Clear Sky Science · en
Analysis of the resistance of small peptides from Periplaneta americana to H2O2-induced apoptosis in KGN Cells based on miRNA-seq
Why this matters for women’s health
Many women today face fertility problems linked to their ovaries aging or failing earlier than expected. One key culprit is the slow loss of the tiny support cells that surround and nourish each egg in the ovary. This study explores an unusual potential ally against that loss: small protein fragments extracted from the American cockroach. The researchers show that these peptides can protect human ovarian support cells from damage caused by oxidative stress and begin to map how they work at the level of gene regulation.
How cell stress threatens fertility
Inside the ovary, egg cells rely on a ring of helper cells called granulosa cells to provide nutrients, hormones, and protection. When these granulosa cells die too early, follicles shrink and disappear, contributing to premature ovarian failure and reduced fertility. One major driver of this unwanted cell death is oxidative stress, a state where aggressive oxygen-containing molecules build up faster than the cell’s defenses can handle. In the lab, the team mimicked this situation by exposing human granulosa-like KGN cells to hydrogen peroxide, a compound that boosts reactive oxygen species and reliably triggers programmed cell death.
Cockroach peptides as unexpected protectors
Periplaneta americana, better known as the American cockroach, is usually thought of as a pest, yet extracts from this insect have long been used in traditional medicine for healing wounds and calming inflammation. In this work, the authors focused on a highly purified fraction rich in very small peptides, called SPPA. They first confirmed that a specific dose of hydrogen peroxide could reduce KGN cell survival to around 60%, a level that signals strong stress without wiping out the entire culture. When they added SPPA after this oxidative insult, several classic markers of damage shifted in a favorable direction: levels of harmful molecules such as reactive oxygen species, nitric oxide, and malondialdehyde dropped, while activity of the protective enzyme superoxide dismutase rose. Microscopy showed fewer cells with shrunken, brightly condensed nuclei, and molecular tests revealed that SPPA lowered pro-death proteins (like Bax and activated caspase-3) and restored the balance toward the survival protein Bcl-2.
Peering into tiny RNA switches
To move beyond these broad protective effects, the researchers examined microRNAs—short, non-coding RNA fragments that fine-tune which genes are turned on or off. Using high-throughput sequencing, they compared three situations: untreated cells, cells stressed by hydrogen peroxide, and stressed cells subsequently treated with SPPA. They identified 162 microRNAs whose activity shifted across these conditions, and then focused on 13 that changed in opposite directions during injury and repair. Some of these microRNAs were boosted by oxidative stress but dialed back after SPPA treatment, while others were suppressed by stress and revived with SPPA. By computationally predicting which genes these 13 microRNAs might regulate, the team found more than 3,500 candidate targets enriched in pathways related to cell death, metabolism, and the function of cellular compartments such as lysosomes.
Key molecular players in the rescue
Among the many microRNAs, three stood out as likely hubs in the protective network: miR-103a-3p, NovelmiRNA-214, and NovelmiRNA-383. Oxidative stress pushed NovelmiRNA-214 and NovelmiRNA-383 upward, in line with a pro-death role, while SPPA brought their levels back down. By contrast, miR-103a-3p dropped under stress but rebounded with SPPA, consistent with a survival-supporting function. Further analysis suggested that these and related microRNAs collectively touch genes involved in apoptosis, cell cycle control, mitochondrial function, and antioxidant defenses. The authors verified the sequencing patterns for five selected microRNAs using quantitative PCR, confirming that the shifts they saw were robust and reproducible.
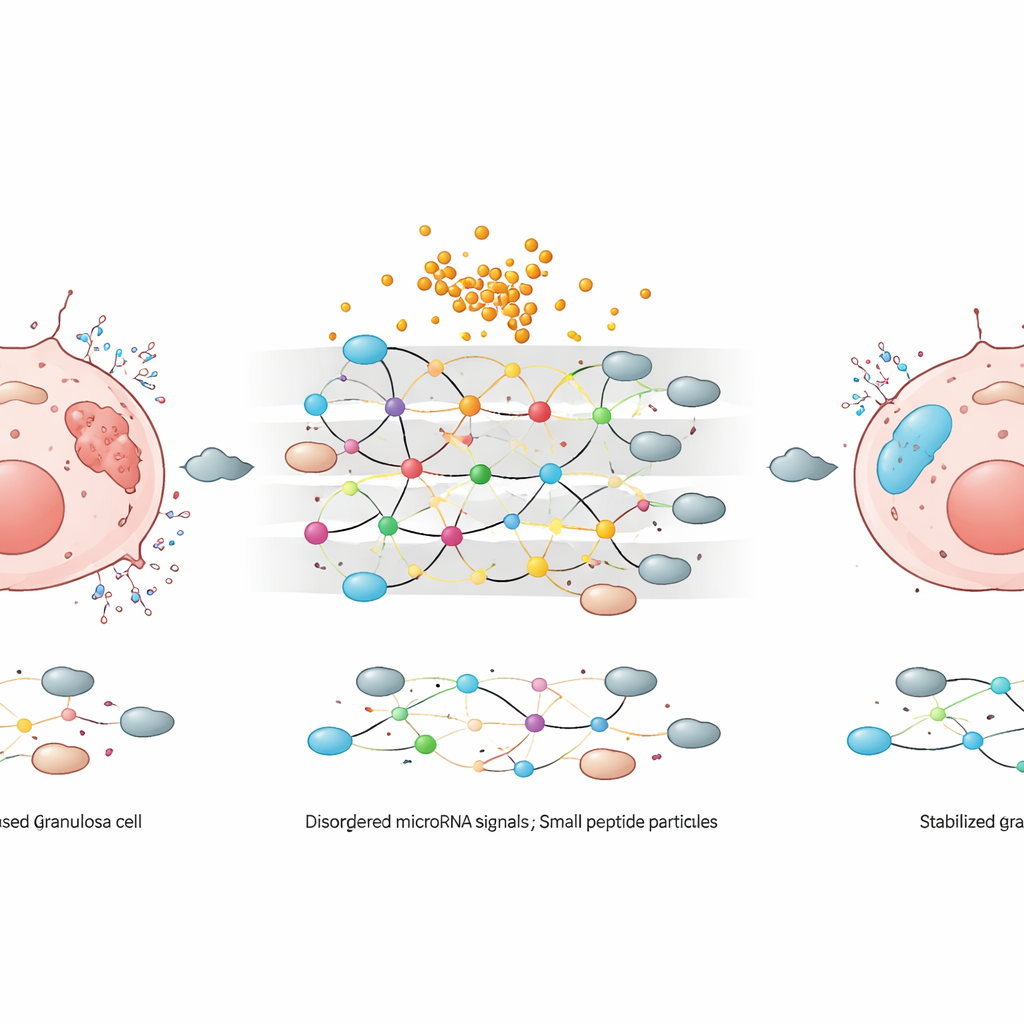
What this could mean for future treatments
Putting the pieces together, the study proposes a model in which hydrogen peroxide–driven oxidative stress pushes ovarian granulosa cells toward death, while SPPA not only reduces the immediate chemical damage but also rewires a network of microRNAs and their target genes to favor survival, controlled self-cleaning of damaged components, and continued cell growth. Although the work is still at the cell-culture stage and cockroach-derived peptides will require careful safety and clinical evaluation, it points to a surprising new direction for protecting ovarian function. By targeting these microRNA-controlled switches—especially those centered on miR-103a-3p, NovelmiRNA-214, and NovelmiRNA-383—future therapies might help safeguard fertility in women whose ovaries are threatened by oxidative stress, chemotherapy, or other damaging influences.
Citation: Xu, L., Jiang, R., Su, J. et al. Analysis of the resistance of small peptides from Periplaneta americana to H2O2-induced apoptosis in KGN Cells based on miRNA-seq. Sci Rep 16, 11500 (2026). https://doi.org/10.1038/s41598-026-41839-y
Keywords: ovarian granulosa cells, oxidative stress, microRNA, apoptosis, fertility preservation