Clear Sky Science · en
Carrot extracellular nanovesicles as carotenoid carriers in an in vitro macular degeneration model
Carrots and Eye Health
Most people have heard that carrots are good for your eyes, but translating that folk wisdom into modern medicine is not straightforward. The pigments in carrots that may protect vision are fragile, hard for the body to absorb, and easily damaged by light and heat. This study explores a clever way to turn ordinary carrot material into tiny natural capsules that could safely ferry protective pigments to vulnerable eye cells involved in age-related macular degeneration, a leading cause of vision loss in older adults.
Tiny Natural Packages from Plants
The researchers focused on microscopic, bubble-like particles naturally produced by plants, known as nanovesicles. These structures are made of lipids, much like cell membranes, and can carry fats, pigments, and other useful molecules. The team isolated such vesicles from two carrot sources: fresh carrot juice and laboratory-grown carrot callus, which is a mass of undifferentiated carrot cells. Using high-speed spinning and density gradients, they separated out these vesicles and confirmed that they were small, roughly spherical particles with properties similar to those of animal cell vesicles already being studied as drug-delivery tools.

Loading Carrot Pigments into Vesicles
Carrots are rich in carotenoids, a family of colorful pigments that includes lutein and zeaxanthin, which naturally accumulate in the center of the human retina. These particular pigments help filter harmful blue light and neutralize reactive oxygen species, both of which are important for slowing damage in age-related macular degeneration. The team measured the native pigment content of their vesicles and found that juice-derived vesicles carried at least 12 carotenoids, including several forms of beta-carotene, lutein, and zeaxanthin. By contrast, vesicles from callus cells contained essentially no detectable carotenoids on their own, making them an almost “blank” carrier.
To turn both vesicle types into targeted pigment carriers, the researchers loaded them with a controlled mixture of lutein and zeaxanthin. They compared simple soaking, which relies on passive diffusion, with electroporation, a technique that briefly opens pores in the vesicle membrane using electrical pulses. Electroporation at specific settings (200 mV, 50 μF) reached encapsulation efficiencies up to about 90% for zeaxanthin and over 50% for lutein in juice-derived vesicles, with similarly high loading in callus-derived vesicles. These results show that plant vesicles can be efficiently packed with delicate, oily pigments that are otherwise hard to handle.
Testing Protection for Eye Cells
The key question was whether these pigment-loaded vesicles could actually shield eye cells from damage. The team used ARPE-19 cells, a laboratory model that mimics the retinal pigment epithelium, a layer critical for nourishing the light-sensing cells in the eye. They exposed these cells to hydrogen peroxide to mimic oxidative stress, then compared several treatments: free lutein/zeaxanthin, unloaded vesicles, and pigment-loaded vesicles from both juice and callus. Cell survival was measured after 24 hours.
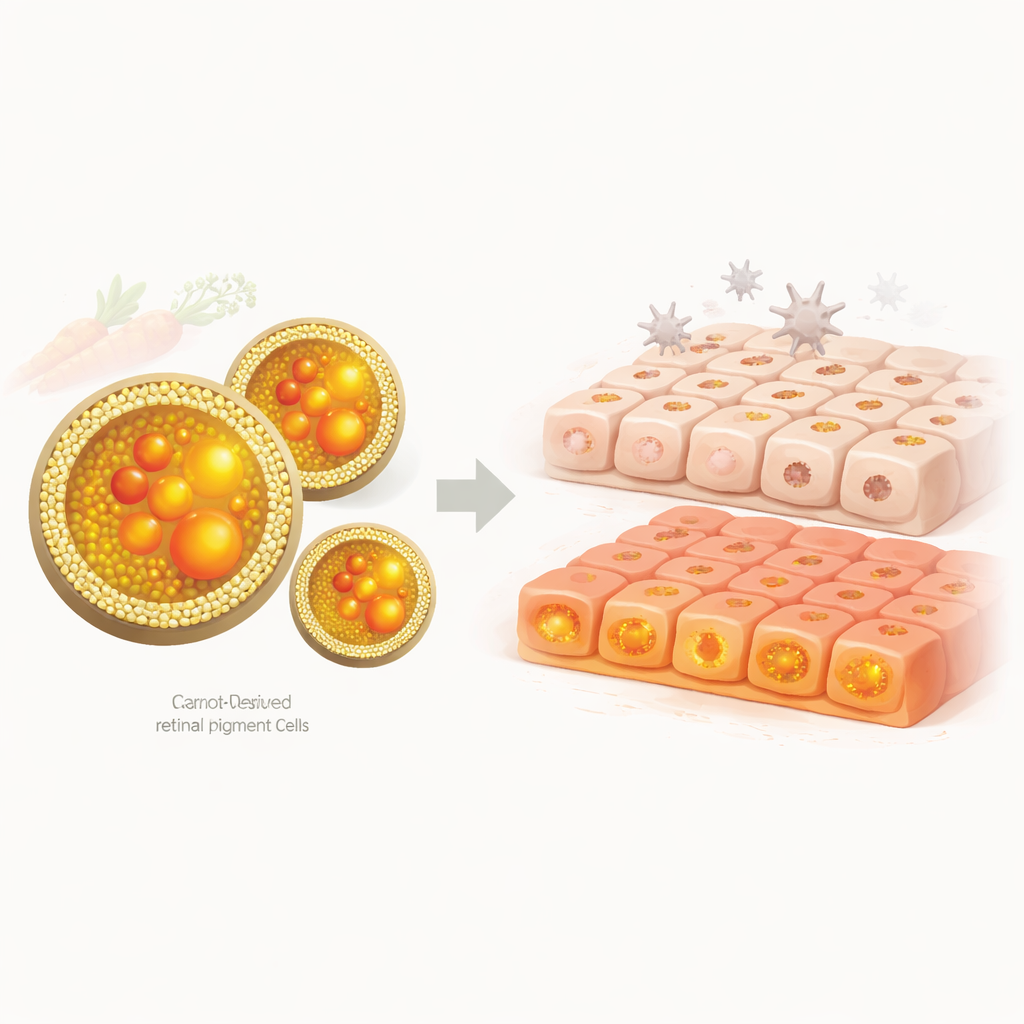
Callus-derived vesicles loaded with lutein and zeaxanthin provided the most impressive protection, maintaining over 95% cell viability under oxidative stress. This was significantly better than free pigments alone and better than pigment-loaded vesicles from juice. Interestingly, unloaded juice vesicles appeared to help as well, likely because they naturally carry beta-carotene and other compounds, although their strong orange color may have interfered with the optical readout of viability. In contrast, unloaded callus vesicles seemed to worsen damage, possibly by shuttling the oxidizing agent into cells, underscoring how important the cargo is for determining vesicle behavior.
What This Could Mean for Future Treatments
To a non-specialist, the takeaway is that the researchers turned carrots into a dual-purpose material: both the source of protective pigments and the source of tiny, biocompatible delivery capsules. They showed that these nanovesicles can be isolated from both regular carrot juice and from lab-grown carrot tissue, loaded efficiently with eye-protective pigments, and used to keep model eye cells alive under stressful conditions. Callus-derived vesicles, which start out pigment-free and seem to release their cargo more readily, emerged as especially promising carriers. While more work is needed in animals and humans, these plant-based nanocapsules offer a potentially safe, scalable, food-derived way to deliver fragile nutrients or drugs not only to the eye but possibly to other sensitive tissues in the brain and heart as well.
Citation: Tapia-Aguayo, A., Cisneros-Pardo, A., De los Santos-González, B.E. et al. Carrot extracellular nanovesicles as carotenoid carriers in an in vitro macular degeneration model. Sci Rep 16, 12603 (2026). https://doi.org/10.1038/s41598-026-41792-w
Keywords: age-related macular degeneration, carotenoids, nanovesicles, drug delivery, ocular health