replace inner-membrane lipids with long fatty tails by versions with shorter tails, which would normally thin and loosen the membrane. They compensated by making many of those tails more saturated, a change that stiffens the membrane and helps preserve its barrier function. Highly resistant strains also stockpiled energy-rich storage lipids and, in some cases, accumulated iron-bound molecules used to scavenge this essential metal.
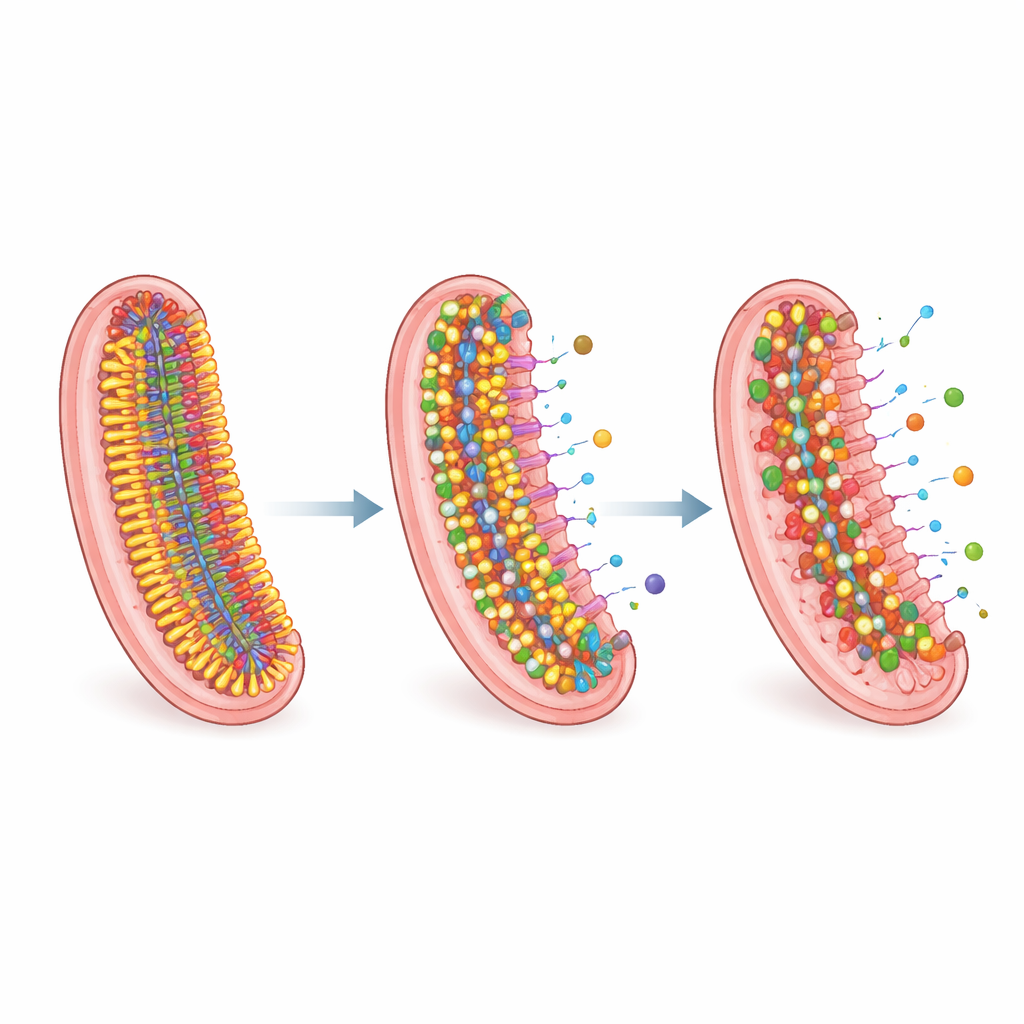
Deep Remodeling of the Waxy Armor
Particular lipid families stood out. Molecules called acylated phosphatidylinositol mannosides, which help organize the inner membrane and link it to outer structures, shifted toward shorter and more saturated forms in resistant strains. Some highly resistant isolates showed a striking loss of outer-envelope lipids known as phthiocerol dimycocerosates and related molecules, while simultaneously building up triacylglycerols, neutral fats that can act as energy stores and chemical sinks. Many clinical strains also lacked the very longest mycolic acids—the giant waxy molecules that contribute to the envelope’s toughness—consistent with strain-specific adjustments in how these complex lipids are built. Together, these changes paint a picture of TB bacteria fine‑tuning their membranes to balance energy cost, structural integrity and resistance to antibiotic entry.
How Plant Compounds Tip the Balance
The synergy between plant compounds and antibiotics appears to exploit these carefully tuned defenses. Piperine is known to inhibit a key drug efflux pump that TB uses to expel antibiotics, likely increasing the amount of rifampicin and isoniazid that stays inside the cell, and may also interact directly with the bacterial enzyme targeted by rifampicin. Thymoquinone, by contrast, drains the bacteria’s energy stores by depleting ATP and NAD, undermining their ability to maintain lipid balance, run efflux pumps and repair damage from rifampicin. When combined with standard drugs, these natural molecules help overwhelm the remodeled lipid shield that supports drug resistance.
What This Means for Future TB Treatment
To a non-specialist, the main lesson is that TB’s resistance is not just a story of mutated genes, but also of a shape-shifting fatty armor that can be rewired to keep drugs out and the microbe alive. This study shows that resistant clinical strains of M. tuberculosis carry distinctive lipid signatures and that carefully chosen natural compounds can weaken the bacterium’s defenses, restoring or boosting the effect of existing antibiotics, particularly rifampicin. Understanding and targeting these lipid-based adaptations may open new paths to treating even the toughest TB infections using smarter combinations of known drugs and safe, lipid‑modulating helpers.
Citation: Zabost, A., Sawicki, R., Jankowski, G. et al. Linking lipid profile alterations to antibiotic tolerance and natural product synergy in drug-resistant Mycobacterium tuberculosis clinical isolates. Sci Rep 16, 11459 (2026). https://doi.org/10.1038/s41598-026-41967-5
Keywords: tuberculosis, antibiotic resistance, bacterial lipids, natural product adjuvants, rifampicin synergy